Abstract
The internal dosimetry schema of the Medical Internal Radiation Dose (MIRD) Committee of the Society of Nuclear Medicine has provided a broad framework for assessment of the absorbed dose to whole organs, tissue subregions, voxelized tissue structures, and individual cellular compartments for use in both diagnostic and therapeutic nuclear medicine. The schema was originally published in 1968, revised in 1976, and republished in didactic form with comprehensive examples as the MIRD primer in 1988 and 1991. The International Commission on Radiological Protection (ICRP) is an organization that also supplies dosimetric models and technical data, for use in providing recommendations for limits on ionizing radiation exposure to workers and members of the general public. The ICRP has developed a dosimetry schema similar to that of the MIRD Committee but has used different terminology and symbols for fundamental quantities such as the absorbed fraction, specific absorbed fraction, and various dose coefficients. The MIRD Committee objectives for this pamphlet are 3-fold: to restate its schema for assessment of absorbed dose in a manner consistent with the needs of both the nuclear medicine and the radiation protection communities, with the goal of standardizing nomenclature; to formally adopt the dosimetry quantities equivalent dose and effective dose for use in comparative evaluations of potential risks of radiation-induced stochastic effects to patients after nuclear medicine procedures; and to discuss the need to identify dosimetry quantities based on absorbed dose that address deterministic effects relevant to targeted radionuclide therapy.
In 1976, the Medical Internal Radiation Dose (MIRD) Committee of the Society of Nuclear Medicine issued MIRD Pamphlet No. 1, Revised, as a supplement to The Journal of Nuclear Medicine (1). The purpose of that document was to update the original MIRD schema issued in 1968 (2,3). The MIRD schema, with examples, was published in didactic format in 1988 and later in 1991 as the MIRD Primer (4). Since that time, the MIRD schema has provided a broad framework for the assessment of absorbed dose to whole organs, tissue subregions, voxelized tissue structures, and individual cellular compartments from internally deposited radionuclides (5,6). At the same time, the International Commission on Radiological Protection (ICRP), whose mission is to establish guidelines regarding accidental, occupational, and patient exposures, formulated an almost identical dosimetry schema that includes physical quantities such as absorbed dose. In addition, the ICRP defined the radiation protection quantities equivalent dose and effective dose to address the relative biological effectiveness (RBE) of all emitted radiations and the differential radiosensitivity of organs to radiation-induced stochastic effects (cancer induction due to mutation of somatic cells or heritable effects due to mutations of germ cells) (7,8). Fundamentally, the computation of absorbed dose in both the MIRD and the ICRP systems is similar, as each uses the concepts of absorbed fraction, specific absorbed fraction, source and target tissue regions, reference computational phantoms, and compartmental models describing biokinetic distributions of activity in the human body. These dosimetry schema differ more in notation than in substance. The purpose of this MIRD pamphlet is 3-fold. First, the Committee restates the MIRD schema for assessment of absorbed dose in a manner consistent with the needs of both the nuclear medicine and radiation protection communities with the goal of standardizing nomenclature. Second, the Committee adopts the dosimetry quantities equivalent dose and effective dose for use in comparative evaluations of potential risks of radiation-induced stochastic effects to patients after nuclear medicine procedures. Finally, the Committee highlights the need for dosimetry quantities to address deterministic effects (due to cell death or impairment of organ function after high absorbed doses and dose rates) associated with targeted radionuclide therapy.
PHYSICAL QUANTITIES
Mean Absorbed Dose Rate
The absorbed dose  is defined as the mean energy imparted to target tissue (or region) rT per unit tissue mass (9). The time-dependent rate at which the absorbed dose is delivered
is defined as the mean energy imparted to target tissue (or region) rT per unit tissue mass (9). The time-dependent rate at which the absorbed dose is delivered 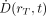 to target tissue rT within a patient from a radioactive material distributed uniformly within source tissue rS at time t after administration is given as:
to target tissue rT within a patient from a radioactive material distributed uniformly within source tissue rS at time t after administration is given as: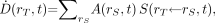 Eq. 1where
Eq. 1where 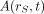 is the time-dependent activity of the radiopharmaceutical in source tissue rS, and
is the time-dependent activity of the radiopharmaceutical in source tissue rS, and  is the radionuclide-specific quantity representing the mean absorbed dose rate to target tissue rT at time t after administration per unit activity present in source tissue rS. S is characteristic of the radionuclide and the age- and sex-specific anatomic model chosen to represent the patient or tissue of interest. The value of S may be based on pre-constructed whole-body computational phantoms representing reference individuals of a given age, sex, total-body mass, and standing height (10,11). Alternatively, the model may be based on segmented images of subject anatomy from either CT or MR images (12). Furthermore, the source and target regions rS and rT, respectively, are those defined within the anatomic model and may represent the full range of configurations including whole organs, suborgan tissue regions, voxels from SPECT or PET images, tumors and cell clusters, individual cells, or cell components (5,6,13). If an absorbed dose distribution is desired as related to voxels defined in a SPECT or PET image, then the MIRD schema is applied at the voxel level, and a dose volume histogram can be derived using the calculated mean absorbed dose per voxel for all voxels in the segmented region of the organ of interest (5).
is the radionuclide-specific quantity representing the mean absorbed dose rate to target tissue rT at time t after administration per unit activity present in source tissue rS. S is characteristic of the radionuclide and the age- and sex-specific anatomic model chosen to represent the patient or tissue of interest. The value of S may be based on pre-constructed whole-body computational phantoms representing reference individuals of a given age, sex, total-body mass, and standing height (10,11). Alternatively, the model may be based on segmented images of subject anatomy from either CT or MR images (12). Furthermore, the source and target regions rS and rT, respectively, are those defined within the anatomic model and may represent the full range of configurations including whole organs, suborgan tissue regions, voxels from SPECT or PET images, tumors and cell clusters, individual cells, or cell components (5,6,13). If an absorbed dose distribution is desired as related to voxels defined in a SPECT or PET image, then the MIRD schema is applied at the voxel level, and a dose volume histogram can be derived using the calculated mean absorbed dose per voxel for all voxels in the segmented region of the organ of interest (5).
Mean Absorbed Dose: Time-Dependent Formulation
The mean absorbed dose 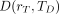 to target tissue rT over a defined dose-integration period TD after administration of the radioactive material to the subject is given as:
to target tissue rT over a defined dose-integration period TD after administration of the radioactive material to the subject is given as: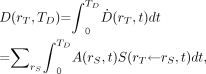 Eq. 2where TD is commonly taken to be infinity, as radionuclides of general use in nuclear medicine have relatively short physical half-lives. In radiologic protection, the dose-integration period is termed the dose-commitment period and is standardized to 50 y for adults (who are assumed to be at a reference age of 20 y at the time of exposure) or a variable time to age 70 y for those exposed as infants, children, or adolescents (8). The unit of the absorbed dose is the joule per kilogram (J kg−1), given the special name gray (Gy).
Eq. 2where TD is commonly taken to be infinity, as radionuclides of general use in nuclear medicine have relatively short physical half-lives. In radiologic protection, the dose-integration period is termed the dose-commitment period and is standardized to 50 y for adults (who are assumed to be at a reference age of 20 y at the time of exposure) or a variable time to age 70 y for those exposed as infants, children, or adolescents (8). The unit of the absorbed dose is the joule per kilogram (J kg−1), given the special name gray (Gy).
If 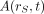 is normalized to a unit administered activity A0 and denoted as
is normalized to a unit administered activity A0 and denoted as 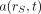 , then the absorbed dose coefficient
, then the absorbed dose coefficient 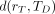 in target tissue rT is given as:
in target tissue rT is given as: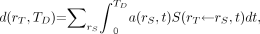 Eq. 3where
Eq. 3where 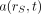 is the fraction of the administered activity in the source tissues rS at time t after administration. In both the ICRP and the MIRD systems, the time-dependent activity in the source tissue is obtained by numeric solution of a set of first-order coupled differential equations defined by compartment models for all organs and suborgan tissues of interest. Alternatively, the time-dependent activity in source tissues of the patient may be obtained directly via quantitative imaging, including planar imaging, SPECT, and PET, or by tissue sampling (e.g., biopsy, blood, or urine collection).
is the fraction of the administered activity in the source tissues rS at time t after administration. In both the ICRP and the MIRD systems, the time-dependent activity in the source tissue is obtained by numeric solution of a set of first-order coupled differential equations defined by compartment models for all organs and suborgan tissues of interest. Alternatively, the time-dependent activity in source tissues of the patient may be obtained directly via quantitative imaging, including planar imaging, SPECT, and PET, or by tissue sampling (e.g., biopsy, blood, or urine collection).
The quantity S is specific to the radionuclide and to the computational phantom defining the spatial relationship and tissue compositions of rS and rT and their intervening tissues in the reference individual or tissue model. S is given as: Eq. 4where Ei is the mean (or individual) energy of the ith nuclear transition, Yi is number of ith nuclear transitions per nuclear transformation (14), Δi is their product (mean energy of the ith transition per nuclear transformation),
Eq. 4where Ei is the mean (or individual) energy of the ith nuclear transition, Yi is number of ith nuclear transitions per nuclear transformation (14), Δi is their product (mean energy of the ith transition per nuclear transformation),  is the absorbed fraction (defined as the fraction of radiation energy Ei emitted within the source tissue rS at time t that is absorbed in the target tissue rT), and
is the absorbed fraction (defined as the fraction of radiation energy Ei emitted within the source tissue rS at time t that is absorbed in the target tissue rT), and  is the time-dependent mass of the target tissue rT in the reference individual. For β-particles whose range in tissue is short relative to the dimensions of the target tissue (i.e., absorbed fraction, ∼1), Ei is typically taken as the mean value of the energy spectrum. If the absorbed fraction varies significantly across the range of spectral energies, then the summation in Equation 4 is replaced by an integral over the β-energy spectrum (Appendix). This circumstance is particularly relevant to cellular dosimetry (13). The specific absorbed fraction
is the time-dependent mass of the target tissue rT in the reference individual. For β-particles whose range in tissue is short relative to the dimensions of the target tissue (i.e., absorbed fraction, ∼1), Ei is typically taken as the mean value of the energy spectrum. If the absorbed fraction varies significantly across the range of spectral energies, then the summation in Equation 4 is replaced by an integral over the β-energy spectrum (Appendix). This circumstance is particularly relevant to cellular dosimetry (13). The specific absorbed fraction 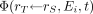 is defined as the ratio of the absorbed fraction and the target mass:
is defined as the ratio of the absorbed fraction and the target mass: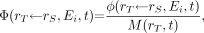 Eq. 5
Eq. 5
such that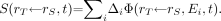 Eq. 6
Eq. 6
Mean Absorbed Dose: Time-Independent Formulation
Examples of situations in which the time dependency of S must be maintained include assessment of the absorbed dose to tumor regions whose mass varies (increases or decreases) over the period of irradiation and assessment of lifetime mean organ doses in subjects exposed to long-lived radionuclides during childhood. In most instances, the time dependency of S may be neglected, as when the source and target masses remain constant over the period of irradiation. Under such conditions, Equation 2 may be reduced to the following time-independent form: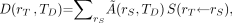 Eq. 7where
Eq. 7where 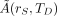 is the time-integrated activity (or total number of nuclear transformations) in source tissue rS over dose-integration period TD such that
is the time-integrated activity (or total number of nuclear transformations) in source tissue rS over dose-integration period TD such that 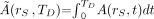 . Whereas S is defined as a ratio of time-dependent rates in Equation 1, it is defined as a ratio of integral quantities in Equation 7 (absorbed dose in rT per nuclear transformation in rS). Furthermore, Equation 3 may be simplified to:
. Whereas S is defined as a ratio of time-dependent rates in Equation 1, it is defined as a ratio of integral quantities in Equation 7 (absorbed dose in rT per nuclear transformation in rS). Furthermore, Equation 3 may be simplified to: Eq. 8where
Eq. 8where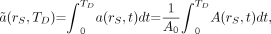 Eq. 9and
Eq. 9and  is the time-integrated activity coefficient. In earlier versions of the MIRD schema,
is the time-integrated activity coefficient. In earlier versions of the MIRD schema,  was termed the residence time τ and given in units of time (i.e., seconds). The value of
was termed the residence time τ and given in units of time (i.e., seconds). The value of 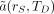 represents the cumulative number of nuclear transformations (Bq s) occurring in source tissue rS over a dose-integration period TD per unit administered activity A0 (Bq). A comparison of dosimetric quantities within the previous version of the MIRD schema, and those of the ICRP, are given in Table 1.
represents the cumulative number of nuclear transformations (Bq s) occurring in source tissue rS over a dose-integration period TD per unit administered activity A0 (Bq). A comparison of dosimetric quantities within the previous version of the MIRD schema, and those of the ICRP, are given in Table 1.
Quantities, Parameters, Symbols, and Units Used in the MIRD and ICRP Dosimetry Schema (Listed in Order of Appearance in Equations 1–17)
QUANTITIES RELEVANT TO THE RISK OF STOCHASTIC EFFECTS
Equivalent Dose
The equivalent dose is a radiation protection quantity defined by the ICRP (7,8) and used to relate absorbed dose to the probability of stochastic health effects in a population exposed to radionuclides or radiation fields, which include a mixture of radiation particle types of varying linear energy transfer (LET). Stochastic effects include biologic outcomes of radiation exposure such as cancer or heritable disease. The equivalent dose 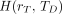 is defined as:
is defined as: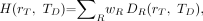 Eq. 10where wR is the radiation-weighting factor for radiation type R, and
Eq. 10where wR is the radiation-weighting factor for radiation type R, and  is the contribution of radiation type R to the mean absorbed dose in target tissue rT. Current ICRP-recommended values of wR are 1.0 for photons, electrons, positrons, and β-particles and 20 for α-particles (8). Some radionuclides used in nuclear medicine (e.g., 99mTc, 123I, 125I, and 201Tl) emit Auger electrons. The radiation-weighting factors of these low-energy electrons, based on their RBE, may be higher than 1.0 when the radionuclide is incorporated into the DNA of the cell nucleus. The ICRP does not give specific recommendations on the value of wR for Auger electron emitters but recommends that its value be determined on a case-by-case basis (7). Guidance is, however, given in the American Association of Physicists in Medicine (AAPM) report no. 49 (15). The AAPM recognized that the RBE (albeit for deterministic effects) caused by Auger electrons emitted by DNA-incorporated radionuclides is similar to that seen for high-LET α-particles and, thus, has recommended a radiation-weighting factor of 20 for this localization of Auger emitters. Furthermore, as there is a linear dependence of the RBE on the subcellular distribution of the Auger electron emitter (16), the AAPM has recommended a linear weighting of the Auger electron contribution to the equivalent dose that is dependent on that subcellular distribution (15). Although not relevant to nuclear medicine, wR is defined for neutrons as an energy-dependent function ranging from 2.5 to 20.7, and the wR for protons is assigned a single value of 2 (8). The unit for equivalent dose is the J kg−1, with the special name sievert (Sv).
is the contribution of radiation type R to the mean absorbed dose in target tissue rT. Current ICRP-recommended values of wR are 1.0 for photons, electrons, positrons, and β-particles and 20 for α-particles (8). Some radionuclides used in nuclear medicine (e.g., 99mTc, 123I, 125I, and 201Tl) emit Auger electrons. The radiation-weighting factors of these low-energy electrons, based on their RBE, may be higher than 1.0 when the radionuclide is incorporated into the DNA of the cell nucleus. The ICRP does not give specific recommendations on the value of wR for Auger electron emitters but recommends that its value be determined on a case-by-case basis (7). Guidance is, however, given in the American Association of Physicists in Medicine (AAPM) report no. 49 (15). The AAPM recognized that the RBE (albeit for deterministic effects) caused by Auger electrons emitted by DNA-incorporated radionuclides is similar to that seen for high-LET α-particles and, thus, has recommended a radiation-weighting factor of 20 for this localization of Auger emitters. Furthermore, as there is a linear dependence of the RBE on the subcellular distribution of the Auger electron emitter (16), the AAPM has recommended a linear weighting of the Auger electron contribution to the equivalent dose that is dependent on that subcellular distribution (15). Although not relevant to nuclear medicine, wR is defined for neutrons as an energy-dependent function ranging from 2.5 to 20.7, and the wR for protons is assigned a single value of 2 (8). The unit for equivalent dose is the J kg−1, with the special name sievert (Sv).
Equations 1–4 can be written in terms of equivalent dose by replacing S with a radiation-weighted S denoted as Sw. The quantity  represents the time-dependent equivalent dose rate in target tissue rT per unit activity present in source tissue rS. Sw is given as:
represents the time-dependent equivalent dose rate in target tissue rT per unit activity present in source tissue rS. Sw is given as: Eq. 11where ER,i and YR,i are the energy and yield, respectively, of the ith radiation of type R, and ΔR,i is their product. As noted, the energies and yields in Equation 11 must be indexed separately by radiation type R. The equivalent dose rate in target tissue rT of the reference individual
Eq. 11where ER,i and YR,i are the energy and yield, respectively, of the ith radiation of type R, and ΔR,i is their product. As noted, the energies and yields in Equation 11 must be indexed separately by radiation type R. The equivalent dose rate in target tissue rT of the reference individual 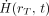 is given as:
is given as: Eq. 12
Eq. 12
The equivalent dose 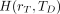 in target tissue rT after intake or administration of the radioactive material in the reference individual over the dose-integration period TD is given as:
in target tissue rT after intake or administration of the radioactive material in the reference individual over the dose-integration period TD is given as: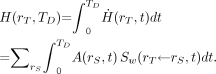 Eq. 13
Eq. 13
Under the condition that Sw may be considered to be time-independent, Equation 13 reduces to: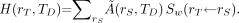 Eq. 14
Eq. 14
If 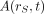 of Equation 13 is replaced by
of Equation 13 is replaced by  (activity in rS per unit activity inhaled, ingested, or intravenously injected), then the equation yields the equivalent dose coefficient
(activity in rS per unit activity inhaled, ingested, or intravenously injected), then the equation yields the equivalent dose coefficient 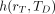 given as:
given as: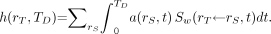 Eq. 15
Eq. 15
Under conditions in which tissue masses remain constant over the period TD, Equation 15 reduces further to: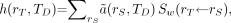 Eq. 16where
Eq. 16where 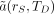 is defined as in Equation 9.
is defined as in Equation 9.
Use of Equivalent Dose in Medical Dose Assessments
As described above, the radiation-weighting factors wR for high-LET radiation (such as α-particles) are ICRP committee–derived values based on representative values of the RBE of that radiation type for causing stochastic effects (7). Accordingly, the equivalent dose, given as the product of the absorbed dose and wR values, is reserved for use in risk assessment associated only with radiation-induced stochastic effects. The wR values are not intended for use in predicting deterministic effects and, if used as such, may result in an overestimation of their occurrence and severity to irradiated tissues (8). Dosimetry quantities of relevance to deterministic effects are discussed later.
Effective Dose
The effective dose E is a radiation protection quantity defined by the ICRP in publications 60 (7) and 103 (8) for establishing annual limits of exposure to workers and members of the general public. The quantity takes into account external radiation fields and internal radionuclide sources that both contribute to low-dose irradiation of tissues and organs. The effective dose supersedes the effective dose equivalent originally defined in ICRP publications 26 and 30 (17,18). For a reference individual and dose-integration period TD (50 y for adults and to age 70 y for nonadults), the effective dose is defined as: Eq. 17where
Eq. 17where 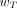 is a tissue-weighting factor for target tissue rT subject to the condition that
is a tissue-weighting factor for target tissue rT subject to the condition that 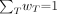 . The sum is performed over all organs and tissues of the human body considered to be sensitive to the induction of stochastic effects. Values of
. The sum is performed over all organs and tissues of the human body considered to be sensitive to the induction of stochastic effects. Values of  are chosen to represent the contribution of individual organs and tissues to overall radiation detriment from stochastic effects. The special named unit for the effective dose is the sievert (Sv). Care must be taken to identify whether the equivalent dose or effective dose is being reported, because the sievert is associated with both these radiation protection quantities.
are chosen to represent the contribution of individual organs and tissues to overall radiation detriment from stochastic effects. The special named unit for the effective dose is the sievert (Sv). Care must be taken to identify whether the equivalent dose or effective dose is being reported, because the sievert is associated with both these radiation protection quantities.
The organs and tissues for which 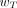 are specified by the ICRP are given in Table 2 (8). They represent mean values for humans averaged over both sexes and all ages and thus do not take account of the characteristics of any one individual. The
are specified by the ICRP are given in Table 2 (8). They represent mean values for humans averaged over both sexes and all ages and thus do not take account of the characteristics of any one individual. The 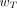 values for the remainder tissues (0.12) apply to the arithmetic mean of the equivalent doses of the 13 organs and tissues for each sex listed in the footnote to Table 2 (including the prostate for males and uterus/cervix for females).
values for the remainder tissues (0.12) apply to the arithmetic mean of the equivalent doses of the 13 organs and tissues for each sex listed in the footnote to Table 2 (including the prostate for males and uterus/cervix for females).
Recommended Tissue-Weighting Factors
On the basis of the results of epidemiologic studies of cancer expression in exposed populations and risk assessments for hereditary effects, a set of 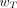 values was chosen by the ICRP according to assigned relative radiation detriment. In addition, the following judgments were applied. First, the detriments from heritable effects and cancer after gonadal irradiation (e.g., to ovaries and testes) were combined to give a
values was chosen by the ICRP according to assigned relative radiation detriment. In addition, the following judgments were applied. First, the detriments from heritable effects and cancer after gonadal irradiation (e.g., to ovaries and testes) were combined to give a 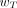 value of 0.08. Second, the thyroid-weighting factor was set to 0.04, representing the higher risk of thyroid cancer in childhood as young children are considered to be particularly sensitive to radiation-induced thyroid cancer. Third, cancer risks in salivary glands and brain, although not precisely quantified, were judged to be greater than those for the other tissues and organs comprising the remainder tissues; each is assigned a
value of 0.08. Second, the thyroid-weighting factor was set to 0.04, representing the higher risk of thyroid cancer in childhood as young children are considered to be particularly sensitive to radiation-induced thyroid cancer. Third, cancer risks in salivary glands and brain, although not precisely quantified, were judged to be greater than those for the other tissues and organs comprising the remainder tissues; each is assigned a 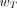 value of 0.01. Fourth, for the purposes of radiologic protection, the
value of 0.01. Fourth, for the purposes of radiologic protection, the 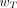 values are assumed to be valid for both sexes and all age groups.
values are assumed to be valid for both sexes and all age groups.
The effective dose for protection of reference persons is based on mean absorbed doses in organs or tissues of the human body and is defined and estimated in a reference individual. The quantity provides a value that takes account of some aspects of the given exposure situation but not the characteristics of a specific individual. In particular, the weighting factors are mean values representing an average over many individuals of both sexes. The reference individual can be either an adult or a child or infant, and in the ICRP system these include the newborn; a 1-, 5-, 10-, and 15-y-old; and the adult (19).
Use of the Effective Dose in Medical Dose Assessment
Effective dose is intended for applications in radiological protection (20). In the context of medical exposures, the effective dose is of value for comparing patient exposures originating from different diagnostic procedures, patient exposures using similar imaging procedures across different hospitals and different nations, and different imaging technologies for the same medical examination. In nuclear medicine, the effective dose is an important tool for conveying the sex- and age-averaged risk of stochastic effects to future populations of patients. As such, the quantity is widely reported in research and clinical protocols for use by Institutional Review Boards as an index of patient risk.
Nevertheless, the limitations of the effective dose for use in nuclear medicine should be clearly understood (21). As shown in Equation 17, the effective dose requires the use of 2 computational phantoms—1 male and 1 female—such that the equivalent organ doses are then averaged and weighted by  . Furthermore, these phantoms should conform to reference 50th percentile individuals as defined in ICRP publication 89 (22). Accordingly, the effective dose for medical exposures cannot be assigned as an index of stochastic risk to a single individual patient (male or female), nor can it be assigned to male or female patients of body morphometries significantly different from those of the ICRP reference individuals. These limitations stem from the fact that wT is both sex- and age-averaged. As a result, the sex-averaged value of
. Furthermore, these phantoms should conform to reference 50th percentile individuals as defined in ICRP publication 89 (22). Accordingly, the effective dose for medical exposures cannot be assigned as an index of stochastic risk to a single individual patient (male or female), nor can it be assigned to male or female patients of body morphometries significantly different from those of the ICRP reference individuals. These limitations stem from the fact that wT is both sex- and age-averaged. As a result, the sex-averaged value of 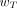 for the breasts given in Table 2 provides no information on the risk of breast cancer in male patients. Similarly, the age-averaged value of
for the breasts given in Table 2 provides no information on the risk of breast cancer in male patients. Similarly, the age-averaged value of  for the thyroid given in Table 2 overemphasizes the risk of thyroid cancer in adult patients and conversely underemphasizes that risk in children. Risks of cancer induction (stochastic effects) and risks of tissue reactions (deterministic effects) differ in both magnitude and import to individual patients receiving diagnostic or therapeutic radiologic procedures. The effective dose is an appropriate quantity for assessing stochastic risk as delivered in diagnostic exposures to populations of patients whose age and sex distribution do not significantly differ from those considered in the derivation of wT (8). The organ-absorbed dose is relevant when assessing the magnitude of deterministic effects in high-dose therapy procedures to individual patients.
for the thyroid given in Table 2 overemphasizes the risk of thyroid cancer in adult patients and conversely underemphasizes that risk in children. Risks of cancer induction (stochastic effects) and risks of tissue reactions (deterministic effects) differ in both magnitude and import to individual patients receiving diagnostic or therapeutic radiologic procedures. The effective dose is an appropriate quantity for assessing stochastic risk as delivered in diagnostic exposures to populations of patients whose age and sex distribution do not significantly differ from those considered in the derivation of wT (8). The organ-absorbed dose is relevant when assessing the magnitude of deterministic effects in high-dose therapy procedures to individual patients.
QUANTITIES RELEVANT TO THE RISK OF DETERMINISTIC EFFECTS
Absorbed dose is the relevant starting quantity for evaluation of the biologic effects of ionizing radiation emitted by administered radiopharmaceuticals. For diagnostic nuclear medicine procedures, radiation-absorbed doses to tissues of the patient are low, and the resulting stochastic risk of cancer or heritable disease is correspondingly low or absent. In therapeutic nuclear medicine, however, absorbed doses to nontargeted tissues can be high and can result in both an increased stochastic risk of cancer and the induction of deterministic effects such as hematologic toxicity, renal failure, gastrointestinal tract toxicity, or lung fibrosis.
RBE-Weighted Dose
When assessing the potential for deterministic effects, the ICRP recommends that the mean absorbed dose to the organ or tissue be weighted by an appropriate value of the RBE for the specified biologic endpoint (8). In this context, the use of RBE is analogous to that for the weighting factor wR in defining the equivalent dose, except that in this case the RBE is a quantity for deterministic endpoints measured under a specific set of experimental conditions rather than a single set of values chosen by committee review of RBE values for stochastic endpoints such as cancer induction. This distinction is important, and failure to appreciate the difference between radiation-weighting factors for stochastic effects and RBE values for deterministic effects in the context of therapy has led to confusion regarding which value is appropriate for weighting tissue-absorbed dose in radionuclide therapy. This difference in biologic endpoint is especially important for α-particle emitters for which the wR is 20 to reflect the relative risk of cancer induction, yet RBE values range from 1 to 8 for cell killing in vivo, depending on the reference radiation, α-particle energy, and biologic endpoint (23–27). RBE values for deterministic effects differ for different biologic endpoints in different organs and tissues. Guidance on appropriate values for the RBE for deterministic effects can be found in ICRP publications 58 (28) and 92 (29), International Commission on Radiation Units and Measurements (ICRU) report 67 (30), and National Council on Radiation Protection and Measurements (NCRP) report no. 104 (31).
As noted previously, the sievert is the special name of the unit assigned to the equivalent dose, which is the product of the tissue-absorbed dose and the radiation-weighting factor wR relevant to stochastic biologic effects. No corresponding special name has been formally adopted to describe the RBE-weighted dose as pertinent to deterministic effects. For example, the unit of the RBE-weighted dose is given as the gray by the ICRP (8) and as the gray-equivalent (Gy-Eq) by the NCRP (32). In addition to absorbed dose–modifying factors that pertain to the radiation type (e.g., α- versus β-particles) and radiation quality (e.g., LET), a variety of other factors also influence the risk of deterministic effects such as dose rate, radiosensitivity, and dose uniformity.
Biologically Effective Dose (BED)
Scientists have acknowledged since the early 1970s that dose rate influences biologic response (33). The BED formalism was developed to compare different fractionation protocols for external radiotherapy (34–38). BED may be thought of as the total physical dose required for a specified biologic effect when it is delivered at a very low dose rate or in many small-dose fractions. Radiobiologic parameters in the BED formulation include α and β, the sensitivity per unit dose and per unit dose squared, respectively, in the linear-quadratic dose-response model, and μ, the rate of repair of sublethal damage (36,37).
In radionuclide therapy, the dose rate is temporally variant, and several investigators have examined the implications of this on the balance between tumor control and normal tissue toxicity (39–42). To date, almost all clinical studies have considered only the total absorbed dose, the majority of which is delivered at an exponentially decreasing dose rate. However, the basis for projecting potential toxicity and justifying initial phase I–administered activity and absorbed dose levels has been the experience with normal organ tolerance in external-beam radiotherapy, the majority of which is delivered in high–dose-rate daily fractions of 2 Gy over a period of 30–40 days. By converting the radionuclide dose-rate profile and the fractionated external-beam dose-delivery profile to a BED, the formulation makes it possible to compare different dose-delivery schemes in terms of likely, tissue-specific, biologic effects.
Equivalent Uniform Dose (EUD)
Dose-volume histograms have been used to summarize the large amount of data present in 3-dimensional distributions of absorbed dose in radionuclide dosimetry studies (43,44). The EUD model takes this one step further by converting the spatially varying absorbed dose distribution into an equivalent uniform absorbed dose value that would yield a biologic response similar to that expected from the nonuniform dose distribution under consideration. The EUD (expressed in Gy) is a single quantity that may be used to compare different dose distributions; its value can also be used to estimate the probability that the magnitude and spatial distribution of the absorbed dose is sufficient for tumor sterilization (45).
Isoeffective Dose
The ICRU and the International Atomic Energy Agency have recently proposed the isoeffective dose for use in high-LET radiation therapy applications (46). The isoeffective dose is defined as the equivalent absorbed dose of low-LET radiation that when delivered under reference conditions would produce the same clinical effects as the high-LET treatment, all other conditions being identical. The quantity is given as the product of the absorbed dose D and a weighting factor wIsoE that includes the effects of multiple variables such as the absorbed dose, dose rate, dose per fraction, radiation quality, and other irradiation conditions known to affect the clinical outcome. Although proposed initially in the context of heavy-ion external-beam radiotherapy, the quantity can in principle be extended to applications in radionuclide therapy.
Dose-response models for deterministic effects are useful for patient treatment planning, and thus dosimetric quantities specific to these high-dose tissue reactions in radionuclide therapy are needed. The Committee is currently addressing this important need. In cases of radionuclides with particulate emissions that have distinct RBE values for the chosen biologic endpoint (e.g., α-emitters), the Committee currently recommends that the absorbed dose always be reported for each particle type, along with the relevant RBE value. In addition, the Committee recommends that radiobiologic model–derived quantities such as the BED, the EUD, or the isoeffective dose be reported with the parameter values used in their corresponding derivations (e.g., α-to-β ratio, dose rate).
CONCLUSION
This pamphlet presents a revised dosimetry schema consistent with the needs of both the nuclear medicine and radiation protection communities, with the goal of standardizing nomenclature between the MIRD and ICRP systems. This revision to the MIRD schema provides the basis for achieving consistent use of quantities, symbols, and units used by both organizations for the assessment of tissue-absorbed dose resulting from internalized radioactivity, whether from medical administrations or accidental or occupational exposures. The ICRP radiation protection quantities equivalent dose and effective dose are adopted by the MIRD committee for assessment of stochastic risk to broad groups of patients administered diagnostic or therapeutic amounts of radiopharmaceuticals. As the radiation- and tissue-weighting factors given by the ICRP are subject to change over time, the MIRD Committee recommends that the absorbed dose to tissues always be presented when reporting the equivalent and effective doses. In addition, as acknowledged by the ICRP in its 2007 recommendations, the effective dose is applied to prospective dose assessment for population-wide exposures (using absorbed doses assessed in reference phantoms) and should not be used to infer stochastic risk to any individual male or female subject. A need, therefore, exists to provide to the nuclear medicine community a radiation dosimetry quantity that can better relate stochastic risk differentiated by both age and sex.
Furthermore, the MIRD Committee recognizes the need to clarify differences in the radiation protection quantities applicable to stochastic effects (e.g., cancer induction) from those dosimetry quantities pertinent to deterministic effects (e.g., normal organ toxicity and tumor cell kill). Dose-response models for deterministic effects are useful for patient treatment planning, and thus dosimetric quantities specific to these high-dose tissue reactions in radionuclide therapy are needed. The Committee is currently engaged in addressing this important need and has provided specific recommendations that the nuclear medicine community can adopt during this interim period.
APPENDIX
Eckerman and Endo (14) have tabulated the energy spectra for β-particle emitters relevant to nuclear medicine using a fixed logarithmic-type energy grid. The tabulation provides for a series of energies E (MeV) ranging from zero to the end-point energy Eo of the spectrum and the number of β-particles per MeV per nuclear transformation emitted at that energy, P(E). P(E) is not averaged over an energy bin which would have involved specifying both a lower and an upper energy value. An example of a binned presentation of a spectrum can be seen in the Cf-252 neutron spectrum included in the data files of Eckerman and Endo (14). The number of β-particles emitted per nuclear transformation Nβ is given by: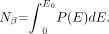 Eq. 1AThe total energy of the β-emissions per nuclear transformation ET is:
Eq. 1AThe total energy of the β-emissions per nuclear transformation ET is: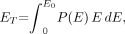 Eq. 2Aand the average energy of the β-spectrum is:
Eq. 2Aand the average energy of the β-spectrum is: Eq. 3AIn the event that the specific absorbed fraction for the source-target regions of interest is energy-dependent, then the contribution of the β-emissions to S is computed as:
Eq. 3AIn the event that the specific absorbed fraction for the source-target regions of interest is energy-dependent, then the contribution of the β-emissions to S is computed as: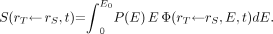 Eq. 4A
Eq. 4A
Footnotes
-
In collaboration with the SNM MIRD Committee: Wesley E. Bolch, A. Bertrand Brill, Darrell R. Fisher, Roger W. Howell, Ruby Meredith, George Sgouros, Stephen R. Thomas (Chair), and Barry W. Wessels.
-
COPYRIGHT © 2009 by the Society of Nuclear Medicine, Inc.
References
- Received for publication July 16, 2008.
- Accepted for publication September 9, 2008.







