Visual Abstract
Abstract
Preclinical data have shown that 161Tb-labeled peptides targeting the somatostatin receptor are therapeutically more effective for peptide receptor radionuclide therapy than are their 177Lu-labeled counterparts. To further substantiate this enhanced therapeutic effect, we performed cellular dosimetry to quantify the absorbed dose to the cell nucleus and compared dose–response curves to evaluate differences in relative biological effectiveness in vitro. Methods: CA20948 cell survival was assessed after treatment with [161Tb]Tb- and [177Lu]Lu-DOTATATE (agonist) and with [161Tb]Tb- and [177Lu]Lu-DOTA-LM3 (antagonist) via a clonogenic assay. Cell binding, internalization, and dissociation assays were performed up to 7 d to acquire time-integrated activity coefficients. Separate S values for each type of particle emission (Auger/internal conversion [IC] electrons and β− particles) were computed via Monte Carlo simulations, while considering spheric cells. Once the absorbed dose to the cell nucleus was calculated, survival curves were fitted to the appropriate linear or linear-quadratic model and corresponding relative biological effectiveness was evaluated. Results: Although the radiopeptide uptake was independent of the radionuclide, [161Tb]Tb-DOTATATE and [161Tb]Tb-DOTA-LM3 delivered a 3.6 and 3.8 times higher dose to the nucleus, respectively, than their 177Lu-labeled counterparts on saturated receptor binding. This increased nucleus-absorbed dose was mainly due to the additional emission of IC and not Auger electrons by 161Tb. When activity concentrations were considered, both [161Tb]Tb-DOTATATE and [161Tb]Tb-DOTA-LM3 showed a lower survival fraction than did labeling with 177Lu. When the absorbed dose to the nucleus was considered, no significant difference could be observed between the dose–response curves for [161Tb]Tb- and [177Lu]Lu-DOTATATE. [161Tb]Tb-DOTA-LM3 showed a linear-quadratic dose response, whereas [161Tb]Tb-DOTATATE showed only a linear dose response within the observed dose range, suggesting additional cell membrane damage by Auger electrons. Conclusion: The IC, rather than Auger, electrons emitted by 161Tb resulted in a higher absorbed dose to the cell nucleus and lower clonogenic survival for [161Tb]Tb-DOTATATE and [161Tb]Tb-DOTA-LM3 than for the 177Lu-labeled analogs. In contrast, [161Tb]Tb-DOTATATE showed no higher dose response than [177Lu]Lu-DOTATATE, whereas for [161Tb]Tb-DOTA-LM3 an additional quadratic response was observed. Because of this quadratic response, potentially caused by cell membrane damage, [161Tb]Tb-DOTA-LM3 is a more effective radiopeptide than [161Tb]Tb-DOTATATE for labeling with 161Tb.
Currently, [177Lu]Lu-DOTA-[Tyr3]octreotate ([177Lu]Lu-DOTATATE) is the standard clinical peptide receptor radionuclide therapy for patients with unresectable or metastatic, progressive, and somatostatin receptor type 2 (SSTR2)–positive gastroenteropancreatic neuroendocrine tumors, as approved by both the European Medicines Agency (2017) and the American Food and Drug Administration (2018). It combines 177Lu, a low-energy β−-emitter, with the SSTR2-agonist octreotate, which has shown therapeutic efficacy, as first demonstrated in the NETTER-1 clinical trial (1). Meanwhile, 161Tb has been identified as a highly promising radioisotope for β−-therapy, with a half-life and a β−-emission spectrum comparable to those of 177Lu. In addition, 161Tb emits per nuclear transition on average 1.4 internal conversion (IC) and 11 Auger electrons, compared with 0.15 IC and 1 Auger electron per 177Lu decay (Supplemental Fig. 2 shows the spectra in detail; supplemental materials are available at http://jnm.snmjournals.org). These additional low-energy electrons are anticipated to enhance therapeutic efficacy because of their shorter range and higher linear energy transfer.
The β−, IC, and Auger electron emissions of 161Tb have an average range in water of 301 µm (continuously-slowing-down approximation), 13 µm, and 97 nm, respectively (estimated with the Geant4 example “range” (2–4)), which makes them particularly effective for treating small metastases, as demonstrated by the metastatic model of Bernhardt et al. (5). The high linear energy transfer of Auger electrons is expected to induce more complex DNA damage, thereby resulting in a higher biological effectiveness when emitted near nuclear DNA (6). Not only the nuclear DNA but also the cell membrane was shown to be radiosensitive to Auger electrons (7). Accordingly, cell membrane–binding antagonists were found to benefit more from the Auger electrons emitted by 161Tb than do internalizing agonists (8).
Numerous studies, both in vitro and in vivo, consistently reported a higher therapeutic effectiveness for 161Tb-labeled radiopharmaceuticals than for their 177Lu-labeled counterparts for equivalent administered activities (8–10). In addition, Monte Carlo simulations have revealed that, for identical administered activities, 161Tb yielded a higher absorbed dose than 177Lu (11). However, these individual reports on the therapeutic response and dosimetry did not provide conclusive evidence of the increased biological effectiveness of 161Tb- versus 177Lu-labeled peptide. Therefore, the aim of this paper was to evaluate the biological effectiveness in vitro by establishing dose–response relationships for the radiolabeled SSTR2 agonists [177Lu]Lu- and [161Tb]Tb-DOTATATE and antagonists [177Lu]Lu- and [161Tb]Tb-DOTA-LM3, to determine which combination proved to be the most effective for peptide receptor radionuclide therapy and the better option for clinical translation.
MATERIALS AND METHODS
Cell Culture and Radiolabeling
CA20948 cells were kindly provided by Prof. Julie Nonnekens (Erasmus MC) (12) and cultured with Dulbecco modified Eagle medium at 37°C and 5% CO2. CA20948 cells (2 × 105) were seeded in 12-well plates the day before the treatments to allow for adherence. Radiolabeling was done at a 161Tb or 177Lu concentration of 400 MBq/mL in 0.1 M NaOAc (pH 4.7) for 15 min at 90°C to obtain a molar activity of 100 MBq/nmol. The radiochemical purity was checked with instant thin-layer chromatography after each radiolabeling (>95%). The radiolytic stability was verified by high-performance liquid chromatography (>90% after 6 h). The precursors DOTATATE (ABX) and DOTA-LM3 (MedChemExpress) were radiolabeled with no-carrier-added 177Lu (ITM) and no-carrier-added 161Tb (SCK CEN). All compounds were used without additional purification.
Colony Survival Assay
The reproductive cell survival after radioligand treatment was evaluated with a colony survival assay. CA20948 cells were incubated for 4 h with a range of activity concentrations (from 1.22 to 1,250 kBq/mL), after which the cells were trypsinized and seeded in a 6-well plate in triplicate (300 cells per well). After 14 d, the colonies (≥50 cells per colony) were fixed and stained with 6% glutaraldehyde and 0.5% crystal violet. The plates were imaged with the transmission light of Fusion FX (Vilber), and colonies were counted with the Analyse Particles function in ImageJ software after thresholding (13). Finally, the number of colonies was normalized to a control condition to obtain the percentage of survival.
Uptake Assays
A saturation binding experiment was performed for activity concentrations from 0.01 to 5 MBq/mL. The results were fitted to the one-site total and nonspecific binding model of Prism (GraphPad):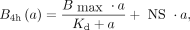 Eq. 1where
Eq. 1where 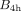 is the number of bound radiopharmaceuticals per cell after 4 h; a is the activity concentration; and
is the number of bound radiopharmaceuticals per cell after 4 h; a is the activity concentration; and  , and NS are fitting parameters representing the maximal specific binding, affinity, and nonspecific binding, respectively. The association and dissociation kinetics were assumed to be independent of the isotope and were determined with only 161Tb- and 177Lu-labeled peptides, respectively. In both uptake assays, the cell membrane and the internalized and medium fractions were collected. More details are described in the supplemental materials (14,15).
, and NS are fitting parameters representing the maximal specific binding, affinity, and nonspecific binding, respectively. The association and dissociation kinetics were assumed to be independent of the isotope and were determined with only 161Tb- and 177Lu-labeled peptides, respectively. In both uptake assays, the cell membrane and the internalized and medium fractions were collected. More details are described in the supplemental materials (14,15).
Cellular Dosimetry
The average absorbed dose to the cell nuclei during the colony survival assay was determined for each added activity concentration a on the basis of the formalism of the MIRD committee (16):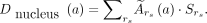 Eq. 2
Eq. 2
The considered source regions 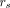 were the cytoplasm, the cell membrane, the neighboring cells, and the activity in the culture medium. The time-integrated activity coefficients
were the cytoplasm, the cell membrane, the neighboring cells, and the activity in the culture medium. The time-integrated activity coefficients  are the total number of nuclear decays within a source region and were based on the data from the saturation binding and kinetic uptake assay. These were multiplied by the corresponding S values, which are the absorbed dose to the cell nucleus per decay within a source region. The S values were simulated with the Monte Carlo particle transportation code Geant4 for a spheric geometry. More detailed information can be found in the supplemental materials (3,17–20).
are the total number of nuclear decays within a source region and were based on the data from the saturation binding and kinetic uptake assay. These were multiplied by the corresponding S values, which are the absorbed dose to the cell nucleus per decay within a source region. The S values were simulated with the Monte Carlo particle transportation code Geant4 for a spheric geometry. More detailed information can be found in the supplemental materials (3,17–20).
Radiobiologic Parameters
The dose–response relationships were obtained by plotting the cell survival fractions against the absorbed doses to the cell nucleus. The linear-quadratic (LQ) model describes the dose response as follows: Eq. 3
Eq. 3
where α represents the cell radiosensitivity to the radiation and β accounts for superposition of radiation damage. In targeted radionuclide therapy, the quadratic contribution is generally so low that the dose–response relationship can often be described by a linear (L) model: Eq. 4
Eq. 4
Both models were fitted with nonlinear regression to the dose–response data, and selection of the best fit was based on the extra-sum-of-squares F test. The fitted α and β values were used to calculate the relative biological effectiveness (RBE),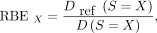 Eq. 5which is the absorbed dose of a reference radiation over the dose of the investigated radiation type that induces an equal biological effect, which was here a survival fraction X, with X = 10% or X = 50%. When possible, the RBEmax, which is the RBE extrapolated to 100% survival, was also calculated as
Eq. 5which is the absorbed dose of a reference radiation over the dose of the investigated radiation type that induces an equal biological effect, which was here a survival fraction X, with X = 10% or X = 50%. When possible, the RBEmax, which is the RBE extrapolated to 100% survival, was also calculated as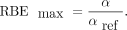 Eq. 6
Eq. 6
An external 137Cs source (γ of 662 keV, 0.6 Gy/min) was used to calculate  with the α and β for the CA20948 cells taken from Chan et al. (21).
with the α and β for the CA20948 cells taken from Chan et al. (21).
Statistical Methods
An unpaired parametric t test and 2-way ANOVA were used to test for statistical significance, with the significance level (α) set to 0.05 (Prism). Curves were fitted with least squares regression, and selection of the best fit was based on the extra-sum-of-squares F test, which was also used to compare and check whether 2 data sets could be fit with a single curve. The curve fitting returned the standard error for the estimated fitting parameters, which was used to calculate the error on the final absorbed dose via standard error propagation rules. Data were expressed as mean ± SD from N = 3 technical and n = 3 biological replicates, unless stated differently.
RESULTS
Clonogenic Survival
Clonogenic survival was significantly reduced after treatment with at least a 0.02 MBq/mL dose of [161Tb]Tb-DOTATATE or [161Tb]Tb-DOTA-LM3 in comparison with untreated controls (P < 0.0003, P < 0.0001), whereas for [177Lu]Lu-DOTATATE this reduction was significant from 0.08 MBq/mL (P < 0.02) onward and for [177Lu]Lu-DOTA-LM3 for a concentration of 0.16 and 1.25 MBq/mL (P = 0.012, P = 0.03, 2-way ANOVA, Fig. 1). Treatment with [161Tb]Tb-DOTATATE and -DOTA-LM3 significantly reduced clonogenic survival compared with the 177Lu-labeled analogs for activity concentrations higher than 0.04 and 0.02 MBq/mL, respectively (P < 0.014, P < 0.027). No significant difference was observed between [161Tb]Tb-DOTATATE and [161Tb]Tb-DOTA-LM3, whereas for [177Lu]Lu-DOTATATE a significantly lower clonogenic survival was observed at 0.625 and 1.25 MBq/mL than for [177Lu]Lu-DOTA-LM3 (P = 0.02, P = 0.01).
Clonogenic survival of CA20948 cells after treatment with [161Tb]Tb- and [177Lu]Lu-DOTATATE and -DOTA-LM3 as function of added activity concentration (MBq/mL). Error bars are defined as ±1 SD (N = 3, n = 6).
Saturation Binding and Uptake Kinetics
The saturation binding results are visualized in Figure 2, whereas fitting parameters are summarized in Supplemental Table 1. No significant differences in 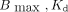 , or NS were observed between 161Tb- and 177Lu-labeled analogs; thus, data were pooled for further analysis. Primarily specific, saturable binding was observed, setting an upper limit to the self-dose achievable with a fixed molar activity. A maximum concentration of 12.5 nM was selected for the colony-forming assay so that the binding would not surpass 90% of the
, or NS were observed between 161Tb- and 177Lu-labeled analogs; thus, data were pooled for further analysis. Primarily specific, saturable binding was observed, setting an upper limit to the self-dose achievable with a fixed molar activity. A maximum concentration of 12.5 nM was selected for the colony-forming assay so that the binding would not surpass 90% of the 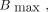 to avoid an increased dose contribution from the radioactive medium. [177Lu]Lu- and [161Tb]Tb-DOTA-LM3 showed a slightly lower
to avoid an increased dose contribution from the radioactive medium. [177Lu]Lu- and [161Tb]Tb-DOTA-LM3 showed a slightly lower  than did [177Lu]Lu- and [161Tb]Tb-DOTATATE (P = 0.04,
than did [177Lu]Lu- and [161Tb]Tb-DOTATATE (P = 0.04, 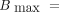 0.78 ± 0.14 vs. 1.1 ± 0.3 × 10−3 fmol/cell), whereas their affinity was significantly higher (P = 0.0012,
0.78 ± 0.14 vs. 1.1 ± 0.3 × 10−3 fmol/cell), whereas their affinity was significantly higher (P = 0.0012,  vs. 1.0 ± 0.4 nM). Nevertheless, the major difference was the higher internalized fraction of [177Lu]Lu- and [161Tb]Tb-DOTATATE (86.4%) than of [177Lu]Lu- and [161Tb]Tb- DOTA-LM3 (10.5%).
vs. 1.0 ± 0.4 nM). Nevertheless, the major difference was the higher internalized fraction of [177Lu]Lu- and [161Tb]Tb-DOTATATE (86.4%) than of [177Lu]Lu- and [161Tb]Tb- DOTA-LM3 (10.5%).
Saturation binding assay representing total binding and nonspecific binding for different added activity concentrations of [161Tb]Tb-DOTATATE with fixed molar activity of 100 MBq/nmol. Error bars are defined as ±1 SD (N = 3, n = 3).
Figure 3 shows the association and dissociation kinetics, along with the fitted curves of the equations (Supplemental Eqs. 5–8). After the incubation phase, [177Lu]Lu- and [161Tb]Tb-DOTATATE showed an exponential excretion of the activity over time, whereas [177Lu]Lu- and [161Tb]Tb-DOTA-LM3 showed an initial internalization before excretion into the medium. The association kinetics of [177Lu]Lu- and [161Tb]Tb-DOTATATE were not significantly different from those of [177Lu]Lu- and [161Tb]Tb-DOTA-LM3 (Supplemental Table 1). The final time–activity curves were obtained after decay correction and a correction for cell division with a doubling time of  h (Supplemental Fig. 1).
h (Supplemental Fig. 1).
(A) Association kinetics during 4 h of incubation with 0.024, 0.78 and 12.5 nM [161Tb]Tb-DOTATATE. (B and C) Dissociation kinetics of 1 nM [177Lu]Lu-DOTATATE (B) and [177Lu]Lu-DOTA-LM3 (C) for all compartments (internalized, cell membrane, and medium fraction). Error bars are defined as ±1 SD (N = 3, n = 2).
Absorbed Dose to the Nucleus
The S values for the dose to the nucleus are summarized in Supplemental Table 2 for each source region, radioisotope, and particle separately. For 177Lu, the highest nuclear S values correspond to the emitted β− particles, whereas for 161Tb, IC electrons have the largest contribution. The difference between the nuclear S values from the cytoplasm 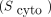 and the cell membrane
and the cell membrane  was relatively low for β− particles and IC electrons (
was relatively low for β− particles and IC electrons (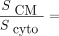 50%–70%), whereas
50%–70%), whereas 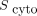 was 24 and 19 times higher than
was 24 and 19 times higher than  for the Auger electrons emitted by 177Lu and 161Tb, respectively. Plotting the total absorbed dose against the activity concentration (Fig. 4) revealed a nonlinear and saturable relationship. At higher activity concentrations, radiolabeled DOTATATE resulted in a higher absorbed dose than did radiolabeled DOTA-LM3, because of DOTATATE’s higher
for the Auger electrons emitted by 177Lu and 161Tb, respectively. Plotting the total absorbed dose against the activity concentration (Fig. 4) revealed a nonlinear and saturable relationship. At higher activity concentrations, radiolabeled DOTATATE resulted in a higher absorbed dose than did radiolabeled DOTA-LM3, because of DOTATATE’s higher 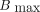 and higher internalization (Fig. 5B). Note that the contribution of the cytoplasm to the absorbed dose in the cell nucleus was still 47% (5.9 Gy/12.5 Gy) and 43% (1.4 Gy/3.4 Gy) for [161Tb]Tb-DOTA-LM3 and [177Lu]Lu-DOTA-LM3, respectively, because of the internalization over time. At lower activity concentrations, the absorbed dose of radiolabeled DOTA-LM3 was higher than that of radiolabeled DOTATATE because of its higher affinity. However, the most significant difference was observed between [161Tb]Tb-DOTATATE and [177Lu]Lu-DOTATATE and between [161Tb]Tb-DOTA-LM3 and [177Lu]Lu-DOTA-LM3, with an average increase of 3.6 and 3.8, respectively. The increased nucleus-absorbed dose of the 161Tb-labeled peptides was primarily due to the additional emission of IC electrons by 161Tb (Fig. 5A) and not by the Auger electrons.
and higher internalization (Fig. 5B). Note that the contribution of the cytoplasm to the absorbed dose in the cell nucleus was still 47% (5.9 Gy/12.5 Gy) and 43% (1.4 Gy/3.4 Gy) for [161Tb]Tb-DOTA-LM3 and [177Lu]Lu-DOTA-LM3, respectively, because of the internalization over time. At lower activity concentrations, the absorbed dose of radiolabeled DOTA-LM3 was higher than that of radiolabeled DOTATATE because of its higher affinity. However, the most significant difference was observed between [161Tb]Tb-DOTATATE and [177Lu]Lu-DOTATATE and between [161Tb]Tb-DOTA-LM3 and [177Lu]Lu-DOTA-LM3, with an average increase of 3.6 and 3.8, respectively. The increased nucleus-absorbed dose of the 161Tb-labeled peptides was primarily due to the additional emission of IC electrons by 161Tb (Fig. 5A) and not by the Auger electrons.
Total absorbed dose to cell nucleus for range of added activity concentrations of both 161Tb- and 177Lu-labeled DOTATATE and DOTA-LM3. Error bands represent ±1 SD.
Absorbed dose to cell nucleus, corresponding to 1 MBq/mL of respective radioligands depicted in x-axis, and contributions from different particles (A) and source regions (B). Error bars are defined as ±1 SD of total absorbed dose calculated over all particle types or all source regions.
Dose Response
The nuclear dose–response data of both [177Lu]Lu- and [161Tb]Tb-DOTATATE were best fitted with a linear model and could be fitted with a common, single curve (P = 0.24; Fig. 6A). This indicated that there was no significant difference between the dose–response relationships of [177Lu]Lu- and [161Tb]Tb-DOTATATE, which was reflected in comparable RBE values (Table 1). On the other hand, within the limited dose range, [161Tb]Tb-DOTA-LM3 fitted better to a linear-quadratic model, whereas [177Lu]Lu-DOTA-LM3 was best described by the linear model. Because the best-fitting model was different for both data sets, a direct comparison was not feasible (Fig. 6B). In addition, because of the limited dose range, and corresponding high survival fractions, the RBE values with survival fractions of 10% or 50% for [177Lu]Lu-DOTA-LM3 were not reported.
Clonogenic survival curves plotted against absorbed dose in cell nucleus (Gy), shown per peptide (A and B) or per radioisotope (C and D). All data are fitted to linear model except for [161Tb]Tb-DOTA-LM3, which is fitted to linear-quadratic model. Fitting parameters are summarized in Table 1. Error bars correspond to ±1 SD, whereas error bands represent 95% confidence interval of curve fit.
Fitting Parameters of Survival Curves
The dose–response data of [177Lu]Lu-DOTATATE and -DOTA-LM3 were better fitted with 2 separate linear curves, indicating a significantly higher dose response for [177Lu]Lu-DOTATATE (P < 0.0001; Fig. 6C). In addition, [161Tb]Tb-DOTATATE and -DOTA-LM3 were also better fitted with 2 separate dose–response curves (P < 0.0001), confirming a higher dose response for [161Tb]Tb-DOTA-LM3 than for [161Tb]Tb-DOTATATE in the higher absorbed-dose range (Fig. 6D). However, for an absorbed dose below 6.9 ± 0.3 Gy, the dose response of [161Tb]Tb-DOTA-LM3 was lower than that of [161Tb]Tb-DOTATATE (Fig. 6D). These observations are again reflected in the calculated RBE values. Note that all RBE values were estimated as lower than 1, indicating a lower dose response than the reference 137Cs irradiation.
DISCUSSION
In previous work, it was shown that treatment with 161Tb-labeled peptides reduced cell survival compared with 177Lu-labeled analogs for the same activity concentration (8–10). Simulations have also shown that the 161Tb emission spectrum resulted in a higher cellular absorbed dose than did the 177Lu emission spectrum (11). Both findings were confirmed by this study for 177Lu- and 161Tb-labeled DOTATATE and DOTA-LM3. In addition, the aim of this study was to extend these findings and compare their dose–response curves and subcellular dose distributions.
We did not observe a significant difference in dose response between [177Lu]Lu- and [161Tb]Tb-DOTATATE, as can be explained by the intracellular dose distributions of the different types of particle emissions. The increased absorbed dose to the nucleus for 161Tb-labeled agonists and antagonists originated predominantly from the higher fraction of IC electron emissions by 161Tb, rather than from the Auger electron emissions. Although IC and Auger electrons are often grouped together, their differences in energy result in a different range and linear energy transfer and, consequently, in a different dose distribution and dose response. More specifically, the range of the Auger electrons was too short (nm range), compared with the CA20948 cell geometry (µm range), to substantially contribute to the absorbed dose to the nucleus from within the cytoplasm or from the cell membrane. Hence, close subcellular targeting (nm range) of radiosensitive structures remains indispensable to exploit the full potential of Auger-emitting radioisotopes, such as 161Tb. Meanwhile, the additional emission of IC electrons by 161Tb resulted in a higher absorbed dose to the cell nucleus but not in a higher dose response, indicating that IC electrons have a similar biological effectiveness as β− particles. Nonetheless, the higher absorbed dose per nuclear transition for 161Tb than for 177Lu because of the emission of IC electrons is important when receptors are saturated, which allows delivery of a higher cellular absorbed dose and thus a more effective treatment.
[161Tb]Tb-DOTA-LM3 showed a linear-quadratic dose–response relationship for the nucleus, whereas the nuclear dose response of [161Tb]Tb-DOTATATE had only a linear component. Since [161Tb]Tb-DOTA-LM3 was initially bound to the cell membrane, the emission of Auger electrons by 161Tb likely caused additional cell membrane damage, resulting in a quadratic dependence of survival on the absorbed dose in the nucleus. Cell membrane damage by Auger electron emissions was also reported by Pouget et al. (7) and has been shown to induce bystander effects in nontargeted cells, which might contribute to the quadratic response (22,23). However, this remains a hypothesis and should be clarified by additional experiments to measure cell membrane damage in combination with cell membrane dosimetry, considering the small dimensions of the cell membrane (7.5–10 nm), the variable location of the radionuclide relative to the membrane, and the short range and cascade behavior of Auger electrons.
When comparing 177Lu-labeled DOTA-LM3 and DOTATATE, we observed a higher nuclear dose response for [177Lu]Lu-DOTATATE, indicating that β− particles caused more lethal cell damage when emissions were near the nucleus, despite their larger range. However, within the colony-forming assay, the trypsinization of the cells might have cleaved a portion of the SSTR2 after the peptide binding (24), resulting in lower binding within the clonogenic assay and thus an overestimation of the actual absorbed dose in the nucleus. This overestimation would be more prominent for the membrane-bound [177Lu]Lu-DOTA-LM3, meaning that the actual dose–response relationships for [177Lu]Lu-DOTA-LM3 and [177Lu]Lu-DOTATATE are probably more similar, as could be expected. This might also explain the observed higher dose response for [161Tb]Tb-DOTATATE than for [161Tb]Tb-DOTA-LM3 in the lower dose range. The trypsinization of the cells might also have led to selection of the least damaged cells, thereby underestimating survival for all radiopeptides (25). Treating the cells after seeding may circumvent this issue.
When we compared our findings with literature values, we found that Chan et al. obtained a lower radiosensitivity (α= 0.08) for [177Lu]Lu-DOTATATE than we did (α = 0.100 ± 0.012), and they also observed a quadratic component (β = 0.003) (21). O’Neill et al. reported a much higher value (α = 0.63 ± 0.07, β < 10−6) (26). Both studies considered only physical decay, whereas radioligand dissociation from the cell and cell division were neglected, potentially overestimating the absorbed dose and underestimating the radiosensitivity. Moreover, O’Neill et al. extrapolated uptake data to lower activity concentrations. Therefore, relating our findings to previously reported findings is not straightforward.
The main limitation of this study was that the resulting absorbed dose ranges of 177Lu-labeled DOTATATE and DOTA-LM3 only partially overlapped with those of the 161Tb-labeled analogs. For future comparison studies, this limitation could be overcome by labeling with a higher molar activity or using a cell type with an even higher SSTR2 expression level. Other limitations of this study were that the use of spheres as an approximation for the adherent cell geometry has been shown to overestimate the nucleus-absorbed dose (27) and the assumption of a homogeneous activity distribution within the cytoplasm has been shown to underestimate the absorbed dose in the nucleus, since fluorescent SSTR2 antibodies were accumulating within the trans-Golgi network after SSTR2 agonist binding (28). Further research will implement more realistic cell geometries. However, even then, the range of Auger electrons will be too limited to significantly contribute to the absorbed dose in the cell nucleus. Finally, a log-normal instead of uniform uptake of activity would increase the survival fractions at higher doses as shown by Neti et al., an effect that may be observed in Figure 6 as a trend toward a plateau phase for [161Tb]Tb-DOTATATE and [177Lu]Lu-DOTA-LM3 (29).
CONCLUSION
Cellular dosimetry and cell survival assays showed that [161Tb]Tb-DOTATATE and [161Tb]Tb-DOTA-LM3 resulted in a higher absorbed dose to the cell nucleus and a lower clonogenic survival than did the 177Lu-labeled analogs for the same activity. In addition, Monte Carlo simulations demonstrated that IC electrons contributed predominantly to the increased absorbed dose to the cell nucleus of 161Tb-labeled DOTATATE and DOTA-LM3, and the range of Auger electrons was too limited to contribute to the cell nucleus–absorbed dose. Evaluation of the dose–response curves revealed that 161Tb instead of 177Lu labeling did not significantly alter the dose response for DOTATATE. On the other hand, the antagonist [161Tb]Tb-DOTA-LM3 increased the biological effectiveness compared with the agonist [161Tb]Tb-DOTATATE for higher nucleus-absorbed doses, as indicated by the linear-quadratic dose–response relationship. This higher dose response may be due to the high membrane-bound fraction of [161Tb]Tb-DOTA-LM3 and the emission of Auger electrons by 161Tb, causing additional damage to the cell membrane. As a result, the SSTR2 antagonist [161Tb]Tb-DOTA-LM3 proved to be a more therapeutically effective peptide for labeling with 161Tb than did the agonist [161Tb]Tb-DOTATATE, making [161Tb]Tb-DOTA-LM3 the preferred combination for clinical translation.
DISCLOSURE
No potential conflict of interest relevant to this article was reported.
KEY POINTS
QUESTION: Does treatment with [161Tb]Tb-DOTATATE or -DOTA-LM3 result in a dose response different from treatment with the 177Lu-labeled peptide analogs?
PERTINENT FINDINGS: The additional absorbed dose to the nucleus from 161Tb-labeled peptides originated from IC and not Auger electrons, which did not result in a different dose–response relationship for [161Tb]Tb- and [177Lu]Lu-DOTATATE but did result in a quadratic response for [161Tb]Tb-DOTA-LM3.
IMPLICATIONS FOR PATIENTS CARE: To exploit the advantage of the Auger electrons of 161Tb, novel radiopharmaceuticals should target radiosensitive compartments of the cell.
ACKNOWLEDGMENTS
We thank Natalie Heynickx, Michelle Andersson, Charlotte Segers, and Anthony Waked for helping with cell culture and uptake experiments and Tomas Opsomer for optimizing the radiolabeling.
Footnotes
Published online Aug. 29, 2024.
- © 2024 by the Society of Nuclear Medicine and Molecular Imaging.
REFERENCES
- Received for publication April 8, 2024.
- Accepted for publication July 17, 2024.