Abstract
Immuno-PET is an appealing concept in the detection of tumors and planning of antibody-based therapy. For this purpose, the long-lived positron emitter 89Zr (half-life, 78.4 h) recently became available. The aim of the present first-in-humans 89Zr immuno-PET study was to assess safety, biodistribution, radiation dose, and quantification of the 89Zr-labeled chimeric monoclonal antibody (cmAb) U36 in patients with head and neck squamous cell carcinoma (HNSCC). In addition, the performance of immuno-PET for detecting lymph node metastases was evaluated, as described previously. Methods: Twenty HNSCC patients, scheduled to undergo surgical tumor resection, received 75 MBq of 89Zr-cmAb U36 (10 mg). Immuno-PET scans were acquired at 1, 24, 72, or 144 h after injection. The biodistribution of the radioimmunoconjugate was evaluated by ex vivo radioactivity measurement in blood and in biopsies from the surgical specimen obtained at 168 h after injection. Uptake levels and residence times in blood, tumors, and organs of interest were derived from quantitative immuno-PET studies, and absorbed doses were calculated using OLINDA/EXM 1.0. The red marrow dose was calculated using the residence time for blood. Results: 89Zr-cmAb U36 was well tolerated by all subjects. PET quantification of blood-pool activity in the left ventricle of the heart showed a good agreement with sampled blood activity (difference equals 0.2% ± 16.9% [mean ± SD]) except for heavy-weight patients (>100 kg). A good agreement was also found for the assessment of mAb uptake in primary tumors (mean deviation, −8.4% ± 34.5%). The mean absorbed red marrow dose was 0.07 ± 0.02 mSv/MBq and 0.09 ± 0.01 mSv/MBq in men and women, respectively. The normal organ with the highest absorbed dose was the liver (mean dose, 1.25 ± 0.27 mSv/MBq in men and 1.35 ± 0.21 mSv/MBq in women), thereafter followed by kidneys, thyroid, lungs, and spleen. The mean effective dose was 0.53 ± 0.03 mSv/MBq in men and 0.66 ± 0.03 mSv/MBq in women. Measured excretion via the urinary tract was less than 3% during the first 72 h. Conclusion: 89Zr immuno-PET can be safely used to quantitatively assess biodistribution, uptake, organ residence times, and radiation dose, justifying its further clinical exploitation in the detection of tumors and planning of mAb-based therapy.
Monoclonal antibodies (mAbs) have been approved for use as diagnostics and therapeutics in a broad range of medical indications, but especially in oncology (1). Immuno-PET, the tracking and quantification of mAbs with PET in vivo, is an exciting novel option to improve diagnostic imaging and to guide mAb-based therapy and has been described previously (2–6).
To enable PET of mAbs, an appropriate positron emitter—with a half-life (t1/2) that is compatible with the time needed to achieve optimal tumor-to-nontumor ratios (typically 2–4 d for intact mAbs)—has to be securely coupled to the targeting molecule. 124I (t1/2, 100.3 h) and 89Zr (t1/2, 78.4 h) are particularly suitable in combination with intact mAbs, because their long half-lives allow imaging at late time points for obtaining maximum information. Although the nonresidualizing positron emitter 124I is particularly suitable for immuno-PET when used in combination with noninternalizing intact mAbs, the residualizing positron emitter 89Zr may be optimal in combination with internalizing intact mAbs, because 89Zr stays in the targeted cell (residualization) after intracellular catabolism of the radioimmunoconjugate (7). 89Zr can also be used as a PET surrogate label for the prediction of the biodistribution and dosimetry of 177Lu-mAb and 90Y-mAb conjugates as used in radioimmunotherapy trials, although deviations have to be anticipated because of subtle differences in the metal–chelate complexes used (8,9).
Although the first clinical immuno-PET studies with 124I-labeled mAbs were performed about 15 y ago, technology for 89Zr immuno-PET became available just recently (10). For this purpose, we developed the large-scale production of pure 89Zr and a strategy for labeling mAbs with 89Zr via a multistep synthesis, using a succinylated derivative of desferrioxamine B (Df) as bifunctional chelate (10). Labeling technology is universal and, therefore, can be used for each individual mAb or other type of protein. In the meantime, several preclinical immuno-PET studies have been performed with 89Zr-labeled mAbs as a prelude to clinical trials, for example, with chimeric (mouse–human) mAb (cmAb) U36 (anti-CD44v6) (10), DN30 (anti-cMet) (11), G250 (anticarbonic anhydrase IX) (12), ibritumomab tiuxetan and rituximab (anti-CD20) (9), bevacizumab (anti–vascular endothelial growth factor) (13), cetuximab (antiepidermal growth factor receptor) (8,14), and trastuzumab (antihuman epidermal growth factor receptor-2) (15).
In a first-in-humans 89Zr immuno-PET clinical trial, we recently determined the diagnostic value of immuno-PET with anti-CD44v6 89Zr-cmAb U36 in patients with head and neck squamous cell carcinoma (HNSCC), who were at high risk of having neck lymph node metastases (2). Twenty HNSCC patients underwent 89Zr-cmAb U36 immuno-PET before surgery. Immuno-PET detected all primary tumors (n = 17) and lymph node metastases in 18 of 25 positive neck levels. For the detection of HNSCC lymph node metastases (and probably distant metastases), immuno-PET with 89Zr-cmAb U36 performs at least as well as CT or MRI.
No radiation dose estimates have been previously described for 89Zr-cmAb U36 or other 89Zr-labeled mAbs. Therefore, the aim of the present study was to assess the safety, biodistribution, radiation dose, and potential for quantification of immuno-PET with 89Zr-cmAb U36 in HNSCC patients using data from the aforementioned clinical trial.
MATERIALS AND METHODS
Patient Study
A total of 20 patients (8 women and 12 men) with histologically proven HNSCC (Table 1), at high risk of having neck lymph node metastasis and, therefore, planned to undergo neck dissection with or without resection of the primary tumor, participated in this study. Other eligibility criteria have been described previously (2). Patients received cmAb U36 IgG radiolabeled with 89Zr (74.9 ± 0.6 MBq). The total administered cmAb U36 dose was 10 mg for all patients. In previous studies, it had been demonstrated that biodistribution is not mAb dose–dependent within the range of 2–52 mg (16,17). Surgery was performed 6–8 d after the administration of radiolabeled cmAb U36.
Patient and Tumor Characteristics
Safety
Before and up to 6 wk after the administration of radiolabeled cmAb U36, routine laboratory analyses were performed, including hemoglobin, hematocrit, mean corpuscular volume, red blood cell count, white blood cell count (including automated differential), platelet count, sodium, potassium, calcium, chloride, creatinine, urea, uric acid, alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase, γ-glutamyl transferase, albumin, glucose, bilirubin, thyroid-stimulating hormone, and urine sediment. Vital signs including pulse rate, blood pressure, temperature, and respiratory rate were recorded before and up to 3 h after injection. On the basis of previous studies with anti-CD44v6 mAbs, cmAb U36 included, no adverse effects were expected (17). Dose rates (μSv/h) were measured at 1, 24, and 72 h after injection at a distance of 100 cm with a γ-radiation dose rate counter (Berthold LB 1230; EG&G). In addition, human anti–cmAb U36 and anti–Df-cmAb U36 antibody responses were assessed (2).
mAb U36
The selection and production of mouse mAb U36 and its chimeric IgG1 derivative cmAb U36 have been described previously (17). mAb U36 binds to the v6 region of CD44 (CD44v6). Expression of CD44v6 was found to be abundant and homogeneous in 96% of all primary HNSCC and HNSCC lymph node metastases (18). In normal tissues, expression has been found in epithelial tissues such as skin, breast and prostate myoepithelium, and bronchial epithelium (19). CD44v6 has been suggested to be involved in tumor formation, tumor cell invasion, metastasis formation, and cancer cell stemness (20,21).
Synthesis of 89Zr-cmAb U36
The synthesis and purification of 89Zr, its coupling to cmAb U36 via the chelate Df (Desferal; Novartis Pharma AG), and the procedures and results of quality tests have been described previously (2). 89Zr emits positrons with a main energy of 897 keV and an abundance of 22.7%. In addition, nonprompt 909-keV photons are emitted at an abundance of 99.9%.
Pharmacokinetics
Serial blood samples were taken from a peripheral vein of the arm opposite the infusion site for the determination of activity at the following time points: 5, 10, and 30 min and 1, 2, 4, 16, 21, 72, and 168 h after the completion of infusion. Urine was collected at intervals of 0–24, 24–48, and 48–72 h after injection to determine renal excretion of 89Zr. Aliquots of blood, plasma, and urine samples were measured for 89Zr activity in an isotope well-counter (1470 Wizard; Wallac), compared with an aliquot retained from the conjugate preparation, and corrected for decay. Blood activity was expressed as the percentage of the injected dose per kilogram.
PET Acquisition
PET scans were obtained at 1 h (all patients except 4 and 16), 24 h (patients 1–6), 72 h (all patients), or 144 h (all patients except 1, 2, and 13) after the intravenous injection of 89Zr-cmAb U36, using a dedicated full-ring PET scanner (ECAT EXACT HR+; CTI/Siemens) as described before (2). Before this study, the PET scanner was calibrated using a standard cylindric calibration phantom filled with a 89Zr solution (5 kBq/cm3). This calibration was performed to verify the quantitative accuracy of the scanner in the presence of 909-keV γ-photons emitted by 89Zr and the impact of emission spillover into the transmission scans. This procedure indicated that activity concentrations measured with the HR+ were accurate within 5%. During image reconstruction, all scans were normalized and corrected for randoms, scatter, attenuation, and decay. Reconstructions were performed using an attenuation- and normalization-weighted ordered-subset expectation maximization (OSEM) algorithm (ECAT software, version 7.2; CTI/Siemens) with 2 iterations and 16 subsets, followed by postsmoothing of the reconstructed image using a gaussian filter of 5 mm in full width at half maximum. Because of the low amount of radioactivity administered to the patients (for radiation exposure reasons), images with attenuation correction showed high noise levels. Therefore, OSEM reconstructions without attenuation (and thus scatter) correction were performed as well and used for visual interpretation only.
Validation of Quantitative 89Zr Immuno-PET
OSEM reconstructions with attenuation correction of the immuno-PET scans were used to investigate the accuracy of quantification 89Zr-cmAb U36 PET in vivo. To this end, three 1.5-cm-diameter regions of interest (ROIs) were defined within the left ventricle of the heart in 3 subsequent image planes using an axial view of the PET images. The average activity concentration within these ROIs, representing PET-assessed blood-pool activity concentration, was directly compared with the activity concentration measured in manual blood samples (sampled blood activity) using a calibrated well-counter to validate the quantitative accuracy of 89Zr PET in vivo.
In addition, the uptake of the radioimmunoconjugate in tumors (percentage injected dose [%ID]/cm3), as assessed from scans acquired at 144 h after injection, was compared with the uptake (%ID/g) derived from tumor biopsies collected at 168 h after injection. %ID/cm3, as derived from the PET scans, was converted to %ID/g using a soft-tissue density factor of 1.04 g/cm3.
Volume-of-Interest (VOI) Definition
The uptake in different organs was determined using manually defined ROIs and the Clinical Application Programming Package (CAPP; provided with the ECAT software). 89Zr uptake was determined in the following organs: lung, liver, spleen, kidney, heart, and, if visible, thyroid. In addition, a semiautomatic VOI was defined over the tumor using in-house–developed software tools (22).
Details of ROI/VOI Definition
As most organs could be visually best identified on the 1-h-after-injection emission scans, most organ 3-dimensional (3D) VOIs were defined manually using this early uptake scan. However, VOIs for the thyroid and tumor were defined on the last scan, because their delineation was optimal at this last time point. Finally, lung regions were defined on reconstructed transmission scans, using the semiautomatic isocontour tool within the CAPP software. All 3D VOIs were defined using a coronal view of the PET images.
A 3D VOI was generated from multiple 2-dimensional ROIs by grouping these ROIs into the VOI. The ROIs on the first and last slices were not used for the VOIs, because these were prone to partial-volume effects (spill-in and spill-out) or sampling errors. The VOIs defined on the 1-h-after-injection scan were saved and imported to the other time frames, for which the organs were not as clearly visible. To allow for projection of VOIs onto scans obtained at other intervals, we scanned patients in the same (patient and bed) position during subsequent studies, using rigid head immobilization devices (e.g., radiotherapy mask) and belts around the patient. To ensure correct positioning of the VOIs, the elevation of the bed was registered for all scans and in the case of differences in patient or bed position, scans were aligned using an in-house–developed software program. Furthermore, in the case of remaining dislocation of VOIs, all VOIs were relocated in 1 step to keep the internal relations intact. For relocation, the alignment of lung VOIs and the contour of the head and neck were verified using reconstructed transmission scans (for each subsequent study) as well. However, if still necessary, VOIs were ungrouped and individual VOIs were relocated to improve the position. After this VOI positioning procedure, all VOIs were saved and projected onto the emission scans. The mean uptake (in Bq/cm3) was then derived using these final VOIs.
The tumor was defined on the 144-h-after-injection scan using a tool that semiautomatically defined a 3D 50% (to maximum) isocontour around the tumor. Then the mean uptake (in Bq/cm3) was calculated. The mean tumor uptake was determined for the latest acquired PET study only, as correct repositioning of the tumor VOI onto earlier scans could not be reliably visually verified because of the lack of signal from the tumor.
All VOIs set on organs and tumors were evaluated by 2 experienced nuclear physicians.
Internal Radiation Dosimetry
The internal radiation dosimetry for the adult human was evaluated through the normalized cumulated activities for each patient provided as input to the OLINDA/EXM 1.0 code (23).
Residence times were calculated for liver, kidneys, lungs, spleen, and the remainder of the body, entering the percentage of the injected dose at each time point for each patient in OLINDA/EXM 1.0 and fitting these data using a monoexponential function. The residence times for the thyroid were calculated using trapezoidal integration. The remaining area under the curve from the end of data collection until infinity was determined by considering physical decay only. The residence time of the remainder of the body was defined as 113 minus the sum of residence times of source organs. A total residence time for the entire body of 113 h is obtained in the case of no biologic clearance. The red marrow dose was estimated using sampled blood clearance data (24,25). The red marrow concentration was assumed to be 30% of the blood activity concentration (conversion factor, 0.3). In addition, the dose from the remainder of the body onto red marrow was considered as well.
RESULTS
Unless otherwise specified, all 89Zr activities are decay-corrected to time of injection.
For 1 patient (patient 18) imaged, not enough data were available for adequate dosimetric analysis. Regarding the evaluation of 89Zr immuno-PET to quantify tumor uptake, 4 patients (patients 1, 2, 13, and 18) were excluded from analysis because of the lack of a scan at 144 h after injection. Another patient (patient 6) was excluded because of delayed surgery 2 wk after injection.
Safety
89Zr-cmAb U36 was found to be safe and well tolerated in all subjects. Neither adverse reactions nor significant changes in earlier mentioned blood and urine parameters that could be related to the study drug were observed. The mean radiation dose rates measured at 1, 24, and 72 h after injection at a distance of 100 cm were 7.0 ± 0.3, 5.7 ± 0.3, and 3.8 ± 0.2 μSv/h, respectively. A human antichimeric antibody (HACA) response developed in patients 9 and 10, and elevated titers were found at 1 and 6 wk after injection, irrespective of whether cmAb U36 IgG or 89Zr-N-sucDf-cmAb U36 was used in the enzyme-linked immunosorbent assay. These data indicate that the response was directed to the protein part of the conjugate and not to the N-sucDf chelate attached to the cmAb.
Biodistribution
Images of a representative male patient between 1 and 144 h after injection of 89Zr-cmAb U36 are presented in Figure 1. Whole-body images obtained directly after the administration of 89Zr-cmAb U36 showed mainly blood-pool activity, with delineation of the heart, lungs, liver, kidneys, spleen, and nose. Uptake of radioactivity for most organs decreased over time, whereas increased uptake was seen only at tumor sites and in the thyroid of some of the patients (patients 1, 6, 7, 10, and 11). The estimated uptake of 89Zr-cmAb U36 in single organs is presented in Table 2. The visual quality of the immuno-PET images varied between different patients. Figure 2 illustrates the difference in visual quality between an average-weight patient (58 kg) and an obese patient (104 kg).
Representative coronal images of male patient with oropharyngeal tumor (indicated by arrows), arranged (left to right) from 1, 24, 72, and 144 h after injection. (A) Increased uptake in time of 89Zr-cmAb U36 in tumor (indicated by arrows), (B) Circulating 89Zr-cmAb U36 in heart and uptake in organs. (C) Maximum-intensity projections of same patient as shown in A and B. Gray scale settings were set for each image independently, for clarity.
Difference in visual quality of coronal PET images obtained at 1 h after injection of 89Zr-cmAb U36 between average-weight patient (58 kg) (A) and obese patient (104 kg) (B).
Uptake of 89Zr-cmAb U36 in Single Organs
PET-Derived Blood-Pool Activity Versus Sampled Blood
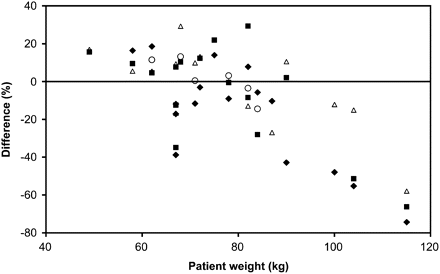
Variation in visual image quality also affected the quantitative analysis of the 89Zr-cmAb U36 immuno-PET images in a proportion of patients, as shown in Figure 3. This plot depicts the percentage under- and overestimation of the blood activity as assessed by immuno-PET. To this end, immuno-PET–derived activity in the left ventricle of the heart was compared with activity in sampled blood. Patients with a weight lower than 100 kg showed, in general, a good agreement with manually sampled data (difference equals 0.2% ± 16.9% [mean ± SD]). Patients with a weight more than 100 kg showed an underestimation of activity by immuno-PET, especially for later images. Figure 4 shows blood kinetics, assessed either by immuno-PET or by blood sampling, for the whole group of evaluable patients (n = 19). For the group of patients as a whole, no statistically significant (P = 0.503) differences were observed between the 2 methods used for assessment of activity. In addition, the small variances suggest a consistency of the pharmacokinetics in these patients, even for those in whom a HACA response developed. Therefore, we decided to use data on all these patients for radiation dose estimations.
Difference between PET-assessed blood-pool activity and sampled blood activity of 89Zr-cmAb U36 as function of patient weight. Difference in activity was assessed at 1 (▵), 24 (○), 72 (▪), and 144 h (♦) after injection.
Mean 89Zr-cmAb U36 activity in blood (Bq/cm3) of study population: PET-assessed blood-pool activity within left ventricle of heart (▪) and sampled blood-pool activity (▴).
Quantification of mAb Uptake in Tumors
Antibody uptake was assessed on 144-h-after-injection scans for all primary tumors and compared with uptake data from biopsies obtained at 168 h after injection. Comparison of %ID/g derived from biopsy and PET data showed a good agreement with slightly lower values for PET (mean deviation, −8.4% ± 34.5%). The mean tumor uptake at 168 h after injection as assessed by biopsies appeared to be 0.019 ± 0.010 %ID/g (range, 0.006–0.038 %ID/g).
Red Marrow Doses
The doses absorbed in the red marrow were estimated in 19 patients, as presented in Table 3. The mean red marrow dose was 0.07 ± 0.02 mSv/MBq in men and 0.09 ± 0.01 mSv/MBq in women.
Absorbed Doses in Red Marrow
Absorbed Doses in Source Organs and Whole Body
Residence times of the source organs were entered into the OLINDA/EXM 1.0 program. The results of the OLINDA/EXM 1.0 analysis are listed in Table 4. The effective dose for each patient was calculated using the available organ dosimetry data of the source organs. Excretion of 89Zr via the urinary pathway was 2.59 ± 1.89 %ID during the first 72 h after injection. As there was also little activity seen in the intestines, most loss of radioactivity was due to physical decay. The mean effective dose for the whole body was 0.53 ± 0.03 mSv/MBq in men and 0.66 ± 0.03 mSv/MBq in women. As an alternative, the effective dose was also calculated by considering the body to be a homogeneous mass, that is, by ignoring organ doses and assuming a residence time of 113 h for the entire patient. This simplified procedure showed only a minor decrease of the estimated effective dose: 0.44 ± 0.002 mSv/MBq in men and 0.54 ± 0.002 mSv/MBq in women. The difference in estimated effective dose between men and women (with vs. without, ignoring the biodistribution) suggests that this difference might be explained by use of different models (men vs. women) within OLINDA/EXM 1.0.
Absorbed Organ Doses
The normal organs with the highest absorbed dose were the liver (mean dose in men, 1.25 ± 0.27 mSv/MBq; in women, 1.35 ± 0.21 mSv/MBq) and kidneys (mean dose in men, 0.82 ± 0.15 mSv/MBq; in women, 1.18 ± 0.26 mSv/MBq).
DISCUSSION
The purpose of this study was to assess the safety and to evaluate the biodistribution, radiation dose, and potential for quantification of immuno-PET with 89Zr-labeled cmAb U36 in HNSCC patients. In this study, the tracer was found to be safe and well tolerated. No adverse events occurred. A HACA response was seen only in 2 patients, whereas none of the antibody responses was directed to the chelate. In all normal organs, the uptake of radioactivity decreased in time. Only in the tumor and in a few patients in the thyroid, uptake increased in time, suggesting specific uptake of 89Zr-cmAb U36. Such variable and sometimes high thyroid uptake was previously observed in HNSCC patients who had been injected with 99mTc-cmAb U36 (16). This result might indicate that in some individuals CD44v6 is expressed in the thyroid.
Furthermore, the advantage of the more detailed images obtained with 89Zr immuno-PET is the possibility of noninvasive quantification. In most of the images, the visual quality was acceptable for defining ROIs; however, there was a variation between images of different patients, and in some images the delineation of organs and tumor was suboptimal. Nevertheless, quantification results seem plausible. The 89Zr calibration procedure indicated that the quantitative accuracy of the scanner was not compromised by the presence of 909-keV γ-photons emitted from 89Zr and due to emission spillover into the transmission scans. In fact, the procedure was repeated for activity concentrations of about 17 kBq/mL (111 MBq in a phantom of 6,283 mL), showing that quantitative accuracy was not affected at higher counting rates as well. Although the 909-keV photons could have resulted in increased dead time and randoms fraction, the 89Zr activity in the field of view is much lower than usually applied (e.g., for 18F-FDG studies, up to 370 MBq of 18F-FDG is applied for patients and about 70 MBq is applied in a 6-L phantom for calibrations). Moreover, the 511-keV photon flux from 89Zr is much lower than that seen in 18F-FDG studies because of lower positron emission abundance. Another effect that could hamper quantification might be emission spillover into the transmission scan. However, emission spillover into the transmission scan is minimized in 2 ways. First, transmission scans are based on coincidence counting of the 511-keV photons emitted by the 68Ge transmission rod sources, thereby reducing the detection of noncoincident emitted photons (although these still result in increased randoms fraction). Second, during transmission scanning a rod windowing technique is applied (26). Rod windowing discards all detected coincidences that do not intersect the rod source (within a certain distance), and, as such, it reduces the influence of scatter, randoms, and emission spillover. To further validate quantitative accuracy, a comparison was made between blood-pool activity seen in the PET images and activity seen in manual samples.
The quantification of blood-pool activity in the left ventricle of the heart agreed well with the sampled blood activity, except for a few heavy-weight patients (>100 kg), suggesting that immuno-PET with 89Zr-cmAb U36 can be used to quantify the radiation dose of the whole body and normal organs of interest. However, as shown earlier with 18F-FDG PET, excessive body weight has negative effects on both quantitative and qualitative scan analysis (27–30). For 89Zr studies, these effects may be even more pronounced because of the low positron emission abundance resulting in lower noise-equivalent counting rates than seen with 18F-FDG studies. The low noise-equivalent counting rates could potentially hamper the accuracy and precision of the scatter correction, but further studies are required to fully understand the negative bias seen with heavier patients. Yet the association between patient weight and bias suggests that this effect is indeed related to scatter-correction issues. Despite this fact, a good agreement between PET and sample-derived blood-pool activities was seen for most other scans and subjects (Fig. 3). In future studies, with modern PET/CT scanners in combination with time-of-flight reconstruction and optimized scan protocols, it is probable that these problems will be of less magnitude, but this would require further validation as well.
Also in the assessment of tumor uptake, a good agreement was found between PET-derived data and data obtained from biopsies, despite the fact that there was a 1-d difference in assessment time (144 h after injection vs. 168 h after injection). This good agreement is quite remarkable because both methods are prone to errors. For example, for uptake assessment in tumor biopsies it is difficult to take a representative part of the tumor. For PET assessment, partial-volume effects will cause an underestimation of the tumor dose. This problem with partial-volume effects is a known limitation of PET, and correction methods are currently being developed to deal with this. At this point we can at least identify patients with low and high tumor uptake; therefore, it is possible to select the patients who may and may not benefit from therapy.
Although 89Zr immuno-PET can become an important tool in the detection and treatment of cancer, there are some limitations to overcome. Drawing of the organ VOIs requires anatomic knowledge and training and is time-consuming. However, with modern PET/CT scanners, 89Zr immuno-PET data can be collected, along with a structural CT image, in 1 scanning session. The aligned CT data can then be used for a more accurate and automated VOI definition.
The mean radiation dose for patients in this study was about 40 mSv, which is high and will limit repeated application of 89Zr immuno-PET. However, the introduction of the new-generation PET/CT scanners will also allow better-quality immuno-PET images to be obtained with a lower 89Zr radioactivity dose. This can be concluded from preliminary PET/CT studies in which 37 MBq of 89Zr-trastuzumab was used for human epidermal growth factor receptor type 2 immuno-PET in breast cancer patients (15). Using 89Zr-cmAb U36 in combination with these scanners would mean an effective dose of about 20 mSv for a whole-body scan. Theoretically, shorter-lived residualizing positron emitters such as 64Cu (t1/2, 12.7 h) and 86Y (t1/2, 14.7 h) might give less dose exposure, although clinical experience with these positron emitters is limited. In the study by Cutler et al., immuno-PET with 370 MBq of 64Cu-labeled intact mAb 1A3 was evaluated for detection of colorectal cancer with a Siemens/CTI ECAT EXACT PET scanner, as was also used in the present study (31,32). The average whole-body dose for these patients was 11.1 mSv. However, because of the short half-life of 64Cu, most of the patients had to be imaged within 24 h after injection to obtain good-quality images. As shown in Figure 1A, for cmAb U36 the delineation of tumors is much better at later time points (72 and 144 h), and, therefore, the use of 64Cu might be a less suitable alternative. For the immuno-PET evaluation of antibody fragments, however, 64Cu and 86Y might be suitable candidates.
CONCLUSION
In the present study, we evaluated the safety, biodistribution, radiation dose, and potential for quantification of immuno-PET with 89Zr-labeled cmAb U36 in HNSCC patients. 89Zr-cmAb U36 was found to be safe and well tolerated in all subjects. Uptake of radioactivity in the heart, lungs, liver, kidneys, and spleen decreased over time, and increased uptake was seen only at tumor sites and in the thyroid of some of the patients. Quantitative analysis, comparing PET-derived blood-pool activity and sampled blood activity, showed good agreement, except for the patients with a body weight of more than 100 kg. Also, a good agreement was found for the assessment of antibody uptake in tumors. The mean effective dose for the whole body was 0.53 ± 0.03 mSv/MBq in men and 0.66 ± 0.03 mSv/MBq in women.
Acknowledgments
This study was supported by the Dutch Cancer Society, grant IKA VU2000-2155. This study was partly performed within the framework of the Center for Translational Molecular Medicine (CTMM; www.ctmm.nl) project AIRFORCE 03O-103.
Footnotes
-
COPYRIGHT © 2009 by the Society of Nuclear Medicine, Inc.
References
- Received for publication May 6, 2009.
- Accepted for publication July 20, 2009.