Visual Abstract
Abstract
Cardiac transthyretin amyloidosis is an infiltrative cardiomyopathy with high mortality. To date, there are no specific biomarkers to directly assess disease activity and response to specific treatments. Our aim was to evaluate scintigraphic changes after treatment with the transthyretin stabilizer tafamidis. Methods: We included patients who had undergone 99mTc-3,3-diphosphono-1,2-propanodicarboxylic acid (99mTc-DPD) scintigraphy before tafamidis initiation and after at least 9 mo. Tracer activity was assessed visually and quantitatively as SUVmax. Results: The study included 14 patients who were on tafamidis for 44 ± 14 mo. We observed regression of Perugini grade in 5 patients, unchanged grade in 9 patients, and regression of mean heart–to–contralateral-lung ratio (P = 0.015) and SUVmax (P = 0.005). There were no changes in N-terminal pro-B-type natriuretic peptide or echocardiographic measures. Conclusion: Treatment with tafamidis results in regression of myocardial 99mTc-DPD uptake. 99mTc-DPD scintigraphy may provide useful imaging biomarkers to assess response to treatment.
Tafamidis is a first-in-class transthyretin stabilizer and currently the only approved treatment for cardiac transthyretin-type amyloidosis (ATTR). Implementation of disease-modifying therapies to stabilize or decelerate this inherently progressive disease has unveiled the need to identify reliable markers of disease activity. This is particularly challenging since it is largely unknown whether amyloid infiltration and the resulting myocardial damage are reversible and at which time point relevant changes can be expected. Current recommendations suggest the evaluation of standard clinical parameters of heart failure severity such as New York Heart Association (NYHA) class, serum biomarkers, and echocardiographic parameters (1). In addition to these traditional but nonspecific markers, further assays may be needed for a comprehensive assessment of response to antiamyloid treatments.
Serial imaging has the potential to identify novel markers of disease activity and disentangle the complex pathophysiology of amyloidosis. MRI with extracellular volume estimation provides a surrogate of myocardial amyloid burden. A previous study demonstrated extracellular volume regression or stabilization in 13 of 16 patients with hereditary cardiac ATTR who received patisiran (2). In the same study, visually assessed tracer uptake declined in 15 of 16 patients with serial 99mTc-labeled 3,3-diphosphono-1,2-propanodicarboxylic acid (99mTc-DPD) scintigraphy. Recently, Rettl et al. showed that treatment with tafamidis delayed the extracellular volume expansion compared with untreated historical controls in serial MRI scans (3). The current study aimed to perform a comprehensive analysis of visual, semiquantitative, and quantitative measures of 99mTc-DPD uptake in patients with cardiac ATTR under long-term tafamidis therapy.
MATERIALS AND METHODS
This analysis retrospectively included patients with cardiac ATTR under tafamidis who had undergone 99mTc-DPD scans, with SPECT/low-dose CT of the chest for diagnostic purposes before treatment initiation and after at least 9 mo of treatment. All patients received echocardiography and laboratory testing as part of a standardized follow-up. Our analysis included SPECT/CT-based quantitation of tracer uptake, which is inherently more accurate than visual or semiquantitative grading methods (4,5). The detailed scintigraphic protocol and quantitative method were previously published (5). Briefly, SPECT/CT systems (Symbia T2 and Intevo; Siemens Healthineers) were calibrated by phantom measurements. We used a whole-heart segmentation method to overcome the limitations of anatomic segmentation in noncontrast low-dose CT. The calculated voxel-based activity concentration was decay-corrected and converted to SUVs normalized to body weight. For comparative analysis, all studies were conducted with identical radiotracer, camera, and acquisition conditions. Two masked nuclear medicine physicians analyzed the images. The following quantitative measures were estimated: heart–to–contralateral-lung (H/CL) ratio, SUVmax, and quantitative ratios of uptake in the myocardium to the blood pool and in the myocardium to the vertebral bone. The institutional review board approved this retrospective study, and the requirement to obtain informed consent was waived (20-9278-BO).
Descriptive statistics are reported as means and SDs for continuous variables and counts and percentages for categoric variables unless indicated otherwise. Variables at baseline and follow-up imaging were compared with the paired t test for interval scaled variables and with the Wilcoxon matched-pairs signed rank test for ordinal scaled variables. All statistical tests and CIs were 2-sided, and P values of less than 0.05 were considered statistically significant. All analyses were performed with GraphPad Prism (version 9.1.1; GraphPad Software).
RESULTS
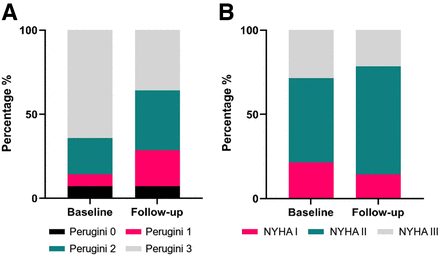
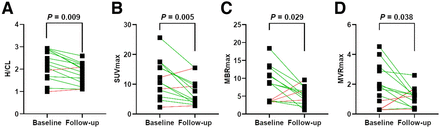
The study included 14 patients, 11 with wild-type and 3 with hereditary cardiac ATTR. The mean age was 76 y, and 79% were male. At baseline, NYHA I class symptoms were reported in 3 patients, NYHA II in 7, and NYHA III in 4. Perugini grades of 0, 1, 2, and 3 were found in 1, 1, 3, and 9 patients, respectively. After uninterrupted tafamidis therapy with 61 mg once daily for 44 ± 14 mo, we reported a stable or improving NYHA class in all but 3 patients (Fig. 1). Scintigraphic images were acquired 197 ± 31 min after injection with a mean injected activity of 547 ± 60 MBq of 99mTc-DPD. In 1 patient, only planar scintigraphy was performed at follow-up. Our analysis showed regression of Perugini grade in 5 patients and an unchanged grade in 9 patients but no increases. One patient improved from Perugini 3 to 1, and the other 4 patients had improvement by 1 grade (Table 1). Quantitative analysis revealed regression of H/CL ratio (2.2 vs. 1.8, P = 0.015), SUVmax (11.9 vs. 7.5, P = 0.005), myocardium–to–blood-pool SUVmax ratio (8.5 vs. 5.2, P = 0.029), and myocardium–to–vertebral bone SUVmax ratio (1.9 vs. 1.1, P = 0.038), as depicted in Figure 2. A representative case of a 73-y-old woman after 57 mo of tafamidis treatment with regression of Perugini grade from 3 to 1 alongside decreasing H/CL ratio (from 2.1 to 1.4) and SUVmax (from 10.7 to 3.4) is shown in Figure 3. Scintigraphic findings of the study population are provided in Figure 4. There were no significant changes in N-terminal pro-B-type natriuretic peptide (4,872 vs. 3,919 pg/mL, P = 0.24) or in several echocardiographic measures of systolic and diastolic function, including left ventricular wall thickness, muscle mass index, global longitudinal strain (mean change: +1%, −11.9 vs. −10.9%, P = 0.29), and ejection fraction (mean change: −2.8%, 53.0 vs. 50.2%, P = 0.08). We found no correlation between absolute changes in laboratory and echocardiographic parameters and H/CL ratio or SUVmax.
Changes in visual Perugini grade (A) and NYHA functional class (B) under treatment with tafamidis.
Study Results
Changes in quantitative measures of 99mTc-DPD uptake at baseline (before tafamidis initiation) and follow-up: H/CL ratio (A), SUVmax (B), MBRmax (myocardium–to–blood-pool SUVmax ratio) (C), and MVRmax (myocardium–to–vertebral bone SUVmax ratio) (D).
Whole-body planar 99mTc-DPD scintigraphy and corresponding SPECT/CT demonstrating regression of myocardial tracer accumulation after tafamidis treatment in 73-y-old woman with hereditary cardiac ATTR. Scintigraphic and SPECT/CT parameters from baseline to follow-up changed accordingly: visual Perugini, 3 vs. 1; SUVmax, 10,7 vs. 3.4; myocardium–to–blood-pool SUVmax ratio, 8.9 vs. 1.3; myocardium–to–vertebral bone SUVmax ratio, 1.9 vs. 0.3. Color scale is in kilocounts.
Planar scintigraphy findings at diagnosis and after treatment with tafamidis.
DISCUSSION
The central finding of this study is that most patients exhibit regression of 99mTc-DPD uptake in response to tafamidis, as assessed by quantitative techniques. Even though the mechanism of bone tracer affinity in ATTR is not yet elucidated, the observed changes generate a new hypothesis regarding the long-term effects of tafamidis beyond the targeted stabilization of transthyretin in its tetrameric form. A possible explanation for the regression of tracer uptake is that the inhibition of de novo amyloidogenesis may enable removal or degradation of the deposited amyloid fibrils and a true reduction in the myocardial amyloid burden. A previous report on 20 untreated cardiac ATTR patients who underwent serial scintigraphic assessment demonstrated no relevant changes in H/CL ratios after a median of 1.5 y (6), supporting the hypothesis that regression of tracer accumulation may reflect subsidence of the disease and a possible treatment effect.
Recently, preliminary data were reported from 2 ongoing randomized controlled trials using the small interfering RNAs vutrisiran and patisiran in patients with ATTR. The HELIOS A trial included patients with hereditary ATTR with polyneuropathy and compared vutrisiran with historic cohorts of patisiran- and placebo-treated patients from the APOLLO trial. In a subgroup of patients with cardiac involvement who were treated with vutrisiran, bone scintigraphy was repeated at 18 mo. Perugini grade was unchanged in 68% (39/57) and was decreased in 28% (16/57) of patients receiving serial scintigraphy, and only 3.5% (2/57) exhibited higher Perugini scores at follow up. H/CL ratio decreased in 65% (31/48) of patients treated with vutrisiran (7). In the APOLLO-B trial, which investigates patisiran versus placebo in patients with cardiac ATTR, scintigraphic changes at 12 mo were one of the selected exploratory endpoints. In 100% of evaluable patients in the patisiran arm (n = 37), Perugini grade decreased or demonstrated no change from baseline at 12 mo, and 38% of patients demonstrated a decrease in Perugini grade. No patients in the placebo arm (n = 28) demonstrated a decrease in Perugini grade at follow-up (8).
However, there is still a lack of robust data to provide evidence of amyloid removal, and these exploratory findings await confirmation by prospective studies with serial multiparametric assessment of myocardial function. Another hypothesis for the regression of uptake could be that antiamyloid treatments offer the myocardium time to start a healing process and, thus, indirectly induce changes in the molecular properties of the deposited amyloid fibrils or the amyloid microenvironment so that the tracer affinity for amyloid is reduced. It is also possible that regression of 99mTc-DPD uptake is a perfusion-dependent phenomenon due to tracer redistribution in other compartments, such as muscles, soft tissues, and bone, despite stability or even progression of myocardial amyloid infiltration.
The strength of the current study lies in the quantitative image analysis and the multiparametric assessment over a long follow-up period. The study carries the inherent limitations of retrospective data analysis. There were no untreated patients with longitudinal 99mTc-DPD scintigraphy who could serve as a control group. The small study sample precludes a safe conclusion regarding the correlation of imaging findings with echocardiographic and biochemical indices of cardiac function or clinical outcomes.
CONCLUSION
In this report, we demonstrate the effect of tafamidis on scintigraphic and SPECT-derived biomarkers and its potential to reflect disease activity. It is of great interest to validate these results in larger cohorts and correlate the observed changes with multiple other disease markers to establish reliable biomarkers for guidance and monitoring of cardiac amyloid burden in response to disease-modifying therapy.
DISCLOSURE
This work was supported by the Universitaetsmedizin Essen Clinician Scientist Academy (UMEA) and the German Research Foundation (DFG, Deutsche Forschungs-Gemeinschaft; research grant FU356/12-1 to Maria Papathanasiou). No other potential conflict of interest relevant to this article was reported.
KEY POINTS
QUESTION: Are there any changes in myocardial bone tracer uptake after treatment with tafamidis for cardiac ATTR?
PERTINENT FINDINGS: In this study involving 14 patients with cardiac ATTR, treatment with the transthyretin stabilizer tafamidis resulted in regression of myocardial tracer uptake at a median of 44 mo after treatment initiation.
IMPLICATIONS FOR PATIENT CARE: Changes in quantitative measures of bone tracer uptake by the myocardium may reflect disease activity and facilitate assessment of response to disease-modifying therapies.
Footnotes
Published online Jun. 8, 2023.
- © 2023 by the Society of Nuclear Medicine and Molecular Imaging.
- Received for publication December 21, 2022.
- Revision received April 14, 2023.