Abstract
The International Atomic Energy Agency sponsored a large, multinational, prospective study to further define PET for risk stratification of diffuse large B-cell lymphoma and to test the hypothesis that international biological diversity or diversity of healthcare systems may influence the kinetics of treatment response as assessed by interim PET (I-PET). Methods: Cancer centers in Brazil, Chile, Hungary, India, Italy, the Philippines, South Korea, and Thailand followed a common protocol based on treatment with R-CHOP (cyclophosphamide, hydroxyadriamycin, vincristine, prednisolone with rituximab), with I-PET after 2–3 cycles of chemotherapy and at the end of chemotherapy scored visually. Results: Two-year survivals for all 327 patients (median follow-up, 35 mo) were 79% (95% confidence interval [CI], 74%–83%) for event-free survival (EFS) and 86% (95% CI, 81%–89%) for overall survival (OS). Two hundred ten patients (64%) were I-PET–negative, and 117 (36%) were I-PET–positive. Two-year EFS was 90% (95% CI, 85%–93%) for I-PET–negative and 58% (95% CI, 48%–66%) for I-PET–positive, with a hazard ratio of 5.31 (95% CI, 3.29–8.56). Two-year OS was 93% (95% CI, 88%–96%) for I-PET–negative and 72% (95% CI, 63%–80%) for I-PET–positive, with a hazard ratio of 3.86 (95% CI, 2.12–7.03). On sequential monitoring, 192 of 312 (62%) patients had complete response at both I-PET and end-of-chemotherapy PET, with an EFS of 97% (95% CI, 92%–98%); 110 of these with favorable clinical indicators had an EFS of 98% (95% CI, 92%–100%). In contrast, the 107 I-PET–positive cases segregated into 2 groups: 58 (54%) achieved PET-negative complete remission at the end of chemotherapy (EFS, 86%; 95% CI, 73%–93%); 46% remained PET-positive (EFS, 35%; 95% CI, 22%–48%). Heterogeneity analysis found no significant difference between countries for outcomes stratified by I-PET. Conclusion: This large international cohort delivers 3 novel findings: treatment response assessed by I-PET is comparable across disparate healthcare systems, secondly a negative I-PET findings together with good clinical status identifies a group with an EFS of 98%, and thirdly a single I-PET scan does not differentiate chemoresistant lymphoma from complete response and cannot be used to guide risk-adapted therapy.
- diffuse large B-cell lymphoma
- positron emission tomography
- prospective observational study
- risk stratification
- risk-adapted therapy
Large studies of diffuse large B-cell lymphoma (DLBCL) in western populations have demonstrated event-free survivals (EFSs) with R-CHOP (cyclophosphamide, hydroxyadriamycin, vincristine, prednisolone with rituximab) of 79% at 3 y in adults aged younger than 60 y and 75% at 2 y in patients of all ages (1,2).
Much effort has been invested to prospectively discriminate between patients with a high probability of prolonged EFS and those who are unlikely to be cured by standard therapy so that their chance of cure may be increased by early intensification. Indicators including the International Prognostic Index (IPI), cell-of-origin tissue phenotype, and gene expression profiles have defined subgroups with predicted better or worse outcomes (3–6). Investigational high-throughput gene sequencing is revealing the biological heterogeneity of DLBCL, which may guide more targeted treatment in the future, but until now prospective personalization of each patient’s treatment remained elusive (7,8).
Proof of principle that the speed of response to treatment as an indicator of tumor chemosensitivity and ultimate cure was first demonstrated to be a powerful predictor of individual outcome in childhood lymphoblastic leukemia (9). PET uses preferential accumulation within tumor cells of 18F-FDG to measure glucose metabolic activity as a surrogate for cell viability. The hope has been that the speed of metabolic response, as judged by reduction in intensity of 18F-FDG uptake by the tumor early in treatment as a marker of chemosensitivity, might similarly identify rapidly responding cases with a high likelihood of cure and incomplete responders who would benefit from early treatment intensification.
A decade of studies has demonstrated that rapid response on an interim PET (I-PET) scan after 2–4 cycles of chemotherapy predicts a comparatively better outcome, but to a variable degree between studies, whereas no study has identified a I-PET–stratified patient group with a high enough probability of treatment failure to provide sound clinical basis for directing therapy (10–12). Most studies report single-center experience in Europe or North America where PET is well established.
At a time when PET scanning is increasing in developing countries, we questioned whether ethnic, economic, and environmental diversity might result in different disease biology or whether advanced disease at presentation, compared with the western world, might confound the utility of risk prediction by an I-PET scan early in treatment. If so, it would limit the global generalizability of data from predominantly Caucasian populations in high-income countries.
To address these issues, the International Atomic Energy Agency (IAEA) initiated a Coordinated Research Project to examine PET monitoring for risk stratification of DLBCL in 5 geographic regions to inform international practice. Coordinated Research Projects, enshrined under Article III of the IAEA’s statute, facilitate the international development of the practical use of atomic energy for peaceful purposes and promote the bringing together of researchers in both developing and industrialized countries to solve a problem of common interest (13).
The primary aim of this prospective international cohort study was to define, with the greater precision afforded by a large cohort, whether the rate of response to treatment as assessed by a mid-treatment I-PET scan could achieve clinically useful prediction of outcomes at 2 y for individual patients. The secondary aim was to establish whether there was clinically important variation in PET-stratified outcomes between participating countries.
MATERIALS AND METHODS
The project was approved by the IAEA and protocol developed jointly at 2 investigator meetings in 2006 and 2008.
Eligibility Criteria and Treatment Protocol
Patients with DLBCL (age, ≥16 y) who had provided informed consent were recruited. Exclusions were cancer within the preceding 5 y, steroid therapy before the staging scan, and no 18F-FDG–avid disease on baseline PET. Diagnosis was based on biopsy with immunohistochemistry and classification by World Health Organization criteria (14). All patients were staged by PET/CT, or PET and CT separately, and iliac crest marrow biopsy.
The treatment protocol was for 6 cycles of R-CHOP at 21-d intervals. To accommodate clinician preference and local practice, up to 8 cycles was permitted. Omission of rituximab was allowed in recognition that some eligible patients might otherwise be excluded for financial reasons. Scan results were reported to treating clinicians, but modification to planned treatment on the basis of the I-PET response was not permitted. Treatment escalation in response to a positive I-PET result was classified as treatment failure (see the “Classification of Events” section).
Radiotherapy
Consolidation radiotherapy, if planned as part of primary treatment (e.g., to sites of bulk disease or to specific sites of extranodal disease), was permitted, as directed by local practice. Preplanned radiotherapy was deemed consolidation only if given after a negative end-treatment PET and confirmation of complete response defined by international criteria (15).
PET Scheduling and Reporting
Scans were required at 3 time points: before treatment, mid treatment (I-PET), and end-chemotherapy (E-PET). The I-PET scan was recommended after 2 cycles of chemotherapy, at a maximum interval from the preceding treatment (median treatment to scan interval, 18 d [interquartile range, 17–21 d]). In recognition of technical and scheduling constraints, I-PET after 3 cycles was permitted and in exceptional circumstances after 4 cycles. The protocol stipulated a minimum of a 4- to 8-wk interval between final chemotherapy and E-PET.
I-PET scan reporting was based on visual assessment and classified into 4 categories: negative/CR (resolution of abnormal 18F-FDG uptake at sites of disease identified on staging PET, with any residual 18F-FDG uptake less than or equal to the mediastinal blood pool), complete response with minimal residual uptake (CR-MRU) (residual low-level 18F-FDG uptake at disease sites greater than mediastinum but less than or equal to physiologic uptake in liver), positive (residual or increased 18F-FDG uptake with intensity greater than liver at a site of known disease), and mixed response (reduction in 18F-FDG uptake at some disease sites, with increased 18F-FDG uptake at other existing or new sites). For outcome analysis, scans scored as CR-MRU were grouped with PET-negative; mixed response was classified as PET-positive.
The study commenced in 2008, before the Deauville 5-point classification was devised in 2009 (15). The scoring scheme used here was based on its antecedent, the validated 5-point London Criteria, modified to combine Deauville-equivalent scores 1 and 2 into a single negative category and scores 4 and 5 into a positive category (16). CR-MRU (equivalent to Deauville 3) was reported as a separate score, as at that time it was uncertain whether or not this level of 18F-FDG uptake represented persistent disease at I-PET.
As recommended for multicenter PET-stratification studies, scans were reviewed by the 4 lead nuclear medicine physicians working together on a common platform at the final collaborator meeting (16,17). Reviewers were masked to clinical details and patient outcomes. Classification of PET response was by consensus.
Classification of Events
Study events were relapse after complete remission; death from any cause; treatment escalation for progressive disease while on treatment; and disease progression or failure to achieve complete remission at end-chemotherapy based on the revised response criteria for PET, with confirmation by biopsy that residual or increased 18F-FDG uptake was due to lymphoma (18). Each patient record was reviewed with the country chief investigator during the final collaborator meeting to ensure correct classification of events.
Research Regulation and Data Protection
Each country gained research ethics approval for the study protocol and patient information from the appropriate national or local Ethics Review Board. Fully informed consent was an inclusion criterion for recruitment. Signed consent forms were kept by the local investigators. To ensure confidentiality while sharing data internationally, cases were assigned a numeric code, and only 2 identifiers for data validation—initials and date of birth—were recorded in the central database (19).
Statistical Methods
Follow-up continued until 75% patients had reached 2 y or died. Cases lost to follow-up were censored at date of last known disease status. Survival was estimated using Kaplan–Meier methods, with the date of first treatment as origin.
The prognostic ability of I-PET is estimated from a Cox proportional hazards model (20). Between-country heterogeneity in the prognostic value of I-PET was explored using a 2-stage meta-analysis. Taking the Cox model based on I-PET as the sole covariate for each country, the overall effect of I-PET was estimated between countries by random country effects calculated using generalized Q statistics (21). Secondly, each country was omitted in turn to identify how each contributed to any differences identified.
To investigate whether I-PET adds prognostic discrimination beyond that of established factors, a Cox model was fitted with IPI and age as covariates. Other variables (stage, performance status, extranodal sites, lactate dehydrogenase [LDH], bulky disease > 5 cm, rituximab treatment, and I-PET timing [2 or 3+ cycles]) were chosen by a process of backward elimination, using a P value of 0.1 as the elimination criteria. Finally I-PET and E-PET classification, as positive or negative, were included as independent variables. A multivariable model was developed using prognostic factors chosen from this model to identify risk categories with a significant degree of prognostic separation. Analyses and graphs were produced using Stata 12 (StataCorp).
RESULTS
Major cancer centers in 9 countries (Sâo Paulo, Brazil; Santiago, Chile; Budapest and Debrecen, Hungary; Mumbai, India; Bologna, Italy; Seoul, South Korea; Manila, Philippines; Bangkok, Thailand; and Ankara, Turkey) from 5 United Nations–defined geographic regions participated in the study. Recruitment commenced in 2008 through September 2011. Of the 383 patients recruited, 56 were excluded, based on predefined eligibility criteria. Twenty-two did not meet recruitment criteria, and 34 could not be analyzed because the scans were not submitted for central review, leading to exclusion of all recruited Turkish cases (Fig. 1). Nine cases classified as primary mediastinal B-cell lymphoma were included. Of the 327 eligible cases, 52 were from low-middle income countries (India, Philippines), 170 from upper-middle (Brazil, Hungary, Thailand), and 105 from high-income countries (Chile, Italy, South Korea).
Consort diagram.
Patient Characteristics
Table 1 provides patient characteristics by country: 35% of patients were older than 60 y. The 8 contributing centers demonstrated good compliance with the protocol; 280 (86%) received rituximab (Table 2). The number of chemotherapy cycles varied between countries, with more than 6 cycles given most commonly for advanced or bulky disease; 66 patients were given consolidation radiotherapy after a negative E-PET result and confirmation of CR. I-PET timing was consistent with the protocol, with 77% scanned after 2 chemotherapy cycles, 99% after 2 or 3 cycles, and 1% after 4 cycles.
Patient and Disease Characteristics
Treatment, Monitoring, and Outcomes
Outcomes
At a median follow-up of 2 y 11 mo, the 2-y survival for all 327 cases was 79% (95% confidence interval [CI], 74%–83%) for EFS and 86% (95% CI, 81%–89%) for OS. Three-year survival was 71% (95% CI, 65%–76%) for EFS and 83% (95% CI, 78%–87%) for OS (Fig. 2). There were 93 events after I-PET, 51 deaths as the primary event and 42 treatment failures or relapses after remission. The 47 cases treated without rituximab had a 2-y EFS of 75% (95% CI, 60%–85%) versus 79% (95% CI, 74%–85%) with rituximab.
Kaplan–Meier plots of EFS (A) and OS (B) for entire eligible cohort. 95% CIs and number of cases at risk are shown; 2-y EFS was 79% (95% CI, 74%–83%) and OS was 86% (95% CI, 81%–89%).
Stratified by I-PET
The I-PET scan was negative in 210 (64%) and positive in 117 (36%) patients. Two-year EFS as stratified by I-PET was 90% (95% CI, 85%–93%) for I-PET–negative patients and 58% (95% CI, 48%–66%) for I-PET–positive patients, with a hazard ratio (HR) of 5.31 (95% CI, 3.29–8.56). Two-year OS as stratified by I-PET was 93% (95% CI, 88%–96%) for I-PET–negative patients and 72% (95% CI, 63%–80%) for I-PET–positive patients, with an HR of 3.86 (95% CI, 2.12–7.03) (Fig. 3).
Kaplan–Meier plots of EFS (A) and OS (B) for cases stratified by I-PET. 95% CIs and number of cases at risk are shown. Two-year EFS: I-PET–negative, 90% (95% CI, 85%–93%); I-PET–positive, 58% (95% CI, 48%–66%); and HR, 5.31 (3.29–8.56). Two-year OS: I-PET–negative, 93% (95% CI, 88%–96%), I-PET–positive, 72% (95% CI, 63%–80%); and HR, 3.86 (95% CI, 2.12–7.03).
Stratified by Sequential I-PET and End-Treatment PET
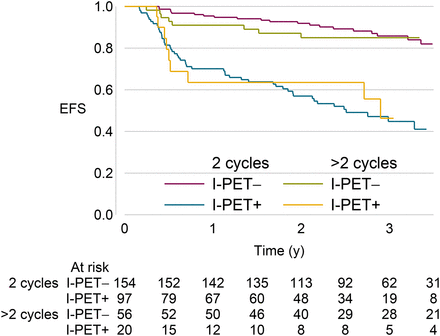
Fifteen cases did not have a study E-PET due to death (n = 12) or early treatment escalation (n = 3). Analysis of the 312 cases with both I-PET and E-PET scans showed that most cases (96%) clustered into 3 prognostic groups (Fig. 4). The largest, 192 (62%) cases, had negative I-PET and E-PET demonstrating a rapid response with an excellent 2-y EFS of 97% (95% CI, 92%–98%) and an OS of 97% (95% CI, 93%–99%).
Kaplan–Meier plots of EFS (A) and OS (B) for cases stratified by both I-PET and E-PET. Number of cases at risk is shown. I-PET–negative/E-PET–negative: EFS, 97% (95% CI, 92%–98%), OS, 97% (95% CI, 93%–99%); I-PET–positive/E-PET–negative: EFS, 86% (95% CI, 73%–93%), OS, 92% (95% CI, 79%–97%); I-PET–negative/E-PET–positive: EFS, 28% (95% CI, 7%–54%), OS, 64% (95% CI, 28%–86%); and I-PET–positive/E-PET–positive: EFS, 35% (95% CI, 22%–48%), OS, 60% (95% CI, 44%–73%).
The second group, 58 (19%) cases, had positive I-PET but negative E-PET and were in clinical remission at the end of chemotherapy; this slow-response group had a 2-y EFS of 86% (95% CI, 73%–93%) and OS of 92% (95% CI, 79%–97%). HRs comparing rapid- and slow-response cases show slow responders to have approximately double the risk of an event by 2 y, compared with those with a negative I-PET (HR for EFS, 2.56 [95% CI, 1.08–6.11]; for OS, 1.83 [95% CI, 0.61–5.51]).
The third largest group, 49 (16%) cases, had positive I-PET and E-PET scans, a 2-y EFS of 35% (95% CI, 22%–48%), and had continuing relapses beyond 2 y. In 7 of these cases, the E-PET appeared to be false-positive, with no residual disease if biopsied or continued clinical remission without additional chemotherapy; none of these 7 cases had consolidation radiotherapy. An additional 13 patients (4%) were I-PET–negative but E-PET–positive with 18F-FDG uptake at a previous or new site (EFS, 28% [7%–54%]); 11 of these patients (85%) had biopsy-confirmed disease progression.
In the slow-response group, bulky disease was more common, 67% versus 50% in the cohort overall (Table 1). Consolidation radiotherapy (after confirmed CR) was given to 19% of patients with bulky disease at diagnosis, compared with 20% of the cohort overall (Table 2).
Between-Country Heterogeneity
Survivals stratified by I-PET were compared for consistency of direction and magnitude across countries. Initial analysis of I-PET–stratified EFS showed modest between-country heterogeneity (P = 0.09; I2 = 65%). With Chile omitted from the analysis (see the “Materials and Methods” section), there was complete absence of heterogeneity among the other 7 countries (I2 = 0%). Heterogeneity for OS across all countries was low and similarly not significant (P = 0.4; I2 = 6.6%) (Fig. 5). Chile’s survival figures were characterized by the highest 2-y EFS (89%) and the lowest proportion of I-PET–positive patients (15%), all of whom had disease progression or died during treatment. These outcomes are sufficient to explain Chile’s noncongruence in the heterogeneity analysis. It is of importance, in the context of this study, that Chile is a high-income country, with healthcare relatively well resourced, and the study patients were predominantly of European origin. We therefore found no relationship between ethnicity, geographic region, or economic status and outcomes stratified by I-PET.
Between-country heterogeneity analysis of outcomes stratified by I-PET: EFS (A) and OS (B). Figures show forest plot of EFS/OS HR and 95% CI for I-PET–positive vs. I-PET–negative cases by country, combined meta-analysis HR, and estimate of heterogeneity, I2. These plots show all countries. Additional analyses explored contribution of each country to any differences identified (data not shown).
Additional Risk Factors
Multivariate analysis including the entire study population was performed to investigate the relative influence on EFS of the IPI and its components, bulky disease, rituximab treatment, timing of I-PET (after 2 vs. 3 cycles), and I-PET response status. IPI, age, stage, extranodal disease in 2 or more sites, and bulky disease were not significant variables, nor were timing of the I-PET (Fig. 6) or the omission of rituximab in 14% of cases. Only I-PET status, performance status, and LDH reached significance: positive I-PET, HR of 4.32 (2.64–7.10); performance status ≥ 2, 1.79 (1.05–3.07); and abnormal LDH, 1.66 (1.03–2.67). I-PET–negative cases with a performance status of 0–1 and normal LDH (n = 110) had an exceptionally good 2-y EFS (98% [92.0%–100%]).
I-PET–stratified EFS for cases by timing of I-PET: after 2 (n = 251) vs. after 3 (n = 73) or 4 (n = 3) cycles of chemotherapy.
The multivariate analysis was repeated to include E-PET status on the 312 cases with end-treatment scans. In this analysis, E-PET was strongly predictive of outcome, with an HR of 14.3 (7.74–22.45), in contrast to I-PET (HR, 1.16 [0.63–2.16]). Performance status and LDH remained the only predictive clinical variables. However, high performance status and raised LDH together with a positive I-PET did not necessarily predict poor outcome; 39% of I-PET–positive cases had these adverse clinical characteristics and yet were in PET-negative remission at completion of chemotherapy, and 84% of these were alive in first remission at 2 y.
Outcome of I-PET CR-MRU
We examined, as a secondary analysis, the outcome of cases in which the I-PET was reported as CR-MRU (n = 88), compared with I-PET–negative with no residual 18F-FDG uptake (n = 122) (Fig. 7). I-PET MRU and I-PET–negative cases had almost identical EFS and OS over time, justifying combining both as I-PET–negative in the analyses.
Outcome of cases with I-PET scans scored as minimal residual uptake. Kaplan–Meier plots compare EFS (A) and OS (B) of cases, with I-PET scans classified visually as negative, MRU, and positive.
DISCUSSION
This study was designed to define the international role of PET for risk stratification of DLBCL and to address the hypothesis that biological diversity or diversity of healthcare in different geographic regions may influence the kinetics of treatment response as assessed by I-PET.
In this cohort of 327 patients, EFSs were close to those reported from recent large European R-CHOP studies (1,2). Similarly, 2-y EFS and OS, stratified by I-PET response after 2–3 cycles chemotherapy, were comparable to recent studies from high-income countries, likewise using visual PET reporting and early I-PET scanning after 2–3 cycles (Table 3) (22–26).
EFS or PFS, Stratified by I-PET, for Studies of DLBCL Without Risk-Adapted Therapy
Analysis for heterogeneity of OS and EFS stratified by I-PET found little difference between countries. Therefore, any population heterogeneity based on clinical risk factors at diagnosis was normalized to deliver remarkable consistency of outcome prediction across participating countries when stratified by I-PET response. Consequently, our hypothesis that international diversity might limit the global applicability of western-generated PET data was not supported. Equally, the consistency of I-PET–stratified survivals across the different healthcare environments justifies pooling data from all countries for the study’s primary analysis.
This large prospective study demonstrates a highly significant difference between EFS of the I-PET–positive and –negative cohorts. Sequential monitoring, by both I-PET and E-PET, identified 4 risk groups with greater and clinically important separation between cases with good and poor prognosis. Two groups had good and 2 poor EFS at 2 y, 97% and 86%, versus 35% and 28%, respectively. The large cohort size not only provided more precise survival estimates, it also enabled a more informative multivariable analysis of clinical risk factors than previously possible. We identified that a substantial subgroup with complete metabolic response at both I-PET and E-PET, coupled with a marker both of more favorable tumor biology (normal LDH) and of patient fitness (good performance status, though not age), had an excellent EFS of 98%.
More important was the revelation that of 107 I-PET–positive cases, more than half (54%) became PET-negative by the end of chemotherapy, and most of these slow responders had durable remissions. This finding not only explains the inability of a single positive I-PET scan to predict poor outcome, it also indicates that to intensify therapy on this basis would put a significant number of patients at risk of unjustified treatment-related toxicity.
A slow but complete response group has been previously noted in 2 much smaller studies. In one, 15 of 25 (60%) patients with positive I-PET achieved complete response by E-PET (23). In another highly selective retrospective database study, 35 of 55 (63%) achieved remission after a positive I-PET (24). These smaller studies provide reassurance that our findings are not a unique phenomenon. Our large prospective cohort provides more definitive evidence that an early I-PET scan cannot be used to guide early treatment escalation.
In some, slow response may represent less chemosensitive disease, reflected by the increased event rate of the I-PET–positive/E-PET–negative subgroup, compared with the rapid response I-PET–negative/E-PET–negative cases reported here. In others, the likely explanation is that persistent 18F-FDG uptake in the tumor mass is due to inflammatory reaction within necrotic tumor rather than residual viable lymphoma (27). A positive I-PET due to inflammatory cells in the absence of tumor has previously been reported in a DLBCL study, which found cases with inflammatory cells only on biopsy had good outcomes (28). It has been suggested that this phenomenon is more common after rituximab (29).
Since the study commenced, there has been growing interest in standardized uptake value reduction (ΔSUVmax) at I-PET as a more sensitive method of separating good- from poor-outcome patients. Recent studies comparing visual with semiquantitative ΔSUVmax for predicting outcomes found ΔSUVmax to better predict progression-free survival (PFS)/EFS after 2 or 4 cycles than visual assessment, and when ΔSUVmax cutoff was optimized to the timing of I-PET, discrimination between those with and without residual lymphoma was further improved (30–32). Early response assessment using ΔSUV or volumetric analysis may evolve to a degree to which it could be justifiably used to guide risk-adapted therapy.
In the global healthcare context explored by this study, quantitative techniques may be less practicable because of the demanding conditions required for accurate and reproducible results. However, our study has demonstrated that multinational collaboration between developed and developing countries to test newer and more demanding methodologies is feasible and informative as well as providing mentoring and training for investigators.
Coordinated Research Projects provide limited financial support. This, and geographic logistics, prevented calibration of PET scanners to a common standard for this study, though there was central review of all PET scans. Central pathologic review was similarly not practicable, though diagnostic support was provided by 2 senior lymphoma pathologists. Despite these potential confounding factors, our outcomes are similar to those from the United States and Europe, with the strength that they reflect local practice.
At a time when health priorities in the developing world are shifting to noncommunicable disease, with calls to make cancer cure a global priority, it is important to demonstrate that data that underpin oncology practice in the developed world can be applied internationally (33). We found that geographic and population diversity did not influence I-PET–stratified outcomes.
CONCLUSION
A decade of investigation has sought to establish I-PET as a reliable indicator to guide early treatment intensification. This large international cohort delivers the strongest evidence that a positive I-PET result does not differentiate chemoresistant residual tumor from complete response, nor does it provide sound basis for early escalation of therapy in individuals with DLBCL.
APPENDIX The International Atomic Energy Agency Lymphoma Study Group
Austria–Vienna (IAEA)
Diana Paez*†, Maurizio Dondi*, Noura El-Haj
Brazil–Sâo Paulo
Juliano Cerci*†, Claudio Meneghetti†, Artur Coutinho, Jose Soares, Debora Levy, Segio Bydtowsky, Juliana Pereria, Renata Costa, Sheila Coelho
Chile–Santiago
Francisco Redondo†, Paulette Conget, Eva Bustamante, Flavia Bruna
France–Paris
Rose Ann Padua*†
Hungary–Budapest, Debrecen
Tamás Masszi†, Botond Timár, Ágota Szepesi, Andrea Sipos, Judith Demeter, Lajos Gergely, Ildikó Garai
India–Mumbai
Reena Nair†, Sumeet Gujral†, Venkatesh Rangarajan, Ranjan Basak
Italy–Bologna
Stefano Fanti*†, Monica Celli†, Pier Luigi Zinzani
South Korea–Seoul
June-Key Chung†, Keon Wook Kang, Jae Seon Eo
Philippines–Manila
Charity Gorospe†, Maejoy Vena Campo, Filipinas Natividad, Mark Piere Dinamay
Thailand–Bangkok
Chirayu Auewarakul†, Narongrit Sritana
Turkey–Ankara
Hilal Ozdag, Isinsu Kuzu†, Nilgun Tekin, Ozem Kucuk, Gulseren Aras, Melike Ozbilgin, Muhit Ozcan, Pervin Topcuolglu
U.K.–London
Robert Carr (Chief Scientific Investigator)*†, Tim Morris†
*Study Management Group.
†Author.
DISCLOSURE
The costs of publication of this article were defrayed in part by the payment of page charges. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734. The study was funded and supported by the International Atomic Energy Agency. No other potential conflict of interest relevant to this article was reported.
Acknowledgments
We thank, in particular, Baldip Khan and Maurizio Dondi for initiating and supporting the project under the umbrella of the IAEA.
Footnotes
Published online Nov. 26, 2014.
- © 2014 by the Society of Nuclear Medicine and Molecular Imaging, Inc.
REFERENCES
- Received for publication July 5, 2014.
- Accepted for publication October 27, 2014.