Abstract
A compendium of about 100 radiopharmaceuticals, based on the OLINDA/EXM version 2.0 software, is presented. A new generation of voxel-based, realistic human computational phantoms developed by the RADAR committee of the Society of Nuclear Medicine and Molecular Imaging, based on 2007 recommendations of the International Commission on Radiological Protection, was used to develop the dose estimates, and the most recent biokinetic models were used as well. These estimates will be made available in electronic form and can be modified and updated as models are changed and as new radiopharmaceuticals are added.
Radiation dose estimates for radiopharmaceuticals require extensive calculations and the use of anthropomorphic body models (computational phantoms). In 2003, the RADAR (RAdiation Dose Assessment Resource) method for internal dose estimation was established (1) and was implemented in the personal computer software code OLINDA/EXM 1.0 (2). The RADAR method is conceptually the same as the MIRD method (3): the time-integrated activity for organs with significant uptake of the radiopharmaceutical is multiplied by dose factors that give the absorbed dose in any target organ per disintegration in any source organ. This process is repeated for all identified source and target organs to obtain a total dose to all target organs. Many have mistakenly stated in publications that the OLINDA/EXM method implements the MIRD method for internal dosimetry, an understandable misconception. OLINDA/EXM implements the RADAR method for internal dose assessment; the first incarnation of this software was called MIRDOSE and did use the MIRD method, but the MIRD committee did not review or approve the software and vigorously opposed the use of its name. Thus, the RADAR group was formed, and the code was appropriately renamed. As noted, the concepts are identical, but the MIRD method uses the term à (cumulated activity) for the time–activity integral and calls the dose factor an S factor, whereas RADAR uses the terms N (number of disintegrations) and dose factor, respectively. The International Commission on Radiological Protection (ICRP) also has a method for internal dose calculations, originally described in ICRP publication 30 (4)—exactly the same concepts but with different symbols for the various quantities used. This schema has been repeated, with modifications, several times, the latest being in ICRP publication 130 (5).
A generic equation for the absorbed dose in any target organ can be given as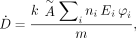 Eq. 1where D is absorbed dose in a target organ (rad or Gy), Ã is cumulated activity (sum of all nuclear transitions that occurred) in a source organ (μCi-h or MBq-s), n is number of radiations with energy E emitted per nuclear transition, E is energy per radiation (MeV), φ is absorbed fraction (fraction of radiation energy emitted from a source organ that is absorbed in a target organ), m is mass of the target region (g or kg), and k is a proportionality constant (rad-g/μCi-h-MeV or Gy-kg/MBq-s-MeV).
Eq. 1where D is absorbed dose in a target organ (rad or Gy), Ã is cumulated activity (sum of all nuclear transitions that occurred) in a source organ (μCi-h or MBq-s), n is number of radiations with energy E emitted per nuclear transition, E is energy per radiation (MeV), φ is absorbed fraction (fraction of radiation energy emitted from a source organ that is absorbed in a target organ), m is mass of the target region (g or kg), and k is a proportionality constant (rad-g/μCi-h-MeV or Gy-kg/MBq-s-MeV).
The MIRD method reduces this equation to the following: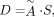 Eq. 2where à is defined as above and S is given by
Eq. 2where à is defined as above and S is given by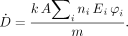 Eq. 3The term n in the later MIRD schema documents was given as Y (yield). In the ICRP system of radiation protection for workers (4), the dose equation is
Eq. 3The term n in the later MIRD schema documents was given as Y (yield). In the ICRP system of radiation protection for workers (4), the dose equation is Eq. 4Here, H is the dose equivalent (the absorbed dose, D, multiplied by a radiation weighting factor, wR, formerly known as a quality factor, Q). US is the number of nuclear transitions that occur in source region S, and SEE is
Eq. 4Here, H is the dose equivalent (the absorbed dose, D, multiplied by a radiation weighting factor, wR, formerly known as a quality factor, Q). US is the number of nuclear transitions that occur in source region S, and SEE is Eq. 5In many ICRP documents, slightly different names are given to some terms, such as AF (absorbed fraction) for φ and f for n, but all the concepts are identical.
Eq. 5In many ICRP documents, slightly different names are given to some terms, such as AF (absorbed fraction) for φ and f for n, but all the concepts are identical.
The RADAR method uses the following simple equation: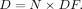 Eq. 6where N is the number of nuclear transitions that occur in source region S, and DF is the dose factor. The dose factor contains the various components shown in the formulas for S and SEE and depends on combining decay data with absorbed fractions, which are derived generally using Monte Carlo simulation of radiation transport in models of the body and its internal structures (organs, tumors, and other structures):
Eq. 6where N is the number of nuclear transitions that occur in source region S, and DF is the dose factor. The dose factor contains the various components shown in the formulas for S and SEE and depends on combining decay data with absorbed fractions, which are derived generally using Monte Carlo simulation of radiation transport in models of the body and its internal structures (organs, tumors, and other structures):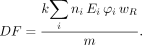 Eq. 7As written, Equations 2, 4, and 6 give only the dose from one source region to one target region, but when all source and target regions are used, a complete table of dose estimates for a radiopharmaceutical is obtained.
Eq. 7As written, Equations 2, 4, and 6 give only the dose from one source region to one target region, but when all source and target regions are used, a complete table of dose estimates for a radiopharmaceutical is obtained.
The current generation of anthropomorphic phantoms began with the development of the Fisher–Snyder phantom (6). This phantom used a combination of geometric shapes—spheres, cylinders, cones—to create a reasonably anatomically accurate representation of the body. All phantoms use Monte Carlo simulations of radiation transport (simulating the creation and transport of photons through these various structures in the body, whose atomic composition and density were based on literature data). The Fisher–Snyder phantom used the data provided in ICRP publication 23 (7). This report provided various anatomic data assumed to represent the average working adult man in the Western hemisphere. Although this was most often applied to adult men, this phantom also contained regions representing organs specific to the adult woman. Using this phantom, radiation doses were calculated for adults on the basis of activity residing in any organ and irradiating any other organ. Absorbed fractions at discrete photon energies were calculated and published by the MIRD committee (8). In addition, dose conversion factors, which, as noted above, they called S values, were developed. These represented the dose to a target region per nuclear transition in a source region; approximately 20 source- and target-region S values were defined and published for over 100 radionuclides (9).
The development of the series of phantoms by Cristy and Eckerman (10), representing children of various ages and adults, allowed dose calculations for individuals different in size and age. Six phantoms were developed, which were assumed to represent children and adults of both sexes. Absorbed fractions for photons at discrete energies were published for these phantoms, which contained approximately 25 source and target regions. Tables of S values never were published but ultimately were made available in the MIRDOSE computer software (11). The 15-y-old phantom was sometimes used as a model of the adult woman.
Stabin et al. developed phantoms for the adult woman, both nonpregnant and at 3 stages of pregnancy (12). These provided more specific models for the adult woman and replaced the Cristy–Eckerman 15-y-old phantom in dose calculations for women. These phantoms attempted to model the changes to the uterus, intestines, bladder, and other organs that occur during pregnancy and included specific models for the fetus, fetal soft tissue, fetal skeleton, and placenta. S values for these phantoms were also made available through the MIRDOSE software.
Other individual-organ models were developed separately and were incorporated in the MIRDOSE and OLINDA/EXM codes, including the brain (13), peritoneal cavity (14), prostate gland (15), bone and marrow (16), and small unit-density spheres (possibly to represent tumors, organs in small animals, or other structures) (17).
In an effort to provide the user community with data needed for dose calculations rapidly and in electronic form, the RADAR group was formed (18). It later attained the status of an official Society of Nuclear Medicine and Molecular Imaging committee. The group maintains an Internet website (www.doseinfo-radar.com) that provides information on internal and external dose assessment; most data are available directly from the site for immediate download. In this paper, we describe the models chosen and the methods applied by RADAR to calculate dose factors for use in internal dose assessment in nuclear medicine. The last time that dose factors for radionuclides of interest in nuclear medicine were published in this format by the MIRD committee was in 1975 (9). Dose factors, principally for nuclides of interest in nuclear medicine, were made available with the distribution of the MIRDOSE software (11), but the factors themselves were never published. The OLINDA/EXM 1.0 and 1.1 software codes (2) superseded the MIRDOSE code but were basically identical in method and in the models used. This article describes a major advance in the technology of computational phantoms—voxel phantoms based on medical images, rather than the stylized phantoms of the 1980s and 1990s—that are implemented in the new version (2.0) of OLINDA/EXM. This new version was used to develop a large compendium of tables of dose estimates for radiopharmaceuticals, based on the best current biokinetic models (available at http://www.doseinfo-radar.com/RADAR-INT-compendium.html; see also the online SNMMI tool that uses data from the compendium at http://www.snmmi.org/dosetool). These models change periodically; thus, their input data are not provided in the OLINDA/EXM codes. Biokinetic data for a new radiopharmaceutical (derived from experiments on animals or humans) or data for existing radiopharmaceuticals (from some literature source) must be entered into OLINDA/EXM to provide tables of dose estimates.
Publication of the technical basis for these factors is important to users. With the advent of electronic publishing, it is possible for the voluminous data tables to be distributed through peer-reviewed journals, with the technical basis for the data being published in a few pages of journal text. We use this method here to document the basis for these new dose estimate tables and facilitate their distribution to users through an electronic format. Also, the tables can be updated at any time, should biokinetic models change, simply with documentation of the date that the change was made and of the differences in the input data.
MATERIALS AND METHODS
New-Generation Computational Phantoms
The methods implemented in OLINDA/EXM version 2.0 are identical to those implemented in versions 1.0 and 1.1, as were described previously (1). The fundamental improvement introduced in version 2.0 was the use of voxel-based, anatomically realistic phantoms, using the adult male and female phantoms developed by Segars (19) and modified to match the organ masses shown in ICRP publication 89 (20). These computational phantoms are based on a technology called nonuniform rational B-splines (NURBS); this method represents organ surfaces in a way that allows them to be easily deformed. As such, the adult male and female phantoms were further deformed to develop computational phantoms for children of various ages, as defined in ICRP publication 89, namely newborns, 1-y-olds, 5-y-olds, 10-y-olds, and 15-y-olds. Unlike the hermaphrodite-stylized models of the 1980s and 1990s (Fig. 1), these NURBS models are separately designed for male and female individuals of each age; thus, this phantom series contains 12 separate phantoms. Absorbed fractions and dose factors were developed for this phantom series (21) and are contained in OLINDA/EXM version 2.0. New, voxel-based realistic phantoms for the pregnant woman at 3, 6, and 9 mo of gestation, developed by the team at Rensselaer Polytechnic Institute (22), were included. The Cristy–Eckerman adult and pediatric phantom series (10) and the pregnant female phantom series of Stabin et al. (12) are also retained in the version 2 code, to facilitate comparisons between the new and old generations of computational phantoms.
(A) Cristy–Eckerman stylized computational phantoms. (B) NURBS realistic voxel-based phantoms.
Computational Phantoms for Animals
With the advent of micro-CT imaging devices for small animals, Segars and Tsui developed computational phantoms for a mouse and a rat from CT images (Fig. 2) (23). Keenan et al. made 2 additional mouse models and 4 additional rat models by scaling the Segars–Tsui models (24). Stabin et al. also developed computational phantoms for male and female beagle dogs (25). Absorbed fractions were developed for all the animal models and are implemented in OLINDA/EXM version 2.0 for the development of dose factors and dose estimates for activity distributions in animals reasonably represented by these models.
Mouse and rat computational phantoms.
Radiopharmaceutical Dose Estimates
Biokinetic models for nearly 100 radiopharmaceuticals were used with the OLINDA/EXM 2.0 adult and pediatric phantoms to develop dose estimate tables. Most of the data were taken from the models proposed by the ICRP task group on radiopharmaceutical dosimetry (26). Male and female tables were generated for 1-y-olds, 5-y-olds, 10-y-olds, 15-y-olds, and adults. Then, a sex-averaged table was developed using the sex-averaging rule described in ICRP publication 103 (Fig. 3; (27)).
Organ (A) and effective dose (B) sex-averaging method proposed in ICRP 103. T = tissue.
The ICRP 30 gastrointestinal tract model (4) was superseded by the human alimentary tract model described in ICRP publication 100 (Fig. 4; (28)).
ICRP 100 human alimentary tract model.
The notable changes are that upper large intestine and lower large intestine from the ICRP 30 model were replaced by right colon, left colon, and rectosigmoid colon. The ICRP 100 model has variable transit times in the various segments of the model for different ages and sexes and for 4 distinct substances (solids, caloric liquids, noncaloric liquids and total diet).
RESULTS
The dose estimate tables give male and female values for dose to approximately 25 target organs, as well as sex-averaged values for the 5 phantom ages considered (1-y-olds, 5-y-olds, 10-y-olds, 15-y-olds, and adults). In a break with tradition in radiopharmaceutical dosimetry, individual organ doses are given in units of equivalent dose (e.g., mSv), as in the ICRP system, and not absorbed dose (e.g., mGy), because quality factors are applied that may be nonunity for α-emitters. OLINDA/EXM 2.0 uses a default radiation weighting factor of 5 for α-emissions; however, all radiation weighting factors in the OLINDA/EXM code are variable and may be adjusted by the user. Effective doses, by definition obtained by applying individual tissue weighting factors, are expressed in the same units as equivalent dose. Sample dose estimate tables are shown in Tables 1–3. Unlike the Cristy–Eckerman phantoms, no breast tissue was assigned in children 10 y old or younger. The bone model used in OLINDA/EXM 2.0 is the same as that used in OLINDA/EXM versions 1.0 and 1.1 (16). Several new organs have been explicitly defined in the RADAR phantoms: esophagus, eyes, salivary glands, and prostate in the male phantoms.
OLINDA/EXM 2.0 Recommended Radiation Dose Estimates for 18F-FDG: Male Data
OLINDA/EXM 2.0 Recommended Radiation Dose Estimates for 18F-FDG: Female Data
OLINDA/EXM 2.0 Recommended Radiation Dose Estimates for 18F-FDG: Sex-Averaged Data
DISCUSSION
Several dose estimate compendia for radiopharmaceuticals have been published. Stabin et al. published a small compendium of doses at Oak Ridge Associated Universities (29). The MIRD committee published 20 dose estimate reports over some 35 y; this is a small number, and many (e.g., selenomethionine) are for radiopharmaceuticals no longer in use. The broadest and most up-to-date compendium has been published by the ICRP task group on radiopharmaceuticals. This was published in several ICRP reports; the data from the various reports were combined in ICRP publication 128 (26). These dose estimates, however, are based on the outdated Cristy–Eckerman phantoms of 1987, which use the organ masses given in the 1973 ICRP report on reference man (7). The dose estimates given in this article are based on the new-generation, voxel-based realistic phantoms, which use the newest ICRP reference organ masses (20). The ICRP group tried to use surrogate organs to estimate doses to some missing organs (e.g., salivary glands), but nonetheless, the use of the outdated stylized phantoms that apply crude geometric shapes to approximate organ geometries is not a defensible idea, given the current technology available. The ICRP does have defined voxel-based adult male and female phantoms (30) but has developed no pediatric or pregnant female voxel-based phantoms. Even these 2 adult models are based on hand-drawn volumes of interest to define organs, with manual sculpting to match the ICRP 89 organ masses.
OLINDA/EXM is a U.S. Food and Drug Administration–approved software tool; the current version (2.0) is currently being distributed. This software tool facilitates the calculation of dose estimates given appropriate input data and generates tables of dose factors. Its widespread use is evidenced in the vast majority of publications involving radiopharmaceutical dosimetry. No software tool has been developed by either the MIRD or ICRP group to calculate dose estimates, as the RADAR group has done.
An important issue in the calculation of dose factors for α-particles is the assignment of a quality factor. Traditionally, a factor of 20 has been applied in radiation protection (4). Some radiobiologic evidence indicates that this value may be as low as 5 (31) or even 1 (32). The authors of MIRD pamphlet 20 (3) state that “[relative biological effectiveness] values range from 1 to 8 for cell killing in vivo, depending on the reference radiation, α-particle energy, and biologic endpoint.” Similar arguments apply to the use of Auger emitters (for which literature values indicate a range of potential relative biological effectiveness values (33)). The OLINDA/EXM code gives suggested radiation weighting factors of 5 for α-emissions and 1 for photons and electrons but allows users to vary the values as desired. Because radiation weighting factors have been applied, dose values for individual organs are not given in units mGy but in units of mSv (as in ICRP publications in which radiation weighting factors are used).
This article gives male and female dose estimates for only 5 ages: 1-y-olds, 5-y-olds, 10-y-olds, 15-y-olds, and adults. As in ICRP publication 103, dose estimates are not provided for newborns, although NURBS models for newborns are available. We decided to develop a separate publication on dose estimates for women in different stages of pregnancy.
All dose estimates cited in this article represent population averages for a particular age and sex and should never be applied to an individual patient. Doses are given for several radiopharmaceuticals that may be used in a therapeutic setting. The individual organ doses give population averages that may be useful for planning purposes. However, the ICRP notes in its publication 128 that “[t]he data are not intended for therapeutic applications of radionuclides. More detailed and patient-specific dosimetry and dose planning should be applied for therapeutic application of radionuclides.” It should also be noted that, although provided, the quantity “effective dose” has no meaning in therapeutic applications and should never be cited.
CONCLUSION
We present here dose estimates for about 100 radiopharmaceuticals based on the new-generation, NURBS-based voxel phantoms, using the ICRP 89–suggested organ masses. This represents a generational change in phantom development, as implemented in the OLINDA/EXM version 2.0 software. The use of an electronic publishing approach permits the publication of this kind of voluminous information in mainstream journals, with the technical basis being presented in printed form in the journal and the many necessarily large data tables provided electronically. Importantly, this compendium is essentially a living document since the dose estimates can be easily updated as biokinetic models change or new radiopharmaeuticals are developed, without the need for paper publication.
DISCLOSURE
No potential conflict of interest relevant to this article was reported.
Footnotes
Published online Sep. 8, 2017.
- © 2018 by the Society of Nuclear Medicine and Molecular Imaging.
REFERENCES
- Received for publication May 18, 2017.
- Accepted for publication July 18, 2017.











