Abstract
During the last decade, a systematic effort to develop a pharmacological treatment for Alzheimer disease (AD) has resulted in drugs being registered for the first time in the US and Europe for this specific indication. The 3 agents registered are cholinesterase inhibitors (ChEIs).
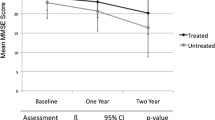
The major therapeutic effect of ChEIs in patients with AD is the maintenance of cognitive function, as compared with placebo, during a 6-month to 1-year period of treatment. Additional drug effects that may occur are the slowing of cognitive deterioration and improvement of behaviour and daily living activities. Comparison of clinical effects of 6 ChEIs demonstrates a rather similar magnitude of improvement in cognitive outcome measures. For some drugs, this level may represent an upper limit, while for others it may be possible to increase the benefit further. In order to maximise and prolong positive drug effects it is important to start treatment early and adjust the dosage during treatment. Recent studies that used this administration strategy have shown that in many patients, the stabilisation effect produced by ChEIs can be prolonged for as long as 36 months.
This long-lasting effect suggests mechanisms of action other than symptomatic ones. In this article, the effects of ChEIs on β-amyloid metabolism are postulated to explain the stabilising (i.e. disease-modifying) effects of the drugs. Evidence for such a mechanism is available at the experimental but not yet at the clinical level.




Similar content being viewed by others
References
Giacobini E. Cholinesterase inhibitors: from the calabar bean to Alzheimer therapy. In: Giacobini E. editor. Cholinesterase and cholinesterase inhibitors: from molecular biology to therapy. London: Martin Dunitz, 2000: 181–226
Asthana S, Raffaele KC, Gerig NH, et al. Neuroendocrine responses to intravenous infusion of physostigmine in patients with Alzheimer disease. Alzheimer Dis Assoc Disord 1999; 13: 102–8
Davis KL, Mohs RC, Marin D, et al. Cholinergic markers in elderly patients with early signs of Alzheimer disease. JAMA 1999; 281: 1401–6
Doody RS, Pratt RD, Perdomo CA. Clinical benefits of done-pezil: results from a long-term phase III extension trial. Neurology 1999; 52Suppl. 2: A174
Mori F, Lai CC, Fusi F, et al. Cholinesterase inhibitors increase secretion of APPs in rat brain cortex. Neuroreport 1995; 6: 633–6
Racchi M, Schmidt B, Koenig G, et al. Treatment with metrifonate promotes soluble amyloid precursor protein release from SH-SY5Y neuroblastoma cells. Alzheimer Dis Assoc Disord 1999; 13: 679–88
Pakaski M, Rakonczay Z, Kasa P. Reversible or irreversible cholinesterase inhibitors cause changes in neuronal amyloid percursor processing and protein kinase C level in vitro. Neurochem Int. In press
Cummings JL. Changes in neuropsychiatric symptoms as outcome measures in clinical trials with cholinergic therapies for Alzheimer disease. Alzheimer Dis Ass Disord 1997; 11(4 Suppl.): 1S–9S
Raskind MA, Cyrus PA, Ruzicka BB, et al. The effects of Metrifonate on the cognitive, behavioral, and functional performance of Alzheimer’s disease patients. J Clin Psychiatry 1999; 60: 318–25
Rogers S, Friedhoff L. Long-term efficacy and safety of done-pezil in the treatment of Alzheimer’s disease. Eur Neuropsychopharmacol 1998; 8: 67–75
Winblad B, Engedal K, Soininen H, et al. Donepezil enhances global function, cognition and activities of daily living compared with placebo one year [abstract 30]. 12th Annual Congress of the European College of Neuropsychopharmacology; 1999 Sep 21–25; London
Mohs R, Doody R, Morris J, et al. Donepezil preserves functional status in Alzheimer’s disease patients [abstract]. Eur Neuropsychopharmacol 1999; 9Suppl. 5: S328
Jelic V, Amberla K, Almkvist O, et al. Long-term tacrine treatment slows the increase of theta power in the EEG of mild Alzheimer patients compared to untreated controls [abstract]. 5th International Geneva/Springfield Symposium on Advances in Alzheimer Therapy; 1998 Apr 15–18; Geneva, 147
Farlow MR, Cyrus PA, Gulanski B. Metrifonate improves the cognitive deficits of Alzheimer’s disease patients in a dose-related manner [abstract A15]. Annual Meeting of the American Geriatrics Society; 1998 May 6–10; Seattle
Anand R, Hartman R, Messina J, et al. Long-term treatment with rivastigmine continue to provide benefits for up to one year [abstract]. 5th International Geneva/Springfield Symposium on Advances in Alzheimer Therapy; 1998 Apr 15–18; Geneva, 18
Rainer M, Mucke HAM. Long-term cognitive benefit from galanthamine in Alzheimer’s disease. Int J Ger Psychiatry 1999; 1: 197–201
Giacobini E. Cholinesterase inhibitors for Alzheimer’s disease therapy, from tacrine to future applications. Neurochem Int 1998 32: 413–9
Giacobini E, Cholinesterase inhibitors do more than inhibit cholinesterase. In: Becker R, Giacobini E, editors. Alzheimer disease: from molecular biology to therapy. Boston: Birkhäuser, 1996, 187–204
Inestrosa N, Alvarez A, Perez CA, et al. Acetylcholinesterase accelerates assembly of amyloid-beta-peptides into Alzheimer’s fibrils: possible role of the peripheral site of the enzyme. Neuron 1996; 16: 881–91
Suh Y-H, Chong YH, Kim S-H, et al. Molecular physiology, biochemistry and pharmacology of Alzheimer’s amyloid precursor protein (APP). Ann N Y Acad Sci 1996; 786: 169–83
Small DH, Sberna G, Li QX, et al. The beta-amyloid protein influence acethylcholinesterase expression, assembly and glycosylation [abstract no. 880S]. 6th International Conference on Alzheimer’s Disease and Related Disorders; 1998 M 18-23; Amsterdam, 209
Hilfers MA, Moore SA. Amyloid-beta peptide induces cyclo-oxygenase-2 expression and activity in brain-derived cells [abstract no. 450.1]. Soc Neuroscience 1999; 25: 1105
Nitsch RM, Slack BE, Wurtman RJ, et al. Release of Alzheimer amyloid precursor derivatives stimulated by activation of muscarinic acetylcholine receptors. Science 1992; 258: 304–7
Becker E, Giacobini E. Mechanisms of cholinesterase inhibition in senile dementia of the Alzheimer type. Drug Dev Res 1988; 12: 163–95
Giacobini E, Desarno P, Clark B, et al. The cholinergic receptor system of the human brain: neurochemical and pharmacological aspects in aging and Alzheimer. In: Nordberg A, Fuxe K, Holmstedt B, editors. Progress in brain research. Amsterdam: Elsevier, 1989: 335–43
von Der Kammer H, Mayhaus M, Albrecht C. et al Muscarinic acetylcholine receptors activate expression of the Erg gene family of transcription factors. J Biol Chem 1998; 273: 10–7
Bareggi SR, Giacobini E. Acetylcholinesterase activity in ventricular and cisternal CSF of dogs. JNeurosci Res 1978; 3: 335–9
Scarsella G, Toschi G, Bareggi SR, et al. Molecular forms of cholinesterase in cerebrospinal fluid, blood plasma and brain tissue of the beagle dog. J Neurosci Res 1979; 4: 19–24
Elble R, Giacobini E, Scarsella GF. Cholinesterase in cerebrospinal fluid. Neurology 1987; 44: 403–7
Mattio T, McIlhany M, Giacobini E, et al. The effects of physostigmine on acethylcholinesterase activity of CSF, plasma and brain. Neuropharmacology 1986; 25: 1167–77
Nordberg A, Hellstrom-Lindahl E, Almqkvist O, et al. Acetylcholinesterase activity in CSF of Alzheimer’s patients after treatment with tacrine. Alzheimer’s Report 1999; 2(6): 347–52
Unni L, Vicari S, Moriearty P, et al. The recovery of cerebrospinal fluid acetylcholinesterase activity in Alzheimer’s disease patients after treatment with metrifonate. Methods Find Exp Clin Pharmacol 2000; 22: 57–61
Hinz VC, Kolb J, Schmidt B. Effects of subchronic administration of metrifonate on cholinergic neurotransmission in rats. Neurochem Res 1998; 23: 933–4
Cummings MD, Cyrus PA, Bieber F, et al. Metrifonate treatment of the cognitive deficits in Alzheimer’s disease. Neurology 1999; 50: 1214–21
Cummings Jl, Cyrus PA, Gulanski B. Metrifonate efficacy in the treatment of psychiatric and behavioral disturbances of Alzheimer’s disease patients [abstract A15]. Annual Meeting of the American Geriatrics Society; 1998 May 6–10; Seattle
Haroutunian V, Wallace WC, Greig N, et al. Induction, secretion and pharmacological regulation of beta-APP in animal model systems [abstract]. 6th International Stockholm/Springfield Symposium in Advances in Alzheimer Therapy; 2000 Apr 5–8, 81
Lahiri DK, Farlow MR, Ge Y et al. Regulation of beta-APP processing with cholinestrase inhibitors in neuronal cell lines [abstract]. 6th International Stockholm/Springfield Symposium in Advances in Alzheimer Therapy; 2000 Apr 5–8, 81
Wallace WC, Bragin V, Robakis NK, et al. Increased biosynthesis of Alzheimer amyloid precursor protein in the cerebral cortex rats with lesions of the nucleus basalis Meynert. Brain Res Mol Brain Res 1991; 10: 173–8
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Giacobini, E. Do Cholinesterase Inhibitors Have Disease-Modifying Effects in Alzheimer’s Disease?. Mol Diag Ther 15, 85–91 (2001). https://doi.org/10.2165/00023210-200115020-00001
Published:
Issue Date:
DOI: https://doi.org/10.2165/00023210-200115020-00001