Abstract
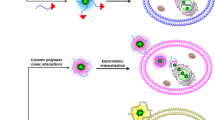
Systemic delivery of adenoviral vectors is a major goal in cancer gene therapy, but is currently prohibited by rapid hepatic uptake of virus following intravenous injection with levels of viable virus in the murine plasma typically falling to less than 0.1% after 30 min. We have used a surface-masking technique based on multivalent copolymers of poly(N-(2-hydroxypropyl)methacrylamide) to ablate all pathways of receptor-mediated infection, combined with dose modulation to achieve partial saturation of nonspecific uptake pathways. Polymer coating gave at least 100-fold decreased hepatic transgene expression at all doses and even high doses of coated virus (pc-virus) showed no weight loss or stimulation of serum transaminases. Low doses of virus and pc-virus (109 viral particles (vp)/mouse) were mainly captured by the liver (assessed by quantitative PCR), although higher doses led to greater fractional persistence in the plasma (measured after 30 min). Coated virus at a dose of 6 × 1011 vp/mouse showed nearly 50% plasma circulation, representing a 3.5-fold greater area under the concentration–time curve (0–30 min) compared to unmodified virus. Such an increase in the bioavailability of adenovirus, coupled with substantial decreases in toxicity and unwanted transgene expression is an important step towards producing systemically available tumour-targeted viruses.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Bergelson JM et al. Isolation of a common receptor for Coxsackie B viruses and adenoviruses 2 and 5. Science 1997; 275: 1320–1323.
Wickham TJ, Mathias P, Cheresh DA, Nemerow GR . Integrins alpha v beta 3 and alpha v beta 5 promote adenovirus internalization but not virus attachment. Cell 1993; 73: 309–319.
Dechecchi MC, Tamanini A, Bonizzato A, Cabrini G . Heparan sulfate glycosaminoglycans are involved in adenovirus type 5 and 2-host cell interactions. Virology 2000; 268: 382–390.
Dechecchi MC et al. Heparan sulfate glycosaminoglycans are receptors sufficient to mediate the initial binding of adenovirus types 2 and 5. J Virol 2001; 75: 8772–8780.
Bergelson JM et al. The murine CAR homolog is a receptor for coxsackie B viruses and adenoviruses. J Virol 1998; 72: 415–419.
Alemany R, Curiel DT . CAR-binding ablation does not change biodistribution and toxicity of adenoviral vectors. Gene Therapy 2001; 8: 1347–1353.
Leissner P et al. Influence of adenoviral fiber mutations on viral encapsidation, infectivity and in vivo tropism. Gene Therapy 2001; 8: 49–57.
Einfeld DA et al. Reducing the native tropism of adenovirus vectors requires removal of both CAR and integrin interactions. J Virol 2001; 75: 11284–11291.
Smith T et al. In vivo hepatic adenoviral gene delivery occurs independently of the coxsackievirus-adenovirus receptor. Mol Ther 2002; 5: 770–779.
Smith TA et al. Adenovirus serotype 5 fiber shaft influences in vivo gene transfer in mice. Hum Gene Ther 2003; 14: 777–787.
Tao N et al. Sequestration of adenoviral vector by Kupffer cells leads to a nonlinear dose response of transduction in liver. Mol Ther 2001; 3: 28–35.
Fisher KD et al. Polymer-coated adenovirus permits efficient retargeting and evades neutralising antibodies. Gene Therapy 2001; 8: 341–348.
Ye X, Jerebtsova M, Ray PE . Liver bypass significantly increases the transduction efficiency of recombinant adenoviral vectors in the lung, intestine, and kidney. Hum Gene Ther 2000; 11: 621–627.
Kasono K et al. Selective gene delivery to head and neck cancer cells via an integrin targeted adenoviral vector. Clin Cancer Res 1999; 5: 2571–2579.
Nicklin SA et al. Selective targeting of gene transfer to vascular endothelial cells by use of peptides isolated by phage display. Circulation 2000; 102: 231–237.
Wickham TJ . Targeting adenovirus. Gene Therapy 2000; 7: 110–114.
Romanczuk H et al. Modification of an adenoviral vector with biologically selected peptides: a novel strategy for gene delivery to cells of choice. Hum Gene Ther 1999; 10: 2615–2626.
Drapkin PT et al. Targeting the urokinase plasminogen activator receptor enhances gene transfer to human airway epithelia. J Clin Invest 2000; 105: 589–596.
Cichon G et al. Titer determination of Ad5 in blood: a cautionary note. Gene Therapy 2003; 10: 1012–1017.
Reid T et al. Intra-arterial administration of a replication-selective adenovirus (dl1520) in patients with colorectal carcinoma metastatic to the liver: a phase I trial. Gene Therapy 2001; 8: 1618–1626.
Nemunaitis J et al. Intravenous infusion of a replication-selective adenovirus (ONYX-015) in cancer patients: safety, feasibility and biological activity. Gene Therapy 2001; 8: 746–759.
Murakami P, McCaman MT . Quantitation of adenovirus DNA and virus particles with the PicoGreen fluorescent dye. Anal Biochem 1999; 274: 283–288.
Rihova B et al. Biocompatibility of N-(2-hydroxypropyl) methacrylamide copolymers containing adriamycin. Immunogenicity, and effect on haematopoietic stem cells in bone marrow in vivo and mouse splenocytes and human peripheral blood lymphocytes in vitro. Biomaterials 1989; 10: 335–342.
Acknowledgements
We are grateful to the Medical Research Council (SJH, VM), Cancer Research UK (KDF) and the National Translational Cancer Network (LWS) for financial support.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Green, N., Herbert, C., Hale, S. et al. Extended plasma circulation time and decreased toxicity of polymer-coated adenovirus. Gene Ther 11, 1256–1263 (2004). https://doi.org/10.1038/sj.gt.3302295
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302295
Keywords
This article is cited by
-
Oncolytic Viruses and Cancer Immunotherapy
Current Oncology Reports (2023)
-
Challenges and progress toward tumor-targeted therapy by systemic delivery of polymer-complexed oncolytic adenoviruses
Cancer Gene Therapy (2022)
-
Pharmacokinetics and tolerance of repeated oral administration of 5-fluorocytosine in healthy dogs
BMC Veterinary Research (2021)
-
A phase 1 trial of the safety, tolerability and biological effects of intravenous Enadenotucirev, a novel oncolytic virus, in combination with chemoradiotherapy in locally advanced rectal cancer (CEDAR)
Radiation Oncology (2020)
-
Vaccine vectors based on Adenovirus 19a/64 exhibit broad cellular tropism and potently restimulate HCMV-specific T cell responses ex vivo
Scientific Reports (2018)