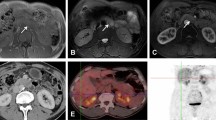
Key Points
-
Pancreatic cancer is frequently diagnosed after the appearance of symptoms, which is too late for curable treatment
-
No blood test currently exists for chronic pancreatitis and the diagnosis can be difficult to make, even with current imaging technologies
-
Treating pancreatic disease at an early stage of the pathogenesis could lead to better prognosis
-
Currently used imaging techniques have various limitations, including difficulty in discriminating between benign and malignant conditions
-
Molecular imaging can augment conventional imaging modalities for the diagnosis of incipient pancreatic diseases
Abstract
Pancreatic diseases, chronic pancreatitis, pancreatic cancer and diabetes mellitus, taken together, occur in >10% of the world population. Pancreatic diseases, as with other diseases, benefit from early intervention and appropriate diagnosis. Although imaging technologies have given clinicians an unprecedented toolbox to aid in clinical decision-making, advances in these technologies and development of molecular-based diagnostic tools could enable physicians to identify diseases at an even earlier stage and, thereby, improve patient outcomes. In this Review, we discuss and identify gaps in the use of imaging techniques for the early detection and appropriate treatment stratification of various pancreatic diseases, including diabetes mellitus, acute and chronic pancreatitis and pancreatic cancer. Imaging techniques discussed are MRI, CT, PET and ultrasonography. Additionally, the identification of new molecular targets for imaging and the development of contrast agents that are able to give molecular information in noninvasive radionuclear imaging and ultrasonography are emerging areas of innovation that could lead to increased diagnostic accuracy and improved patient outcomes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Berger, H. G. et al. (eds) The Pancreas (Blackwell Publishing Ltd., 2008)
Coté, G. A., Smith, J., Sherman, S. & Kelly, K. Technologies for imaging the normal and diseased pancreas. Gastroenterology 144, 1262–1271.e1 (2013).
Li, H., Hu, Z., Chen, J. & Guo, X. Comparison of ERCP, EUS, and ERCP combined with EUS in diagnosing pancreatic neoplasms: a systematic review and meta-analysis. Tumour Biol. 35, 8867–8874 (2014).
Bushberg, J. T. The Essential Physics of Medical Imaging. (Lippincott Williams & Wilkins, 2002).
Lindner, J. R. Microbubbles in medical imaging: current applications and future directions. Nat. Rev. Drug Discov. 3, 527–532 (2004).
Rickes, S. et al. Contrast-enhanced sonography in pancreatic diseases. Eur. J. Radiol. 64, 183–188 (2007).
Barr, R. G. Off-label use of ultrasound contrast agents for abdominal imaging in the United States. J. Ultrasound Med. 32, 7–12 (2013).
Worhunsky, D. J. et al. Pancreatic neuroendocrine tumours: hypoenhancement on arterial phase computed tomography predicts biological aggressiveness. HPB (Oxford) 16, 304–311 (2014).
D'Onofrio, M., Zamboni, G., Faccioli, N., Capelli, P. & Pozzi Mucelli, R. Ultrasonography of the pancreas. 4. Contrast-enhanced imaging. Abdom. Imaging 32, 171–181 (2007).
Robles-Medranda, C. Confocal endomicroscopy: is it time to move on? World J. Gastrointest. Endosc. 8, 1–3 (2016).
Yachida, S. et al. Distant metastasis occurs late during the genetic evolution of pancreatic cancer. Nature 467, 1114–1117 (2010).
Diagnosis of Diabetes and Prediabetes | National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK). niddk.nih.gov Available at: https://www.niddk.nih.gov/health-information/diabetes/diagnosis-diabetes-prediabetes (Accessed: 7 December 2015).
Chen, R., Pan, S., Brentnall, T. A. & Aebersold, R. Proteomic profiling of pancreatic cancer for biomarker discovery. Mol. Cell. Proteom. 4, 523–533 (2005).
Farr, R. J., Joglekar, M. V., Taylor, C. J. & Hardikar, A. A. Circulating non-coding RNAs as biomarkers of β cell death in diabetes. Pediatr. Endocrinol. Rev. 11, 14–20 (2013).
Melo, S. A. et al. Glypican-1 identifies cancer exosomes and detects early pancreatic cancer. Nature 523, 177–182 (2015).
Rhim, A. D. et al. Detection of circulating pancreas epithelial cells in patients with pancreatic cystic lesions. Gastroenterology 146, 647–651 (2014).
Chari, S. T. et al. Early detection of sporadic pancreatic cancer: summative review. Pancreas 44, 693–712 (2015).
Klibanov, A. L. et al. Targeting of ultrasound contrast material. An in vitro feasibility study. Acta Radiol. Suppl. 412, 113–120 (1997).
Klibanov, A. Targeted delivery of gas-filled microspheres, contrast agents for ultrasound imaging. Adv. Drug Deliv. Rev. 37, 139–157 (1999).
Foygel, K. et al. Detection of pancreatic ductal adenocarcinoma in mice by ultrasound imaging of thymocyte differentiation antigen 1. Gastroenterology 145, 885–894.e3 (2013).
Pogue, B. W., Leblond, F., Krishnaswamy, V. & Paulsen, K. D. Radiologic and near-infrared/optical spectroscopic imaging: where is the synergy? AJR Am. J. Roentgenol. 195, 321–332 (2010).
Rudin, M. & Weissleder, R. Molecular imaging in drug discovery and development. Nat. Rev. Drug Discov. 2, 123–131 (2003).
Whitcomb, D. C. Clinical practice. Acute pancreatitis. N. Engl. J. Med. 354, 2142–2150 (2006).
Lankisch, P. G., Apte, M. & Banks, P. A. Acute pancreatitis. Lancet 386, 85–96 (2015).
Lévy, P., Domínguez-Muñoz, E., Imrie, C., Löhr, M. & Maisonneuve, P. Epidemiology of chronic pancreatitis: burden of the disease and consequences. United European Gastroenterol. J. 2, 345–354 (2014).
Olsen, T. S. The incidence and clinical relevance of chronic inflammation in the pancreas in autopsy material. Acta Pathol. Microbiol. Scand. A 86A, 361–365 (1978).
Shimizu, M., Hayashi, T., Saitoh, Y. & Itoh, H. Interstitial fibrosis in the pancreas. Am. J. Clin. Pathol. 91, 531–534 (1989).
Stamm, B. H. Incidence and diagnostic significance of minor pathologic changes in the adult pancreas at autopsy: a systematic study of 112 autopsies in patients without known pancreatic disease. Hum. Pathol. 15, 677–683 (1984).
Whitcomb, D. C. & Pogue-Geile, K. Pancreatitis as a risk for pancreatic cancer. Gastroenterol. Clin. North Am. 31, 663–678 (2002).
Raimondi, S., Lowenfels, A. B., Morselli-Labate, A. M., Maisonneuve, P. & Pezzilli, R. Pancreatic cancer in chronic pancreatitis; aetiology, incidence, and early detection. Best Pract. Res. Clin. Gastroenterol. 24, 349–358 (2010).
Neff, C. C., Simeone, J. F., Wittenberg, J., Mueller, P. R. & Ferrucci, J. T. Inflammatory pancreatic masses. Problems in differentiating focal pancreatitis from carcinoma. Radiology 150, 35–38 (1984).
Thoeni, R. F. Imaging of acute pancreatitis. Radiol. Clin. North Am. 53, 1189–1208 (2015).
Bollen, T. L. Acute pancreatitis: international classification and nomenclature. Clin. Radiol 71, 121–133 (2016).
Singh, V. K. et al. An assessment of the severity of interstitial pancreatitis. Clin. Gastroenterol. Hepatol. 9, 1098–1103 (2011).
van Santvoort, H. C. et al. A conservative and minimally invasive approach to necrotizing pancreatitis improves outcome. Gastroenterology 141, 1254–1263 (2011).
Ljutic, D., Piplovic-Vukovic, T., Raos, V. & Andrews, P. Acute renal failure as a complication of acute pancreatitis. Ren. Fail. 18, 629–633 (1996).
Tenner, S., Baillie, J., DeWitt, J., Vege, S. S. & American College of Gastroenterology American College of Gastroenterology guideline: management of acute pancreatitis. Am. J. Gastroenterol. 108, 1400–1415, 1416 (2013).
Golea, A., Badea, R., Socaciu, M., Diaconu, B. & Iacob, D. Quantitative analysis of tissue perfusion using contrast-enhanced transabdominal ultrasound (CEUS) in the evaluation of the severity of acute pancreatitis. Med. Ultrason. 12, 198–204 (2010).
Siracusano, S. et al. The current role of contrast-enhanced ultrasound (CEUS) imaging in the evaluation of renal pathology. World J. Urol. 29, 633–638 (2011).
Hasebroock, K. M. & Serkova, N. J. Toxicity of MRI and CT contrast agents. Expert Opin. Drug Metab. Toxicol. 5, 403–416 (2009).
Ripollés, T., Martínez, M. J., López, E., Castelló, I. & Delgado, F. Contrast-enhanced ultrasound in the staging of acute pancreatitis. Eur. Radiol. 20, 2518–2523 (2010).
Steer, M. L., Waxman, I. & Freedman, S. Chronic pancreatitis. N. Engl. J. Med. 332, 1482–1490 (1995).
Gumaste, V. V., Roditis, N., Mehta, D. & Dave, P. B. Serum lipase levels in nonpancreatic abdominal pain versus acute pancreatitis. Am. J. Gastroenterol. 88, 2051–2055 (1993).
Choueiri, N. E., Balci, N. C., Alkaade, S. & Burton, F. R. Advanced Imaging of chronic pancreatitis. Curr. Gastroenterol. Rep. 12, 114–120 (2010).
Siddiqi, A. J. & Miller, F. Chronic pancreatitis: ultrasound, computed tomography, and magnetic resonance imaging features. Semin. Ultrasound CT MRI 28, 384–394 (2007).
Bali, M. A. et al. Quantification of pancreatic exocrine function with secretin-enhanced magnetic resonance cholangiopancreatography: normal values and short-term effects of pancreatic duct drainage procedures in chronic pancreatitis. Initial results. Eur. Radiol. 15, 2110–2121 (2005).
Calculli, L. et al. Exocrine pancreatic function assessed by secretin cholangio-Wirsung magnetic resonance imaging. Hepatobiliary Pancreat. Dis. Int. 7, 192–195 (2008).
Bor, R., Madácsy, L., Fábián, A., Szepes, A. & Szepes, Z. Endoscopic retrograde pancreatography: when should we do it? World J. Gastrointest. Endosc. 7, 1023–1031 (2015).
Zuccaro, G. & Sivak, M. V. Endoscopic ultrasonography in the diagnosis of chronic pancreatitis. Endoscopy 24 (Suppl. 1), 347–349 (1992).
Catalano, M. F. et al. EUS-based criteria for the diagnosis of chronic pancreatitis: the Rosemont classification. Gastrointest. Endosc. 69, 1251–1261 (2009).
Gardner, T. B. & Levy, M. J. EUS diagnosis of chronic pancreatitis. Gastrointest. Endosc. 71, 1280–1289 (2010).
Azemoto, N. et al. Utility of contrast-enhanced transabdominal ultrasonography to diagnose early chronic pancreatitis. Biomed. Res. Int. 2015, 393124 (2015).
Iglesias-Garcia, J., Lariño-Noia, J., Abdulkader, I., Forteza, J. & Dominguez-Muñoz, J. E. Quantitative endoscopic ultrasound elastography: an accurate method for the differentiation of solid pancreatic masses. Gastroenterology 139, 1172–1180 (2010).
Iglesias-Garcia, J., Dominguez-Muñoz, J. E., Castiñeira-Alvariño, M., Luaces Regueira, M. & Lariño-Noia, J. Quantitative elastography associated with endoscopic ultrasound for the diagnosis of chronic pancreatitis. Endoscopy 45, 781–788 (2013).
Kongkam, P. et al. Combination of EUS-FNA and elastography (strain ratio) to exclude malignant solid pancreatic lesions: a prospective single-blinded study. J. Gastroenterol. Hepatol. 30, 1683–1689 (2015).
de Jong, K. et al. High prevalence of pancreatic cysts detected by screening magnetic resonance imaging examinations. Clin. Gastroenterol. Hepatol. 8, 806–811 (2010).
Laffan, T. A. et al. Prevalence of unsuspected pancreatic cysts on MDCT. AJR Am. J. Roentgenol. 191, 802–807 (2008).
Brugge, W. R., Lauwers, G. Y., Sahani, D., Fernández-del, C. C. & Warshaw, A. L. Cystic neoplasms of the pancreas. N. Engl. J. Med. 351, 1218–1226 (2004).
de Jong, K., Bruno, M. J. & Fockens, P. Epidemiology, diagnosis, and management of cystic lesions of the pancreas. Gastroenterol. Res. Pract. 2012, 147465 (2012).
Grützmann, R., Niedergethmann, M., Pilarsky, C., Klöppel, G. & Saeger, H. D. Intraductal papillary mucinous tumors of the pancreas: biology, diagnosis, and treatment. Oncologist 15, 1294–1309 (2010).
Adimoolam, V. et al. Endoscopic ultrasound identifies synchronous pancreas cystic lesions not seen on initial cross-sectional imaging. Pancreas 40, 1070–1072 (2011).
Anand, N., Sampath, K. & Wu, B. U. Cyst features and risk of malignancy in intraductal papillary mucinous neoplasms of the pancreas: a meta-analysis. Clin. Gastroenterol. Hepatol. 11, 913–921; quiz e59–e60 (2013).
D'Onofrio, M. et al. Pancreatic multicenter ultrasound study (PAMUS). Eur. J. Radiol. 81, 630–638 (2012).
Beyer-Enke, S. A., Hocke, M., Ignee, A., Braden, B. & Dietrich, C. F. Contrast enhanced transabdominal ultrasound in the characterisation of pancreatic lesions with cystic appearance. JOP 11, 427–433 (2010).
D'Onofrio, M. et al. Comparison of contrast-enhanced sonography and MRI in displaying anatomic features of cystic pancreatic masses. AJR Am. J. Roentgenol. 189, 1435–1442 (2007).
Hocke, M., Cui, X.-W., Domagk, D., Ignee, A. & Dietrich, C. F. Pancreatic cystic lesions: the value of contrast-enhanced endoscopic ultrasound to influence the clinical pathway. Endosc. Ultrasound 3, 123–130 (2014).
Giovannini, M. et al. Results of a phase I-II study on intraductal confocal microscopy (IDCM) in patients with common bile duct (CBD) stenosis. Surg. Endosc. 25, 2247–2253 (2011).
Sultana, A. et al. What is the best way to identify malignant transformation within pancreatic IPMN: a systematic review and meta-analyses. Clin. Transl. Gastroenterol. 6, e130 (2015).
Kim, S.-L. et al. The effect of PPAR-γ agonist on 18F-FDG uptake in tumor and macrophages and tumor cells. Nucl. Med. Biol. 36, 427–433 (2009).
Cornish, T. C. & Hruban, R. H. Pancreatic intraepithelial neoplasia. Surg. Pathol. Clin. 4, 523–535 (2011).
Jemal, A., Siegel, R., Xu, J. & Ward, E. Cancer statistics, 2010. CA Cancer J. Clin. 60, 277–300 (2010).
Allen, P. J. & Brennan, M. F. A. Selective approach to resection of cystic lesions of the pancreas: results from 539 consecutive patients. Ann. Surg. 245, 825–826 (2007).
Pancreatic Cancer Action Network. Pancreatic Cancer Facts 2016 [online], (2016).
Rahib, L. et al. Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 74, 2913–2921 (2014).
National Cancer Institute SEER stat fact sheets: pancreatic cancer. http://seer.cancer.gov/statfacts/html/pancreas.html (2015).
Al-Hawary, M. M., Francis, I. R. & Anderson, M. A. Pancreatic solid and cystic neoplasms: diagnostic evaluation and intervention. Radiol. Clin. North Am. 53, 1037–1048 (2015).
Valls, C. et al. Dual-phase helical CT of pancreatic adenocarcinoma: assessment of resectability before surgery. AJR Am. J. Roentgenol. 178, 821–826 (2002).
Lee, E. S. & Lee, J. M. Imaging diagnosis of pancreatic cancer: a state-of-the-art review. World J. Gastroenterol. 20, 7864–7877 (2014).
Bronstein, Y. L. et al. Detection of small pancreatic tumors with multiphasic helical CT. AJR Am. J. Roentgenol. 182, 619–623 (2004).
Tsunoda, T. et al. Staging and treatment for patients with pancreatic cancer. How small is an early pancreatic cancer? J. Hepatobiliary Pancreat. Surg. 5, 128–132 (1998).
Sahani, D. V., Shah, Z. K., Catalano, O. A., Boland, G. W. & Brugge, W. R. Radiology of pancreatic adenocarcinoma: current status of imaging. J. Gastroenterol. Hepatol. 23, 23–33 (2008).
Harewood, G. C. & Wiersema, M. J. Endosonography-guided fine needle aspiration biopsy in the evaluation of pancreatic masses. Am. J. Gastroenterol. 97, 1386–1391 (2002).
Templeton, A. W. & Brentnall, T. A. Screening and surgical outcomes of familial pancreatic cancer. Surg. Clin. North Am. 93, 629–645 (2013).
Helmstaedter, L. & Riemann, J. F. Pancreatic cancer —EUS and early diagnosis. Langenbecks Arch. Surg. 393, 923–927 (2008).
Pietryga, J. A. & Morgan, D. E. Imaging preoperatively for pancreatic adenocarcinoma. J. Gastrointest. Oncol. 6, 343–357 (2015).
Hicklin, D. J. & Ellis, L. M. Role of the vascular endothelial growth factor pathway in tumor growth and angiogenesis. J. Clin. Oncol. 23, 1011–1027 (2005).
Longo, R., Cacciamani, F., Naso, G. & Gasparini, G. Pancreatic cancer: from molecular signature to target therapy. Crit. Rev. Oncol. Hematol. 68, 197–211 (2008).
Korc, M. Pathways for aberrant angiogenesis in pancreatic cancer. Mol. Cancer 2, 8 (2003).
Tonra, J. R. et al. Synergistic antitumor effects of combined epidermal growth factor receptor and vascular endothelial growth factor receptor-2 targeted therapy. Clin. Cancer Res. 12, 2197–2207 (2006).
Spano, J.-P. et al. Efficacy of gemcitabine plus axitinib compared with gemcitabine alone in patients with advanced pancreatic cancer: an open-label randomised phase II study. Lancet 371, 2101–2108 (2008).
Itakura, J. et al. Enhanced expression of vascular endothelial growth factor in human pancreatic cancer correlates with local disease progression. Clin. Cancer Res. 3, 1309–1316 (1997).
Büchler, P. et al. Target therapy using a small molecule inhibitor against angiogenic receptors in pancreatic cancer. Neoplasia 9, 119–127 (2007).
Higgins, K. J., Abdelrahim, M., Liu, S., Yoon, K. & Safe, S. Regulation of vascular endothelial growth factor receptor-2 expression in pancreatic cancer cells by Sp proteins. Biochem. Biophys. Res. Commun. 345, 292–301 (2006).
Luo, J. et al. Pancreatic cancer cell-derived vascular endothelial growth factor is biologically active in vitro and enhances tumorigenicity in vivo. Int. J. Cancer 92, 361–369 (2001).
Shi, Q. et al. Constitutive Sp1 activity is essential for differential constitutive expression of vascular endothelial growth factor in human pancreatic adenocarcinoma. Cancer Res. 61, 4143–4154 (2001).
Korpanty, G., Carbon, J. G., Grayburn, P. A., Fleming, J. B. & Brekken, R. A. Monitoring response to anticancer therapy by targeting microbubbles to tumor vasculature. Clin. Cancer Res. 13, 323–330 (2007).
Deshpande, N., Ren, Y., Foygel, K., Rosenberg, J. & Willmann, J. K. Tumor angiogenic marker expression levels during tumor growth: longitudinal assessment with molecularly targeted microbubbles and US imaging. Radiology 258, 804–811 (2011).
Pysz, M. A. et al. Vascular endothelial growth factor receptor type 2-targeted contrast-enhanced US of pancreatic cancer neovasculature in a genetically engineered mouse model: potential for earlier detection. Radiology 274, 790–799 (2015).
Quail, D. F. & Joyce, J. A. Microenvironmental regulation of tumor progression and metastasis. Nat. Med. 19, 1423–1437 (2013).
Drifka, C. R. et al. Periductal stromal collagen topology of pancreatic ductal adenocarcinoma differs from that of normal and chronic pancreatitis. Mod. Pathol. 28, 1470–1480 (2015).
Ouban, A., Muraca, P., Yeatman, T. & Coppola, D. Expression and distribution of insulin-like growth factor-1 receptor in human carcinomas. Hum. Pathol. 34, 803–808 (2003).
Zhou, H. et al. IGF1 receptor targeted theranostic nanoparticles for targeted and image-guided therapy of pancreatic cancer. ACS Nano 9, 7976–7991 (2015).
Thomas, S. et al. Development of secreted protein and acidic and rich in cysteine (SPARC) targeted nanoparticles for the prognostic molecular imaging of metastatic prostate cancer. J. Nanomed. Nanotechnol. 2, http://dx.doi.org/10.4172/2157-7439.1000112 (2011).
Neuzillet, C. et al. Stromal expression of SPARC in pancreatic adenocarcinoma. Cancer Metastasis Rev. 32, 585–602 (2013).
Kelly, K. A. et al. Targeted nanoparticles for imaging incipient pancreatic ductal adenocarcinoma. PLoS Med. 5, e85 (2008).
Shin, S. J. et al. Unexpected gain of function for the scaffolding protein plectin due to mislocalization in pancreatic cancer. Proc. Natl Acad. Sci. USA 110, 19414–19419 (2013).
Bausch, D. et al. Plectin-1 as a novel biomarker for pancreatic cancer. Clin. Cancer Res. 17, 302–309 (2011).
US National Library of Medicine. ClinicalTrials.gov, https://clinicaltrials.gov/ct2/show/NCT01962909 (2013).
Kannagi, R., Izawa, M., Koike, T., Miyazaki, K. & Kimura, N. Carbohydrate-mediated cell adhesion in cancer metastasis and angiogenesis. Cancer Sci. 95, 377–384 (2004).
Dimastromatteo, J., Houghton, J. L., Lewis, J. S. & Kelly, K. A. Challenges of pancreatic cancer. Cancer J. 21, 188–193 (2015).
Viola-Villegas, N. T. et al. Applying PET to broaden the diagnostic utility of the clinically validated CA19.9 serum biomarker for oncology. J. Nucl. Med. 54, 1876–1882 (2013).
Houghton, J. L. et al. Site-specifically labeled CA19.9-targeted immunoconjugates for the PET, NIRF, and multimodal PET/NIRF imaging of pancreatic cancer. Proc. Natl Acad. Sci. USA 112, 15850–15855 (2015).
Bünger, S., Laubert, T., Roblick, U. J. & Habermann, J. K. Serum biomarkers for improved diagnostic of pancreatic cancer: a current overview. J. Cancer Res. Clin. Oncol. 137, 375–389 (2011).
Boonstra, M. C. et al. Preclinical evaluation of a novel CEA-targeting near-infrared fluorescent tracer delineating colorectal and pancreatic tumors. Int. J. Cancer 137, 1910–1920 (2015).
Jiang, T. et al. Tumor imaging by means of proteolytic activation of cell-penetrating peptides. Proc. Natl Acad. Sci. USA 101, 17867–17872 (2004).
Nguyen, Q. T. et al. Surgery with molecular fluorescence imaging using activatable cell-penetrating peptides decreases residual cancer and improves survival. Proc. Natl Acad. Sci. USA 107, 4317–4322 (2010).
Metildi, C. A. et al. Ratiometric activatable cell-penetrating peptides label pancreatic cancer, enabling fluorescence-guided surgery, which reduces metastases and recurrence in orthotopic mouse models. Ann. Surg. Oncol. 22, 2082–2087 (2014).
Pociot, F. & Lernmark, Å. Genetic risk factors for type 1 diabetes. Lancet 387, 2331–2339 (2016).
Ribaric, S. The rationale for insulin therapy in Alzheimer's disease. Molecules 21, E689 (2016).
de la Monte, S. M. & Wands, J. R. Alzheimer's disease is type 3 diabetes-evidence reviewed. J. Diabetes Sci. Technol. 2, 1101–1113 (2008).
Diabetes Atlas DiabetesAtlas.org http://www.diabetesatlas.org/ (2016).
Kim, D. L., Kim, S. D., Kim, S. K., Park, S. & Song, K. H. Is an oral glucose tolerance test still valid for diagnosing diabetes mellitus? Diabetes Metab. J. 40, 118–128 (2016).
Rahier, J., Guiot, Y., Goebbels, R. M., Sempoux, C. & Henquin, J. C. Pancreatic β-cell mass in European subjects with type 2 diabetes. Diabetes Obes. Metab. 10, 32–42 (2008).
Perry, T. & Greig, N. H. The glucagon-like peptides: a double-edged therapeutic sword? Trends Pharmacol. Sci. 24, 377–383 (2003).
Reiner, T. et al. Accurate measurement of pancreatic islet β-cell mass using a second-generation fluorescent exendin-4 analog. Proc. Natl Acad. Sci. USA 108, 12815–12820 (2011).
Wild, D. et al. [Lys40(Ahx-DTPA-111In)NH2]exendin-4, a very promising ligand for glucagon-like peptide-1 (GLP-1) receptor targeting. J. Nucl. Med. 47, 2025–2033 (2006).
Wicki, A. et al. [Lys40(Ahx-DTPA-111In)NH2]-exendin-4 is a highly efficient radiotherapeutic for glucagon-like peptide-1 receptor-targeted therapy for insulinoma. Clin. Cancer Res. 13, 3696–3705 (2007).
Wu, Z. et al. In vivo imaging of transplanted islets with 64Cu-DO3A-VS-Cys40-Exendin-4 by targeting GLP-1 receptor. Bioconjug. Chem. 22, 1587–1594 (2011).
Connolly, B. M. et al. Ex vivo imaging of pancreatic β cells using a radiolabeled GLP-1 receptor agonist. Mol. Imaging Biol. 14, 79–87 (2012).
Brand, C. et al. In vivo imaging of GLP-1R with a targeted bimodal PET/fluorescence imaging agent. Bioconjug. Chem. 25, 1323–1330 (2014).
Behnam Azad, B. et al. Synthesis and evaluation of optical and PET GLP-1 peptide analogues for GLP-1R imaging. Mol. Imaging 14, 1–16 (2015).
Henquin, J.-C., Nenquin, M., Stiernet, P. & Ahren, B. In vivo and in vitro glucose-induced biphasic insulin secretion in the mouse: pattern and role of cytoplasmic Ca2+ and amplification signals in β-cells. Diabetes 55, 441–451 (2006).
Antkowiak, P. F. et al. Noninvasive assessment of pancreatic β-cell function in vivo with manganese-enhanced magnetic resonance imaging. Am. J. Physiol. Endocrinol. Metab. 296, E573–E578 (2009).
Antkowiak, P. F., Stevens, B. K., Nunemaker, C. S., McDuffie, M. & Epstein, F. H. Manganese-enhanced magnetic resonance imaging detects declining pancreatic β-cell mass in a cyclophosphamide-accelerated mouse model of type 1 diabetes. Diabetes 62, 44–48 (2013).
Ablamunits, V., Quintana, F., Reshef, T., Elias, D. & Cohen, I. R. Acceleration of autoimmune diabetes by cyclophosphamide is associated with an enhanced IFN-γ secretion pathway. J. Autoimmun. 13, 383–392 (1999).
Lubag, A. J. M., De Leon-Rodriguez, L. M., Burgess, S. C. & Sherry, A. D. Noninvasive MRI of β-cell function using a Zn2+-responsive contrast agent. Proc. Natl Acad. Sci. USA 108, 18400–18405 (2011).
Acknowledgements
The authors thank S. L. Hasbrouck, Curry School of Education, University of Virginia, for her kind help reading and editing the manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed equally to the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Dimastromatteo, J., Brentnall, T. & Kelly, K. Imaging in pancreatic disease. Nat Rev Gastroenterol Hepatol 14, 97–109 (2017). https://doi.org/10.1038/nrgastro.2016.144
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrgastro.2016.144
This article is cited by
-
PancrESS – a meta-analysis resource for understanding cell-type specific expression in the human pancreas
BMC Genomics (2024)
-
Aktuelle CT-Entwicklungen für die Diagnostik von Erkrankungen des Pankreas
Die Radiologie (2023)
-
Clinical significance of pancreatic calcifications: a 15-year single-center observational study
European Journal of Medical Research (2022)
-
ImmunoPET/NIRF/Cerenkov multimodality imaging of ICAM-1 in pancreatic ductal adenocarcinoma
European Journal of Nuclear Medicine and Molecular Imaging (2021)
-
Utility of ultrasound in acute pancreatitis
Abdominal Radiology (2020)