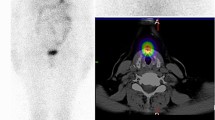
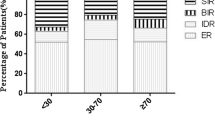
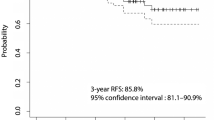
Abstract
Radioiodine is considered an effective and low-risk therapy modality of advanced differentiated thyroid cancer. For patients without lymph-node or distant metastases and low stages of the primary tumor, debate is ongoing about the necessity of thyroid remnant tissue ablation in an adjuvant setting. On the basis of evidence from retrospective studies, and until results of ongoing controlled prospective randomized trials become available, 131I ablation of remnant thyroid tissue in patients with primary tumors >1 cm is advisable. For thyroid remnant ablation, individual dosimetry is not obligatory. By contrast, the effectiveness of 131I therapy of locally advanced and/or metastatic disease can be improved by individual dosimetry. For practical reasons, an approach delivering the maximal possible radiation dose to the tumor without exceeding a critical blood dose of approximately 2 Gy seems advantageous. The availability of recombinant human TSH (rhTSH) has improved the quality of life of patients and reduces the radiation exposure of healthy nonthyroid tissue compared with TSH stimulation through levothyroxine withdrawal. In patients with distant metastases, rhTSH stimulation is possible only in off-label use, from which especially elderly and frail patients may benefit, as they most severely suffer from hypothyroidism caused by thyroid hormone withdrawal.
Key Points
-
Radioiodine is an effective and low-risk therapy of advanced differentiated thyroid cancer
-
For low-risk patients, the indication for radioiodine (131I) therapy is subject of debate
-
For thyroid remnant ablation, individual dosimetry is not obligatory
-
The effectiveness of 131I therapy of advanced differentiated thyroid cancer can be improved by individual dosimetry
-
Stimulation with recombinant human TSH (rhTSH) for 131I ablation maintains quality of life and lowers the radiation exposure of healthy nonthyroid tissue compared with TSH stimulation through levothyroxine withdrawal
-
For 131I therapy of advanced disease, rhTSH stimulation in off-label use might benefit elderly and frail patients
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Change history
12 January 2012
In the version of this article initially published online the legend of Figure 1 should read "The 5th edition of the UICC TNM system was used to classify patients. We performed a visual, illustrative comparison using data on expected survival on the basis of group composition regarding age and sex for each stage group. These data were obtained from the German Federal Bureau of Statistics (Bundesamt für Statistik). No further statistical analysis was performed, as the visual illustration of the good prognosis of patients with differentiated thyroid cancer was the only goal of this figure." The error has been corrected for the HTML and PDF versions of the article.
References
Cooper, D. S. et al. Revised American Thyroid Association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid 19, 1167–1214 (2009).
Pacini, F. et al. European consensus for the management of patients with differentiated thyroid carcinoma of the follicular epithelium. Eur. J. Endocrinol. 154, 787–803 (2006).
Pacini, F., Castagna, M. G., Brilli, L. & Pentheroudakis, G. Thyroid cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 21 (Suppl. 5), v214–v219 (2010).
Luster, M. et al. Guidelines for radioiodine therapy of differentiated thyroid cancer. Eur. J. Nucl. Med. Mol. Imaging 35, 1941–1959 (2008).
Mazzaferri, E. L. & Jhiang, S. M. Long-term impact of initial surgical and medical therapy on papillary and follicular thyroid cancer. Am. J. Med. 97, 418–428 (1994).
Eskandari, S. et al. Thyroid Na+/I− symporter. Mechanism, stoichiometry, and specificity. J. Biol. Chem. 272, 27230–27238 (1997).
Van Nostrand, D. Sialoadenitis secondary to 131I therapy for well-differentiated thyroid cancer. Oral Dis. 17, 154–161 (2011).
Iyer, N. G., Morris, L. G., Tuttle, R. M., Shaha, A. R. & Ganly, I. Rising incidence of second cancers in patients with low-risk (T1N0) thyroid cancer who receive radioactive iodine therapy. Cancer doi:10.1002/cncr.26070.
Schlumberger, M. J. Papillary and follicular thyroid carcinoma. N. Engl. J. Med. 338, 297–306 (1998).
Verburg, F. A., de Keizer, B., Lips, C. J., Zelissen, P. M. & de Klerk, J. M. Prognostic significance of successful ablation with radioiodine of differentiated thyroid cancer patients. Eur. J. Endocrinol. 152, 33–37 (2005).
Mazzaferri, E. L. & Kloos, R. T. Clinical review 128: Current approaches to primary therapy for papillary and follicular thyroid cancer. J. Clin. Endocrinol. Metab. 86, 1447–1463 (2001).
Tubiana, M. et al. Long-term results and prognostic factors in patients with differentiated thyroid carcinoma. Cancer 55, 794–804 (1985).
Simpson, W. J., Panzarella, T., Carruthers, J. S., Gospodarowicz, M. K. & Sutcliffe, S. B. Papillary and follicular thyroid cancer: impact of treatment in 1578 patients. Int. J. Radiat. Oncol. Biol. Phys. 14, 1063–1075 (1988).
Utiger, R. D. Follow-up of patients with thyroid carcinoma. N. Engl. J. Med. 337, 928–930 (1997).
Verburg, F. A., Dietlein, M., Lassmann, M., Luster, M. & Reiners, C. Why radioiodine remnant ablation is right for most patients with differentiated thyroid carcinoma. Eur. J. Nucl. Med. Mol. Imaging 36, 343–346 (2009).
Shattuck, T. M., Westra, W. H., Ladenson, P. W. & Arnold, A. Independent clonal origins of distinct tumor foci in multifocal papillary thyroid carcinoma. N. Engl. J. Med. 352, 2406–2412 (2005).
Sherman, S. I., Tielens, E. T., Sostre, S., Wharam, M. D. Jr & Ladenson, P. W. Clinical utility of posttreatment radioiodine scans in the management of patients with thyroid carcinoma. J. Clin. Endocrinol. Metab. 78, 629–634 (1994).
Tenenbaum, F., Corone, C., Schlumberger, M. & Parmentier, C. Thyroglobulin measurement and postablative iodine-131 total body scan after total thyroidectomy for differentiated thyroid carcinoma in patients with no evidence of disease. Eur. J. Cancer 32A, 1262 (1996).
Travagli, J. P. et al. Combination of radioiodine (131I) and probe-guided surgery for persistent or recurrent thyroid carcinoma. J. Clin. Endocrinol. Metab. 83, 2675–2680 (1998).
Taylor, T. et al. Outcome after treatment of high-risk papillary and non-Hurthle-cell follicular thyroid carcinoma. Ann. Intern. Med. 129, 622–627 (1998).
Hay, I. D. et al. Papillary thyroid carcinoma managed at the Mayo Clinic during six decades (1940–1999): temporal trends in initial therapy and long-term outcome in 2,444 consecutively treated patients. World J. Surg. 26, 879–885 (2002).
Sawka, A. M. et al. Clinical review 170: a systematic review and metaanalysis of the effectiveness of radioactive iodine remnant ablation for well-differentiated thyroid cancer. J. Clin. Endocrinol. Metab. 89, 3668–3676 (2004).
Pacini, F. et al. Post-surgical use of radioiodine (131I) in patients with papillary and follicular thyroid cancer and the issue of remnant ablation: a consensus report. Eur. J. Endocrinol. 153, 651–659 (2005).
Rosario, P. W. et al. Is adjuvant therapy useful in patients with papillary carcinoma smaller than 2 cm? Thyroid 17, 1225–1228 (2007).
Sawka, A. M. et al. An updated systematic review and commentary examining the effectiveness of radioactive iodine remnant ablation in well-differentiated thyroid cancer. Endocrinol. Metab. Clin. North Am. 37, 457–480 (2008).
DeGroot, L. J., Kaplan, E. L., Shukla, M. S., Salti, G. & Straus, F. H. Morbidity and mortality in follicular thyroid cancer. J. Clin. Endocrinol. Metab. 80, 2946–2953 (1995).
Mazzaferri, E. L. Thyroid remnant 131I ablation for papillary and follicular thyroid carcinoma. Thyroid 7, 265–271 (1997).
Samaan, N. A. et al. The results of various modalities of treatment of well differentiated thyroid carcinomas: a retrospective review of 1,599 patients. J. Clin. Endocrinol. Metab. 75, 714–720 (1992).
Robbins, R. J. et al. Real-time prognosis for metastatic thyroid carcinoma based on 2-[18F]fluoro-2-deoxy-D-glucose-positron emission tomography scanning. J. Clin. Endocrinol. Metab. 91, 498–505 (2006).
Demidchik, Y. E. et al. Comprehensive clinical assessment of 740 cases of surgically treated thyroid cancer in children of Belarus. Ann. Surg. 243, 525–532 (2006).
Reiners, C., Demidchik, Y. E., Drozd, V. M. & Biko, J. Thyroid cancer in infants and adolescents after Chernobyl. Minerva Endocrinol. 33, 381–395 (2008).
Dottorini, M. E., Lomuscio, G., Mazzucchelli, L., Vignati, A. & Colombo, L. Assessment of female fertility and carcinogenesis after iodine-131 therapy for differentiated thyroid carcinoma. J. Nucl. Med. 36, 21–27 (1995).
Hay, I. D. et al. Long-term outcome in 215 children and adolescents with papillary thyroid cancer treated during 1940 through 2008. World J. Surg. 34, 1192–1202 (2010).
Biko, J. et al. Favourable course of disease after incomplete remission on 131I therapy in children with pulmonary metastases of papillary thyroid carcinoma: 10 years follow-up. Eur. J. Nucl. Med. Mol. Imaging 38, 651–655 (2011).
Verburg, F. A. et al. Dosimetry-guided high-activity 131I therapy in patients with advanced differentiated thyroid carcinoma: initial experience. Eur. J. Nucl. Med. Mol. Imaging 37, 896–903 (2010).
Lassmann, M., Reiners, C. & Luster, M. Dosimetry and thyroid cancer: the individual dosage of radioiodine. Endocr. Relat Cancer 17, R161–R172 (2010).
Verburg, F. A., Mäder, U., Luster, M. & Reiners, C. Primary tumor diameter as a risk factor for advanced disease features of differentiated thyroid carcinoma. Clin. Endocrinol. (Oxf.) 71, 291–297 (2008).
Verburg, F. A., Mäder, U., Luster, M. & Reiners, C. Histology does not influence prognosis in differentiated thyroid carcinoma when accounting for age, tumour diameter, invasive growth and metastases. Eur. J. Endocrinol. 160, 619–624 (2009).
Seidlin, S. M., Marinelli, L. D. & Oshry, E. Radioactive iodine therapy: Effect on functioning metastases of adenocarcinoma of the thyroid. JAMA 132, 838–847 (1946).
Tuttle, R. M. et al. Thyroid carcinoma. J. Natl Compr. Canc. Netw. 8, 1228–1274 (2010).
Hackshaw, A. & Mallick, U. Low versus high I-131 dose for remnant ablation in differentiated thyroid cancer. Clin. Med. Res. 7, 1–3 (2009).
Mallick, U., Harmer, C. & Hackshaw, A. The HiLo trial: a multicentre randomised trial of high- versus low-dose radioiodine, with or without recombinant human thyroid stimulating hormone, for remnant ablation after surgery for differentiated thyroid cancer. Clin. Oncol. (R. Coll. Radiol.) 20, 325–326 (2008).
Doi, S. A. & Woodhouse, N. J. Ablation of the thyroid remnant and 131I dose in differentiated thyroid cancer. Clin. Endocrinol. (Oxf.) 52, 765–773 (2000).
Hackshaw, A., Harmer, C., Mallick, U., Haq, M. & Franklyn, J. A. 131I activity for remnant ablation in patients with differentiated thyroid cancer: A systematic review. J. Clin. Endocrinol. Metab. 92, 28–38 (2007).
Doi, S. A., Woodhouse, N. J., Thalib, L. & Onitilo, A. Ablation of the thyroid remnant and I-131 dose in differentiated thyroid cancer: a meta-analysis revisited. Clin. Med. Res. 5, 87–90 (2007).
Schlumberger, M. et al. Radioactive iodine treatment and external radiotherapy for lung and bone metastases from thyroid carcinoma. J. Nucl. Med. 37, 598–605 (1996).
Menzel, C. et al. “High-dose” radioiodine therapy in advanced differentiated thyroid carcinoma. J. Nucl. Med. 37, 1496–1503 (1996).
Van Nostrand, D. in Thyroid Cancer: A Comprehensive Guide to Clinical Management 2nd edn (eds Wartofsky, L. & Van Nostrand, D.) 411–425 (Humana Press, Totowa, 2006).
Samuel, A. M., Rajashekharrao, B. & Shah, D. H. Pulmonary metastases in children and adolescents with well-differentiated thyroid cancer. J. Nucl. Med. 39, 1531–1536 (1998).
Chiesa, C. et al. Individualized dosimetry in the management of metastatic differentiated thyroid cancer. Q. J. Nucl. Med. Mol. Imaging 53, 546–561 (2009).
Hänscheid, H. et al. Iodine biokinetics and dosimetry in radioiodine therapy of thyroid cancer: procedures and results of a prospective international controlled study of ablation after rhTSH or hormone withdrawal. J. Nucl. Med. 47, 648–654 (2006).
Benua, R. S., Cicale, N. R., Sonenberg, M. & Rawson, R. W. The relation of radioiodine dosimetry to results and complications in the treatment of metastatic thyroid cancer. AJR 1962, 171–182 (1962).
Maxon, H. R. et al. Relation between effective radiation dose and outcome of radioiodine therapy for thyroid cancer. N. Engl. J. Med. 309, 937–941 (1983).
Maxon, H. R. III et al. Radioiodine-131 therapy for well-differentiated thyroid cancer—a quantitative radiation dosimetric approach: outcome and validation in 85 patients. J. Nucl. Med. 33, 1132–1136 (1992).
Dorn, R. et al. Dosimetry-guided radioactive iodine treatment in patients with metastatic differentiated thyroid cancer: largest safe dose using a risk-adapted approach. J. Nucl. Med. 44, 451–456 (2003).
Tuttle, R. M. et al. Empiric radioactive iodine dosing regimens frequently exceed maximum tolerated activity levels in elderly patients with thyroid cancer. J. Nucl. Med. 47, 1587–1591 (2006).
Kulkarni, K. et al. The relative frequency in which empiric dosages of radioiodine would potentially overtreat or undertreat patients who have metastatic well-differentiated thyroid cancer. Thyroid 16, 1019–1023 (2006).
Hänscheid, H., Lassmann, M., Luster, M., Kloos, R. & Reiners, C. Blood dosimetry from a single measurement of the whole body radioiodine retention in patients with differentiated thyroid carcinoma. Endocr. Relat. Cancer 16, 1283–1289 (2009).
Hänscheid, H. et al. Success of the postoperative 131I therapy in young Belarusian patients with differentiated thyroid cancer after Chernobyl depends on the radiation absorbed dose to the blood and the thyroglobulin level. Eur. J. Nucl. Med. Mol. Imaging 38, 1296–1302 (2011).
Verburg, F. A. et al. The absorbed dose to the blood is a better predictor of ablation success than the administered (131)I activity in thyroid cancer patients. Eur. J. Nucl. Med. Mol. Imaging 38, 673–680 (2011).
Van Nostrand, D. et al. Dosimetrically determined doses of radioiodine for the treatment of metastatic thyroid carcinoma. Thyroid 12, 121–134 (2002).
Flux, G. D. et al. A dose-effect correlation for radioiodine ablation in differentiated thyroid cancer. Eur. J. Nucl. Med. Mol. Imaging 37, 270–275 (2010).
Sgouros, G. et al. Patient-specific dosimetry for 131I thyroid cancer therapy using 124I PET and 3-dimensional-internal dosimetry (3D-ID) software. J. Nucl. Med. 45, 1366–1372 (2004).
Bolch, W. E., Eckerman, K. F., Sgouros, G. & Thomas, S. R. MIRD pamphlet No. 21: a generalized schema for radiopharmaceutical dosimetry—standardization of nomenclature. J. Nucl. Med. 50, 477–484 (2009).
Prideaux, A. R. et al. Three-dimensional radiobiologic dosimetry: application of radiobiologic modeling to patient-specific 3-dimensional imaging-based internal dosimetry. J. Nucl. Med. 48, 1008–1016 (2007).
Mazzaferri, E. L. & Massoll, N. Management of papillary and follicular (differentiated) thyroid cancer: new paradigms using recombinant human thyrotropin. Endocr. Relat. Cancer 9, 227–247 (2002).
Maxon, H. R. et al. Low iodine diet in I-131 ablation of thyroid remnants. Clin. Nucl. Med. 8, 123–126 (1983).
Sawka, A. M. et al. Basis for physician recommendations for adjuvant radioiodine therapy in early-stage thyroid carcinoma: principal findings of the Canadian–American thyroid cancer survey. Endocr. Pract. 14, 175–184 (2008).
Morsch, E. P., Vanacor, R., Furlanetto, T. W. & Schmid, H. Two weeks of a low-iodine diet are equivalent to 3 weeks for lowering urinary iodine and increasing thyroid radioactive iodine uptake. Thyroid 21, 61–67 (2011).
Weintraub, B. D. & Szkudlinski, M. W. Development and in vitro characterization of human recombinant thyrotropin. Thyroid 9, 447–450 (1999).
Haugen, B. R. et al. A comparison of recombinant human thyrotropin and thyroid hormone withdrawal for the detection of thyroid remnant or cancer. J. Clin. Endocrinol. Metab. 84, 3877–3885 (1999).
Duntas, L. H. & Biondi, B. Short-term hypothyroidism after levothyroxine-withdrawal in patients with differentiated thyroid cancer: clinical and quality of life consequences. Eur. J. Endocrinol. 156, 13–19 (2007).
Luster, M., Felbinger, R., Dietlein, M. & Reiners, C. Thyroid hormone withdrawal in patients with differentiated thyroid carcinoma: a one hundred thirty-patient pilot survey on consequences of hypothyroidism and a pharmacoeconomic comparison to recombinant thyrotropin administration. Thyroid 15, 1147–1155 (2005).
Dow, K. H., Ferrell, B. R. & Anello, C. Quality-of-life changes in patients with thyroid cancer after withdrawal of thyroid hormone therapy. Thyroid 7, 613–619 (1997).
Chow, S. M. et al. Health-related quality-of-life study in patients with carcinoma of the thyroid after thyroxine withdrawal for whole body scanning. Laryngoscope 116, 2060–2066 (2006).
Tagay, S. et al. Health-related quality of life, anxiety and depression in thyroid cancer patients under short-term hypothyroidism and TSH-suppressive levothyroxine treatment. Eur. J. Endocrinol. 153, 755–763 (2005).
Schroeder, P. R. et al. A comparison of short-term changes in health-related quality of life in thyroid carcinoma patients undergoing diagnostic evaluation with recombinant human thyrotropin compared with thyroid hormone withdrawal. J. Clin. Endocrinol. Metab. 91, 878–884 (2006).
Remy, H. et al. 131I effective half-life and dosimetry in thyroid cancer patients. J. Nucl. Med. 49, 1445–1450 (2008).
Vaiano, A. et al. Comparison between remnant and red-marrow absorbed dose in thyroid cancer patients submitted to 131I ablative therapy after rh-TSH stimulation versus hypothyroidism induced by L-thyroxine withdrawal. Nucl. Med. Commun. 28, 215–223 (2007).
Luster, M. et al. Comparison of radioiodine biokinetics following the administration of recombinant human thyroid stimulating hormone and after thyroid hormone withdrawal in thyroid carcinoma. Eur. J. Nucl. Med. Mol. Imaging 30, 1371–1377 (2003).
Löffler, M., Weckesser, M., Franzius, C., Kies, P. & Schober, O. Iodine excretion during stimulation with rhTSH in differentiated thyroid carcinoma. Nuklearmedizin 42, 240–243 (2003).
Rosário, P. W., Borges, M. A. & Purisch, S. Preparation with recombinant human thyroid-stimulating hormone for thyroid remnant ablation with 131I is associated with lowered radiotoxicity. J. Nucl. Med. 49, 1776–1782 (2008).
Frigo, A. et al. Chromosome translocation frequency after radioiodine thyroid remnant ablation: a comparison between recombinant human thyrotropin stimulation and prolonged levothyroxine withdrawal. J. Clin. Endocrinol. Metab. 94, 3472–3476 (2009).
Chen, A. Y., Jemal, A. & Ward, E. M. Increasing incidence of differentiated thyroid cancer in the United States, 1988–2005. Cancer 115, 3801–3807 (2009).
Tuttle, R. M., Leboeuf, R. & Shaha, A. R. Medical Management of thyroid cancer: a risk adapted approach. J. Surg. Oncol. 97, 712–716 (2008).
Gramza, A. & Schuff, K. G. Recombinant human thyroid stimulating hormone in 2008: focus on thyroid cancer management. Onco. Targets Ther. 1, 87–101 (2009).
Pacini, F. et al. Radioiodine ablation of thyroid remnants after preparation with recombinant human thyrotropin in differentiated thyroid carcinoma: results of an international, randomized, controlled study. J. Clin. Endocrinol. Metab. 91, 926–932 (2006).
Elisei, R. et al. Follow-up of low-risk differentiated thyroid cancer patients who underwent radioiodine ablation of postsurgical thyroid remnants after either recombinant human thyrotropin or thyroid hormone withdrawal. J. Clin. Endocrinol. Metab. 94, 4171–4179 (2009).
Luster, M. et al. Use of recombinant human thyrotropin before radioiodine therapy in patients with advanced differentiated thyroid carcinoma. J. Clin. Endocrinol. Metab. 85, 3640–3645 (2000).
de Keizer, B. et al. Tumour dosimetry and response in patients with metastatic differentiated thyroid cancer using recombinant human thyrotropin before radioiodine therapy. Eur. J. Nucl. Med. Mol. Imaging 30, 367–373 (2003).
Robbins, R. J., Driedger, A. & Magner, J. Recombinant human thyrotropin-assisted radioiodine therapy for patients with metastatic thyroid cancer who could not elevate endogenous thyrotropin or be withdrawn from thyroxine. Thyroid 16, 1121–1130 (2006).
Borget, I. et al. Length and cost of hospital stay of radioiodine ablation in thyroid cancer patients: comparison between preparation with thyroid hormone withdrawal and thyrogen. Eur. J. Nucl. Med. Mol. Imaging 35, 1457–1463 (2008).
Borget, I. et al. Sick leave for follow-up control in thyroid cancer patients: comparison between stimulation with Thyrogen and thyroid hormone withdrawal. Eur. J. Endocrinol. 156, 531–538 (2007).
Mernagh, P. et al. Cost-effectiveness of using recombinant human TSH prior to radioiodine ablation for thyroid cancer, compared with treating patients in a hypothyroid state: the German perspective. Eur. J. Endocrinol. 155, 405–414 (2006).
Zanotti-Fregonara, P. et al. Overview on the use of recombinant human thyrotropin in thyroid cancer of follicular cell origin. Minerva Endocrinol. 33, 53–65 (2008).
Barbaro, D. et al. Recombinant human TSH and ablation of post-surgical thyroid remnants in differentiated thyroid cancer: the effect of pre-treatment with furosemide and furosemide plus lithium. Eur. J. Nucl. Med. Mol. Imaging 37, 242–249 (2010).
Tala Jury, H. P. et al. Lack of association between urinary iodine excretion and successful thyroid ablation in thyroid cancer patients. J. Clin. Endocrinol. Metab. 95, 230–237 (2010).
Author information
Authors and Affiliations
Contributions
All authors researched the data for the article and provided a substantial contribution to discussions of the content. C. Reiners, H. Hänscheid, M. Luster and F. A. Verburg contributed equally to writing the article. All authors reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
C. Reiners, M. Luster, M. Lassmann and F. A. Verburg declare an association with the following company: Genzyme. C. Reiners and M. Lassmann have received research support from Genzyme. M. Luster and F. A. Verburg have received research support and speakers fees from Genzyme. H. Hänscheid declares no competing interests.
Rights and permissions
About this article
Cite this article
Reiners, C., Hänscheid, H., Luster, M. et al. Radioiodine for remnant ablation and therapy of metastatic disease. Nat Rev Endocrinol 7, 589–595 (2011). https://doi.org/10.1038/nrendo.2011.134
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2011.134
This article is cited by
-
Effects of BRAFV600E mutation on Na+/I- symporter expression in papillary thyroid carcinoma
Journal of Huazhong University of Science and Technology [Medical Sciences] (2016)
-
Development of a dry distillation technology for the production of 131I using medium flux reactor for radiopharmaceutical applications
Journal of Radioanalytical and Nuclear Chemistry (2015)
-
Differentiated thyroid cancer—personalized therapies to prevent overtreatment
Nature Reviews Endocrinology (2014)
-
Physiological sodium concentrations enhance the iodide affinity of the Na+/I− symporter
Nature Communications (2014)
-
The number of 131I therapy courses needed to achieve complete remission is an indicator of prognosis in patients with differentiated thyroid carcinoma
European Journal of Nuclear Medicine and Molecular Imaging (2014)