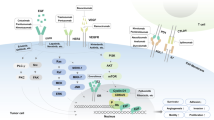
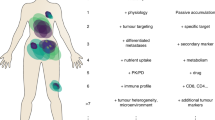
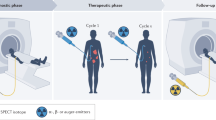
Key Points
-
Imaging technologies exploit the interaction of various forms of energy with tissues to non-invasively visualize the body.
-
Many imaging techniques were originally developed for human use, but have recently been scaled-down to allow the high-resolution imaging of mice. This is highly relevant, because as genomics provides us with better animal models of disease, imaging readouts can be used to evaluate novel therapeutics.
-
Imaging methods offer several advantages over other current practices in drug discovery, examples of which are discussed in this article:
-
The use of imaging endpoints instead of time-consuming dissection and histology can significantly decrease the workload involved in tissue analysis and thereby speed up the evaluation of drug candidates.
-
Imaging can provide biomarkers of a disease process and thus help to define stratified study groups.
-
As imaging methods are non-invasive, they allow for longitudinal studies in a single animal. This increases the statistical relevance of a study, allows for more clinically relevant study designs and decreases the number of animals required.
-
Imaging can also provide important information on the optimal timing and dosing of drugs.
-
Emerging molecular-imaging tools can provide much earlier surrogate markers of therapy success than is currently possible.
Abstract
Imaging sciences have grown exponentially during the past three decades, and many techniques, such as magnetic resonance imaging, nuclear tomographic imaging and X-ray computed tomography, have become indispensable in clinical use. Advances in imaging technologies and imaging probes for humans and for small animals are now extending the applications of imaging further into drug discovery and development, and have the potential to considerably accelerate the process. This review summarizes some of the recent developments in conventional and molecular imaging, and highlights their impact on drug discovery.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Weissleder, R. Scaling down imaging: molecular mapping of cancer in mice. Nature Rev. Cancer 2, 11–18 (2002). Review of in vivo mouse imaging technologies.
Weissleder, R. & Ntziachristos, V. Shedding light onto live molecular targets. Nature Med. 9, 123–128 (2003). This manuscript reviews emerging novel optical in vivo imaging technologies.
Rudin, M. et al. In vivo magnetic resonance methods in pharmaceutical research: current status and perspectives. NMR Biomed. 12, 69–97 (1999). Review of MR technologies in drug development.
Beckmann, N., Mueggler, T., Allegrini, P. R., Laurent, D. & Rudin, M. From anatomy to the target: contributions of magnetic resonance imaging to preclinical pharmaceutical research. Anat. Rec. 265, 85–100 (2001).
Jain, R. K., Munn, L. L. & Fukumura, D. Dissecting tumour pathophysiology using intravital microscopy. Nature Rev. Cancer 2, 266–276 (2002).
Phelps, M. E. Inaugural article: positron emission tomography provides molecular imaging of biological processes. Proc. Natl Acad. Sci. USA 97, 9226–9233 (2000). Review of PET imaging applications.
Fischman, A. J., Alpert, N. M. & Rubin, R. H. Pharmacokinetic imaging: a noninvasive method for determining drug distribution and action. Clin. Pharmacokinet. 41, 581–602 (2002). Review of the role of PET in the study of drug pharmacokinetics.
Reese, T., Bochelen, D., Sauter, A., Beckmann, N. & Rudin, M. Magnetic resonance angiography of the rat cerebrovascular system without the use of contrast agents. NMR Biomed. 12, 189–196 (1999).
Villringer, A. et al. Dynamic imaging with lanthanide chelates in normal brain: contrast due to magnetic susceptibility effects. Magn. Reson. Med. 6, 164–174 (1988).
Roussel, S. A., van Bruggen, N., King, M. D. & Gadian, D. G. Identification of collaterally perfused areas following focal cerebral ischemia in the rat by comparison of gradient echo and diffusion-weighted MRI. J. Cereb. Blood Flow Metab. 15, 578–586 (1995).
Moseley, M., Wendland, M. & Kucharczyk, J. Magnetic resonance imaging of diffusion and perfusion. Top. Magn. Reson. Imaging 3, 50–67 (1991).
Sauter, A. & Rudin, M. Calcium antagonists reduce the extent of infarction in rat middle cerebral artery occlusion model as determined by quantitative magnetic resonance imaging. Stroke 17, 1228–1234 (1986).
Rausch, M., Baumann, D., Neubacher, U. & Rudin, M. In vivo visualization of phagocytotic cells in rat brains after transient ischemia by USPIO. NMR Biomed. 15, 278–283 (2002).
Sauter, A. et al. Recovery of function in cytoprotected cerebral cortex in rat stroke model assessed by functional MRI. Magn. Reson. Med. 47, 759–765 (2002).
Cristofanilli, M., Charnsangavej, C. & Hortobagyi, G. N. Angiogenesis modulation in cancer research: novel clinical approaches. Nature Rev. Drug Discov. 1, 415–426 (2002).
Tatum, J. L. & Hoffman, J. M. Role of imaging in clinical trials of antiangiogenesis therapy in oncology. Acad. Radiol. 7, 798–799 (2000).
Bhujwalla, Z. M. et al. The physiological environment in cancer vascularization, invasion and metastasis. Novartis Found Symp. 240, 23–38; discussion 38-45, 152-153, (2001).
Lewin, M. et al. In vivo assessment of vascular endothelial growth factor-induced angiogenesis. Int. J. Cancer 83, 798–802 (1999).
Petrovsky, A., Weissleder, W., Shalinsky, D., Hu-Lowe, D. & Bogdanov, A. Non-invasive magnetic resonance imaging (MRI) of vascular parameters affected by VEGF–receptor tyrosine kinase inhibition in a human xenograft model. Proc. Am. Assoc. Cancer Res. 43, 1081 (2002).
Peterfy, C. G. Magnetic resonance imaging of rheumatoid arthritis: the evolution of clinical applications through clinical trials. Semin. Arthritis Rheum. 30, 375–396 (2001).
McConnell, M. V. et al. MRI of rabbit atherosclerosis in response to dietary cholesterol lowering. Arterioscler. Thromb. Vasc. Biol. 19, 1956–1959 (1999).
Chinnaiyan, A. M. et al. Combined effect of tumor necrosis factor-related apoptosis-inducing ligand and ionizing radiation in breast cancer therapy. Proc. Natl Acad. Sci. USA 97, 1754–1759 (2000).
Jennings, D. et al. Early response of prostate carcinoma xenografts to docetaxel chemotherapy monitored with diffusion MRI. Neoplasia 4, 255–262 (2002).
Evelhoch, J. L. et al. Applications of magnetic resonance in model systems: cancer therapeutics. Neoplasia 2, 152–165 (2000).
Cherry, S. R. Fundamentals of positron emission tomography and applications in preclinical drug development. J. Clin. Pharmacol. 41, 482–491 (2001).
Del Guerra, A. & Belcari, N. Advances in animal PET scanners. Q. J. Nucl. Med. 46, 35–47 (2002).
Green, L. A. et al. Noninvasive methods for quantitating blood time–activity curves from mouse PET images obtained with fluorine-18-fluorodeoxyglucose. J. Nucl. Med. 39, 729–734 (1998).
Chatziioannou, A., Tai, Y. C., Doshi, N. & Cherry, S. R. Detector development for microPET II: a 1 microl resolution PET scanner for small animal imaging. Phys. Med. Biol. 46, 2899–2910 (2001).
Brady, F. et al. Radiolabelled tracers and anticancer drugs for assessment of therapeutic efficacy using PET. Curr. Pharm. Des. 7, 1863–1892 (2001).
Halldin, C., Gulyas, B. & Farde, L. PET studies with carbon-11 radioligands in neuropsychopharmacological drug development. Curr. Pharm. Des. 7, 1907–1929 (2001). Review of PET in the development of drugs for mental health disorders.
Fischman, A. J. et al. Pharmacokinetics of 18F fleroxacin in patients with acute exacerbations of chronic bronchitis and complicated urinary tract infection studied by positron emission tomography. Antimicrob. Agents Chemother. 40, 659–664 (1996).
Fischman, A. J. et al. Positron emission tomographic analysis of central 5-hydroxytryptamine2 receptor occupancy in healthy volunteers treated with the novel antipsychotic agent, ziprasidone. J. Pharmacol. Exp. Ther. 279, 939–947 (1996).
Aboagye, E. O. et al. Cancer Research UK procedures in manufacture and toxicology of radiotracers intended for pre-phase I positron emission tomography studies in cancer patients. Br. J. Cancer 86, 1052–1056 (2002).
Gibson, R. E. et al. Non-invasive radiotracer imaging as a tool for drug development. Curr. Pharm. Des. 6, 973–989 (2000).
Fischman, A. J. et al. Pharmacokinetics of 18F-labeled fluconazole in healthy human subjects by positron emission tomography. Antimicrob. Agents Chemother. 37, 1270–1277 (1993).
Capdeville, R., Buchdunger, E., Zimmermann, J. & Matter, A. Glivec (STI571, imatinib), a rationally developed, targeted anticancer drug. Nature Rev. Drug Discov. 1, 493–502 (2002).
Coussens, L. M., Fingleton, B. & Matrisian, L. M. Matrix metalloproteinase inhibitors and cancer: trials and tribulations. Science 295, 2387–2392 (2002).
Warner, R. R. & O'Dorisio T. M. Radiolabeled peptides in diagnosis and tumor imaging: clinical overview. Semin. Nucl. Med. 32, 79–83 (2002).
Becker, A. et al. Receptor-targeted optical imaging of tumors with near-infrared fluorescent ligands. Nature Biotechnol. 19, 327–331 (2001).
Bugaj, J. E., Achilefu, S., Dorshow, R. B. & Rajagopalan, R. Novel fluorescent contrast agents for optical imaging of in vivo tumors based on a receptor-targeted dye–peptide conjugate platform. J. Biomed. Opt. 6, 122–133 (2001).
Tung, C. H., Lin, Y., Moon, W. K. & Weissleder, R. A receptor-targeted near-infrared fluorescence probe for in vivo tumor imaging. Chembiochem 3, 784–786 (2002).
Weissleder, R. et al. In vivo magnetic resonance imaging of transgene expression. Nature Med. 6, 351–355 (2000).
Hogemann, D., Ntziachristos, V., Josephson, L. & Weissleder, R. High throughput magnetic resonance imaging for evaluating targeted nanoparticle probes. Bioconjug. Chem. 13, 116–121 (2002).
Sipkins, D. A. et al. Detection of tumor angiogenesis in vivo by αVβ3-targeted magnetic resonance imaging. Nature Med. 4, 623–626 (1998).
Burns, H. D. et al. Positron emission tomography neuroreceptor imaging as a tool in drug discovery, research and development. Curr. Opin. Chem. Biol. 3, 388–394 (1999).
Hartvig, P., Bergstrom, M., Antoni, G. & Langstrom, B. Positron emission tomography and brain monoamine neurotransmission — entries for study of drug interactions. Curr. Pharm. Des. 8, 1417–1434 (2002).
Fowler, J. S. et al. Positron emission tomography studies of dopamine-enhancing drugs. J. Clin. Pharmacol. (Suppl.) 13–16 (1999).
Hietala, J. Ligand–receptor interactions as studied by PET: implications for drug development. Ann. Med. 31, 438–443 (1999).
Laakso, A. & Hietala, J. PET studies of brain monoamine transporters. Curr. Pharm. Des. 6, 1611–1623 (2000).
Passchier, J. & van Waarde, A. Visualisation of serotonin-1A (5-HT1A) receptors in the central nervous system. Eur. J. Nucl. Med. 28, 113–129 (2001).
Pilowsky, L. S. Probing targets for antipsychotic drug action with PET and SPET receptor imaging. Nucl. Med. Commun. 22, 829–833 (2001).
Volkow, N. D., Ding, Y. S., Fowler, J. S. & Gatley, S. J. Imaging brain cholinergic activity with positron emission tomography: its role in the evaluation of cholinergic treatments in Alzheimer's dementia. Biol. Psychiatry 49, 211–220 (2001).
Waarde, A. Measuring receptor occupancy with PET. Curr. Pharm. Des. 6, 1593–1610 (2000).
Martinez, D. et al. Differential occupancy of somatodendritic and postsynaptic 5HT(1A) receptors by pindolol: a dose-occupancy study with 11C WAY 100635 and positron emission tomography in humans. Neuropsychopharmacology 24, 209–229 (2001).
Louie, A. Y. et al. In vivo visualization of gene expression using magnetic resonance imaging. Nature Biotechnol. 18, 321–325 (2000).
Perez, J. M., Josephson, L., O'Loughlin, T., Hogemann, D. & Weissleder, R. Magnetic relaxation switches capable of sensing molecular interactions. Nature Biotechnol. 20, 816–820 (2002).
Bremer, C., Tung, C. H. & Weissleder, R. In vivo molecular target assessment of matrix metalloproteinase inhibition. Nature Med. 7, 743–748 (2001).
Mahmood, U., Tung, C. H., Tang, Y. & Weissleder, R. Feasibility of in vivo multichannel optical imaging of gene expression: experimental study in mice. Radiology 224, 446–451 (2002).
Lewin, M. et al. Tat peptide-derivatized magnetic nanoparticles allow in vivo tracking and recovery of progenitor cells. Nature Biotechnol. 18, 410–414 (2000).
Sharma, V., Luker, G. D. & Piwnica-Worms, D. Molecular imaging of gene expression and protein function in vivo with PET and SPECT. J. Magn. Reson. Imaging 16, 336–351 (2002).
Zhao, M., Kircher, M. F., Josephson, L. & Weissleder, R. Differential conjugation of tat peptide to superparamagnetic nanoparticles and its effect on cellular uptake. Bioconjug. Chem. 13, 840–844 (2002).
Tjuvajev, J. G. et al. Imaging the expression of transfected genes in vivo. Cancer Res. 55, 6126–6132 (1995).
Gambhir, S. S. et al. Imaging adenoviral-directed reporter gene expression in living animals with positron emission tomography. Proc. Natl Acad. Sci. USA 96, 2333–2338 (1999).
Li, X., Zhang, S., Zhao, P., Kovacs, Z. & Sherry, A. D. Synthesis and NMR studies of new DOTP-like lanthanide(III) complexes containing a hydrophobic substituent on one phosphonate side arm. Inorg. Chem. 40, 6572–6579 (2001).
Bornhop, D. J. et al. Fluorescent tissue site-selective lanthanide chelate, Tb-PCTMB for enhanced imaging of cancer. Anal. Chem. 71, 2607–2615 (1999).
Tromberg, B. J. et al. Non-invasive in vivo characterization of breast tumors using photon migration spectroscopy. Neoplasia 2, 26–40 (2000).
Chance, B. Near-infrared (NIR) optical spectroscopy characterizes breast tissue hormonal and age status. Acad. Radiol. 8, 209–210 (2001).
Hanlon, E. B. et al. Prospects for in vivo Raman spectroscopy. Phys. Med. Biol. 45, R1–R59 (2000).
Brown, E. B. et al. In vivo measurement of gene expression, angiogenesis and physiological function in tumors using multiphoton laser scanning microscopy. Nature Med. 7, 864–868 (2001).
Ntziachristos, V., Tung, C. H., Bremer, C. & Weissleder, R. Fluorescence molecular tomography resolves protease activity in vivo. Nature Med. 8, 757–760 (2002).
Marten, K. et al. Detection of dysplastic intestinal adenomas using enzyme-sensing molecular beacons in mice. Gastroenterology 122, 406–414 (2002).
Ito, S. et al. Detection of human gastric cancer in resected specimens using a novel infrared fluorescent anti-human carcinoembryonic antigen antibody with an infrared fluorescence endoscope in vitro. Endoscopy 33, 849–853 (2001).
Zonios, G., Bykowski, J. & Kollias, N. Skin melanin, hemoglobin, and light scattering properties can be quantitatively assessed in vivo using diffuse reflectance spectroscopy. J. Invest. Dermatol. 117, 1452–1457 (2001).
Kuroiwa, T., Kajimoto, Y. & Ohta, T. Development and clinical application of near-infrared surgical microscope: preliminary report. Minim. Invasive. Neurosurg. 44, 240–242 (2001).
Ntziachristos, V., Ripoll, J. & Weissleder, R. Would near-infrared fluorescence signals propagate through large human organs for clinical studies? Opt. Lett. 27, 333–335 (2002).
Contag, C. H. et al. Visualizing gene expression in living mammals using a bioluminescent reporter. Photochemistry & Photobiology 4, 523–531 (1997).
Hastings, J. W. Chemistries and colors of bioluminescent reactions: a review. Gene 173, 5–11 (1996).
Bhaumik, S. & Gambhir, S. S. Optical imaging of Renilla luciferase reporter gene expression in living mice. Proc. Natl Acad. Sci. USA 99, 377–382 (2002).
Nicholson, J. K., Connelly, J., Lindon, J. C. & Holmes, E. Metabonomics: a platform for studying drug toxicity and gene function. Nature Rev. Drug Discov. 1, 153–161 (2002).
Author information
Authors and Affiliations
Corresponding author
Related links
Related links
FURTHER INFORMATION
Academy of Molecular Imaging (AMI)
Encyclopedia of Life Sciences
Molecular Imaging (MOLI) Database
Society for Magnetic Resonance in Medicine (ISMRM)
Society for Molecular Imaging (SMI)
Glossary
- MAGNETIC RESONANCE IMAGING
-
(MRI). A powerful diagnostic imaging method that uses radiowaves in the presence of a magnetic field to extract information from certain atomic nuclei (most commonly hydrogen). It is primarily used for producing anatomical images, but also gives information on the physico-chemical state of tissues, flow, diffusion, motion and, more recently, molecular targets.
- X-RAY COMPUTED TOMOGRAPHY
-
(CT). As generated X-rays pass through different types of tissue, they are deflected or absorbed to different degrees. CT uses X-rays to obtain three-dimensional images by rotating an X-ray source around the subject and measuring the intensity of transmitted X-rays from different angles.
- POSITRON EMISSION TOMOGRAPHY
-
(PET). A tomographic imaging technique that detects nuclides as they decay by positron emission.
- SEROTONIN SYSTEM
-
This is an important neurotransmission network involved in aggressive and sexual behaviour, depression, anxiety, the sleep–wake cycle, moods, thermoregulation and other functions.
- FLUORESCENCE-MEDIATED TOMOGRAPHY
-
A tomographic reconstruction method developed for in vivo imaging of fluorescent probes. Images of deep structures are mathematically reconstructed by solving diffusion equations, under the assumption that photons have been scattered many times.
- FLUORESCENCE REFLECTANCE IMAGING
-
A simple method of image acquisition similar to fluorescence microscopy, except that different optics allow image acquisition of whole animals. Mostly suited for surface tumours or surgically exposed tumours.
Rights and permissions
About this article
Cite this article
Rudin, M., Weissleder, R. Molecular imaging in drug discovery and development. Nat Rev Drug Discov 2, 123–131 (2003). https://doi.org/10.1038/nrd1007
Issue Date:
DOI: https://doi.org/10.1038/nrd1007
This article is cited by
-
Ir(III)-based Ratiometric Hypoxic Probe for Cell Imaging
Chinese Journal of Polymer Science (2023)
-
Practical Guidance for Developing Small-Molecule Optical Probes for In Vivo Imaging
Molecular Imaging and Biology (2023)
-
Carboranes in drug discovery, chemical biology and molecular imaging
Nature Reviews Chemistry (2022)
-
Optical Imaging of Epigenetic Modifications in Cancer: A Systematic Review
Phenomics (2022)
-
Dual-locked spectroscopic probes for sensing and therapy
Nature Reviews Chemistry (2021)