Abstract
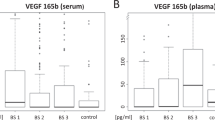
Antiangiogenic therapies that target VEGF or its receptors have become a mainstay of cancer therapy in multiple malignancies. However, the clinical efficacy of these agents is less than originally anticipated and, in most settings, requires the addition of cytotoxic chemotherapy suggesting that, as for other targeted therapies, VEGF inhibitors will require selection of patient subpopulations to achieve maximal clinical benefit. Without the identification and use of predictive biomarkers for VEGF-targeted agents, and other agents that target the vasculature, further improvements in current clinical outcomes are unlikely. Exciting new data presented in 2011 at the ESMO conference showed that retrospective evaluation of plasma concentrations of VEGF-A predicted progression-free survival and/or overall survival benefit from bevacizumab in phase III trials in certain tumour types; prospective evaluation of the assay is required. This endeavour should be followed by further biomarker research, requiring inter-laboratory collaboration and high-quality, adequately powered clinical trials.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Folkman, J. Tumor angiogenesis: therapeutic implications. N. Engl. J. Med. 28 5, 1182–1186 (1971).
Langer, R., Brem, H., Falterman, K., Klein, M. & Folkman, J. Isolations of a cartilage factor that inhibits tumor neovascularization. Science 193, 70–72 (1976).
Boehm, T., Folkman, J., Browder, T. & O'Reilly, M. S. Antiangiogenic therapy of experimental cancer does not induce acquired drug resistance. Nature 39 0, 404–407 (1997).
O'Reilly, M. S. et al. Endostatin: an endogenous inhibitor of angiogenesis and tumor growth. Cell 88, 277–285 (1997).
Brem, H. & Folkman, J. Analysis of experimental antiangiogenic therapy. J. Pediatr. Surg. 28, 445–451 (1993).
Citeline. TrialTrove [online], (2012).
National Cancer Institute. Angiogenesis inhibitors—factsheet [online], (2011).
Cannistra, S. A. et al. Phase II study of bevacizumab in patients with platinum-resistant ovarian cancer or peritoneal serous cancer. J. Clin. Oncol. 25, 5180–5186 (2007).
Raymond, E. et al. Sunitinib malate for the treatment of pancreatic neuroendocrine tumors. N. Engl. J. Med. 364, 501–513 (2011).
Jayson, G. C. et al. Evaluation of plasma VEGFA as a potential predictive pan-tumour biomarker for bevacizumab [abstract 804]. Eur. J. Cancer 47 (Suppl. 1), S96 (2011).
Miles, D. et al. Plasma biomarker analyses in the AVADO phase III randomized study of first-line bevacizumab + docetaxel in patients with HER2-negative metastatic breast cancer [abstract P2-16-04]. Cancer Res. 70 (Suppl.), 235 (2010).
Hurwitz, H. et al. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N. Engl. J. Med. 350, 2335–2342 (2004).
Kristensen, G. et al. Result of interim analysis of overall survival in the GCIG ICON7 phase III randomized trial of bevacizumab in women with newly diagnosed ovarian cancer [abstract]. J. Clin. Oncol. 29 (Suppl.), LBA5006 (2011).
Llovet, J. M. et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 359, 378–390 (2008).
Yang, J. C. et al. A randomized trial of bevacizumab, an anti-vascular endothelial growth factor antibody, for metastatic renal cancer. N. Engl. J. Med. 349, 427–434 (2003).
Friedman, H. S. et al. Bevacizumab alone and in combination with irinotecan in recurrent glioblastoma. J. Clin. Oncol. 27, 4733–4740 (2009).
Sandler, A. et al: Paclitaxel-carboplatin alone or with bevacizumab for non-small-cell lung cancer. N. Engl. J. Med. 355, 2542–2550 (2006).
National Cancer Institute. FDA approval for bevacizumab [online], (2011).
Hauschild, A. et al. Results of a phase III, randomized, placebo-controlled study of sorafenib in combination with carboplatin and paclitaxel as second-line treatment in patients with unresectable stage III or stage IV melanoma. J. Clin. Oncol. 27, 2823–2830 (2009).
Kindler, H. L. et al. Gemcitabine plus bevacizumab compared with gemcitabine plus placebo in patients with advanced pancreatic cancer: phase III trial of the Cancer and Leukemia Group B (CALGB 80303). J. Clin. Oncol. 28, 3617–3622 (2010).
Van Cutsem, E. et al. Phase III trial of bevacizumab in combination with gemcitabine and erlotinib in patients with metastatic pancreatic cancer. J. Clin. Oncol. 27, 2231–2237 (2009).
Kelly, W. K. et al. A randomized, double-blind, placebo-controlled phase III trial comparing docetaxel, prednisone, and placebo with docetaxel, prednisone, and bevacizumab in men with metastatic castration-resistant prostate cancer (mCRPC): survival results of CALGB 90401 [abstract]. J. Clin. Oncol. 28 (Suppl.), LBA4511 (2010).
Martin, M. et al. Motesanib, or open-label bevacizumab, in combination with paclitaxel, as first-line treatment for HER2-negative locally recurrent or metastatic breast cancer: a phase 2, randomised, double-blind, placebo-controlled study. Lancet Oncol. 12, 369–376 (2011).
Spigel, D. R. et al. Randomized, double-blind, placebo-controlled, phase II trial of sorafenib and erlotinib or erlotinib alone in previously treated advanced non-small-cell lung cancer. J. Clin. Oncol. 29, 2582–2589 (2011).
Scagliotti, G. et al. Phase III study of carboplatin and paclitaxel alone or with sorafenib in advanced non-small-cell lung cancer. J. Clin. Oncol. 28, 1835–1842 (2010).
Cheng, A. et al. Phase III trial of sunitinib (Su) versus sorafenib (So) in advanced hepatocellular carcinoma (HCC) [abstract]. J. Clin. Oncol. 29 (Suppl.), a4000 (2011).
Sanofi-Aventis. Sanofi-Aventis and Regeneron report top-line results from phase III study with aflibercept (VEGF Trap) in second-line non-small cell lung cancer [online], (2011).
Van Cutsem, E. et al. Intravenous (IF) aflibercept versus placebo in combination with irinotecan/5-FU (FOLFIRI) for second-line treatment of metastatic colorectal cancer (MCC): results of a multinational phase III trial (EFC10262-VELOUR) [abstract]. Ann. Oncol. 22 (Suppl. 5), aO–0024 (2011).
Ellis, L. M. & Hicklin, D. J. VEGF-targeted therapy: mechanisms of anti-tumour activity. Nat. Rev. Cancer 8, 579–591 (2008).
Saltz, L. B. et al. Bevacizumab in combination with oxaliplatin-based chemotherapy as first-line therapy in metastatic colorectal cancer: a randomized phase III study. J. Clin. Oncol. 26, 2013–2019 (2008).
O'Connor, J. P. et al. Quantifying antivascular effects of monoclonal antibodies to vascular endothelial growth factor: insights from imaging. Clin. Cancer Res. 15, 6674–6682 (2009).
Wedge, S. R. et al. AZD2171: a highly potent, orally bioavailable, vascular endothelial growth factor receptor-2 tyrosine kinase inhibitor for the treatment of cancer. Cancer Res. 65, 4389–4400 (2005).
Wedam, S. B. et al. Antiangiogenic and antitumor effects of bevacizumab in patients with inflammatory and locally advanced breast cancer. J. Clin. Oncol. 24, 769–777 (2006).
Willett, C. G. et al. Direct evidence that the VEGF-specific antibody bevacizumab has antivascular effects in human rectal cancer. Nat. Med. 10, 145–147 (2004).
Winkler, F. et al. Kinetics of vascular normalization by VEGFR2 blockade governs brain tumor response to radiation: role of oxygenation, angiopoietin-1, and matrix metalloproteinases. Cancer Cell 6, 553–563 (2004).
Batchelor, T. T. et al. AZD2171, a pan-VEGF receptor tyrosine kinase inhibitor, normalizes tumor vasculature and alleviates edema in glioblastoma patients. Cancer Cell 11, 83–95 (2007).
Jackson, A., O'Connor, J. P., Parker, G. J. & Jayson, G. C. Imaging tumor vascular heterogeneity and angiogenesis using dynamic contrast-enhanced magnetic resonance imaging. Clin. Cancer Res. 13, 3449–3459 (2007).
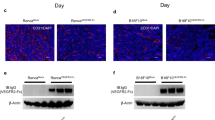
Li, J.-L. et al. DLL4-Notch signaling mediates tumor resistance to anti-VEGF therapy in vivo. Cancer Res. 71, 6073–6083 (2011).
Duff, S. E. et al. Vascular endothelial growth factors and receptors in colorectal cancer: implications for anti-angiogenic therapy. Eur. J. Cancer 42, 112–117 (2006).
Smith, N. R. et al. Vascular endothelial growth factor receptors VEGFR-2 and VEGFR-3 are localized primarily to the vasculature in human primary solid cancers. Clin. Cancer Res. 16, 3548–3561 (2010).
Ricci-Vitiani, L. et al. Tumour vascularization via endothelial differentiation of glioblastoma stem-like cells. Nature 468, 824–828 (2010).
Medici, D. et al. Conversion of vascular endothelial cells into multipotent stem-like cells. Nat. Med. 16, 1400–1406 (2010).
Shojaei, F. et al. Tumor refractoriness to anti-VEGF treatment is mediated by CD11b+Gr1+ myeloid cells. Nat. Biotech. 25, 911–920 (2007).
Grothey, A. et al. Bevacizumab beyond first progression is associated with prolonged overall survival in metastatic colorectal cancer: results from a large observational cohort study (BRiTE). J. Clin. Oncol. 26, 5326–5334 (2008).
US National Library of Medicine. ClinicalTrials.gov [online], (2011).
Shojaei, F. et al. G-CSF-initiated myeloid cell mobilization and angiogenesis mediate tumor refractoriness to anti-VEGF therapy in mouse models. Proc. Natl Acad. Sci. USA 106, 6742–6747 (2009).
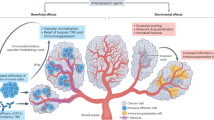
Motz, G. T. & Coukos, G. The parallel lives of angiogenesis and immunosuppression: cancer and other tales. Nat. Rev. Immunol. 11, 702–711 (2011).
Gabrilovich, D. I. et al. Production of vascular endothelial growth factor by human tumors inhibits the functional maturation of dendritic cells. Nat. Med. 10, 1096–1103 (1996).
Murukesh, N., Dive, C. & Jayson, G. C. Biomarkers of angiogenesis and their role in the development of VEGF inhibitors. Br. J. Cancer 102, 8–18 (2010).
Gourley, C. et al. Establishing a molecular taxonomy for epithelial ovarian cancer (EOC) from 363 formalin-fixed paraffin embedded (FFPE) specimens [abstract]. J. Clin. Oncol. 29 (Suppl.), a5000 (2011).
Jubb, A. M. et al. Impact of vascular endothelial growth factor-A expression, thrombospondin-2 expression, and microvessel density on the treatment effect of bevacizumab in metastatic colorectal cancer. J. Clin. Oncol. 24, 217–227 (2006).
Fabi, A. et al. HER2 protein and gene variation between primary and metastatic breast cancer: significance and impact on patient care. Clin. Cancer Res. 17, 2055–2064 (2011).
Van Cutsem, E. et al. Rash as a marker for the efficacy of gemcitabine plus erlotinib-based therapy in pancreatic cancer: results from the AViTA study [abstract]. ASCO Gastrointestinal Cancers Symp. a117 (2009).
Kang, Y. et al. AVAGAST: A randomized, double-blind, placebo-controlled, phase III study of first-line capecitabine and cisplatin plus bevacizumab or placebo in patients with advanced gastric cancer (AGC) [abstract]. J. Clin. Oncol. 28 (Suppl. 18), LBA4007 (2011).
Khambata-Ford, S. et al. Expression of epiregulin and amphiregulin and K-ras mutation status predict disease control in metastatic colorectal cancer patients treated with cetuximab. J. Clin. Oncol. 25, 3230–3237 (2007).
Mitchell, C. L. et al. Identification of early predictive imaging biomarkers and their relationship to serological angiogenic markers in patients with ovarian cancer with residual disease following cytotoxic therapy. Ann. Oncol. 21, 1982–1989 (2010).
RECIST. RECIST 1.1 [online], (2010).
Choi, H. et al. Correlation of computed tomography and positron emission tomography in patients with metastatic gastrointestinal stromal tumor treated at a single institution with imatinib mesylate: proposal of new computed tomography response criteria. J. Clin. Oncol. 25, 1753–1759 (2007).
O'Connor, J. P., Jackson, A., Parker, G. J. & Jayson, G. C. DCE-MRI biomarkers in the clinical evaluation of antiangiogenic and vascular disrupting agents. Br. J. Cancer 96, 189–195 (2007).
O'Connor, J. P. et al. DCE-MRI biomarkers of tumour heterogeneity predict CRC liver metastasis shrinkage following bevacizumab and FOLFOX-6. Br. J. Cancer 105, 139–145 (2011).
Shojaei, F., Singh, M., Thompson, J. D. & Ferrara, N. Role of Bv8 in neutrophil-dependent angiogenesis in a transgenic model of cancer progression. Proc. Natl Acad. Sci. USA 105, 2640–2645 (2008).
You, W.-K. et al. VEGF and c-Met blockade amplify angiogenesis inhibition in pancreatic islet cancer. Cancer Res. 71, 4758–4768 (2011).
Hanrahan, E. O. et al. Distinct patterns of cytokine and angiogenic factor modulation and markers of benefit for vandetanib and/or chemotherapy in patients with non-small-cell lung cancer. J. Clin. Oncol. 2 8, 193–201 (2010).
Leenders, W. P. et al. Antiangiogenic therapy of cerebral melanoma metastases results in sustained tumor progression via vessel co-option. Clin. Cancer Res. 10, 6222–6230 (2004).
Pezzella, F. et al. Non-small-cell lung carcinoma tumor growth without morphological evidence of neo-angiogenesis. Am. J. Pathol. 151, 1417–1423 (1997).
Stessels, F. et al. Breast adenocarcinoma liver metastases, in contrast to colorectal cancer liver metastases, display a non-angiogenic growth pattern that preserves the stroma and lacks hypoxia. Br. J. Cancer 90, 1429–1436 (2004).
Sardari Nia, P., Hendriks, J., Friedel, G., Van Schil, P. & Van Marck, E. Distinct angiogenic and non-angiogenic growth patterns of lung metastases from renal cell carcinoma. Histopathology 5 1, 354–361 (2007).
Hida, K. et al. Tumor-associated endothelial cells with cytogenetic abnormalities. Cancer Res. 64, 8249–8255 (2004).
Sheldon, H. et al. New mechanism for notch signaling to endothelium at a distance by Delta-like 4 incorporation into exosomes. Blood 116, 2385–2394 (2010).
Miller, K. D. E2100: a phase III trial of paclitaxel versus paclitaxel/bevacizumab for metastatic breast cancer. Clin Breast Cancer 3, 421–422 (2003).
Perren, T. J. et al. A phase 3 trial of bevacizumab in ovarian cancer. N. Engl. J. Med. 36 5, 2484–2496 (2011).
Author information
Authors and Affiliations
Contributions
All the authors contributed to researching data for the article, discussion of content, and to writing and editing the article prior to submission.
Corresponding author
Ethics declarations
Competing interests
G. C. Jayson receives grant/research support from AstraZeneca, Genentech and Roche. He has sat on advisory boards for AstraZeneca, Aveo, Genentech, Merck and Roche. D. J. Hicklin is a stock holder at Merck. L. M. Ellis declares that he serves as an ad hoc consultant for Genentech/Roche and Sanofi-Aventis.
Rights and permissions
About this article
Cite this article
Jayson, G., Hicklin, D. & Ellis, L. Antiangiogenic therapy—evolving view based on clinical trial results. Nat Rev Clin Oncol 9, 297–303 (2012). https://doi.org/10.1038/nrclinonc.2012.8
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrclinonc.2012.8
This article is cited by
-
Angiogenic signaling pathways and anti-angiogenic therapy for cancer
Signal Transduction and Targeted Therapy (2023)
-
Targeting angiogenesis in oncology, ophthalmology and beyond
Nature Reviews Drug Discovery (2023)
-
A pharmacogenetic interaction analysis of bevacizumab with paclitaxel in advanced breast cancer patients
npj Breast Cancer (2022)
-
Integrin alpha9 emerges as a key therapeutic target to reduce metastasis in rhabdomyosarcoma and neuroblastoma
Cellular and Molecular Life Sciences (2022)
-
Tumor angiogenesis: causes, consequences, challenges and opportunities
Cellular and Molecular Life Sciences (2020)