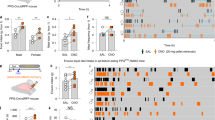
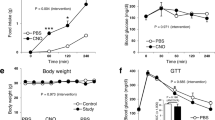
Abstract
Glucagon–like peptide 1 (GLP1) is postulated to regulate blood glucose and satiety, but the biological importance of GLP1 as an incretin and neuropeptide remains controversial. The regulation of nutrient–induced insulin secretion is dependent on the secretion of incretins, gut–derived peptides that potentiate insulin secretion from the pancreatic islets1. To ascertain the relative physiological importance of GLP1 as a regulator of feeding behavior and insulin secretion, we have generated mice with a targeted disruption of the GLP1 receptor gene (GLP1R). These GLP1R−/− mice are viable, develop normally but exhibit increased levels of blood glucose following oral glucose challenge in association with diminished levels of circulating insulin. It is surprising that they also exhibit abnormal levels of blood glucose following intraperitoneal glucose challenge. Intracerebroventricular administration of GLP1 inhibited feeding in wild–type mice but not in GLP1R−/− mice; however, no evidence for abnormal body weight or feeding behavior was observed in GLP1R−/− mice. These observations demonstrate that GLP1 plays a central role in the regulation of glycemia; however, disruption of GLP1/GLP1R signaling in the central nervous system is not associated with perturbation of feeding behavior or obesity in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Creutzfeldt, W. & Ebert, R. New developments in the incretin concept today. Diabetologi 28, 565–573 (1985).
Mojsov, S., Weir, G.C. & Habener, J.F., Glucagon-like peptide I (7–37) co-encoded in the glucagon gene is potent stimulator of insulin release in the perfused rat pancreas. J. Clin. Invest. 79, 616–619 (1987).
Kreymann, B., Ghatei, M.A., Williams, G. & Bloom, S.R. Glucagon-like peptide-1 7–36:A physiological incretin in man. Lancet ii, 1300–304 (1987).
Holst, J.J., Orskov, C., Nielsen, O.V. & Schwartz, T.W. Truncated glucagon-like peptide I, an insulin-releasing hormone from the distal gut. FERS Lett. 211, 169–174 (1987).
Fehmann, H.-C., Goke, R. & Goke, B. Cell and molecular biology of the incretin hormones glucagon-like peptide 1 and glucose-dependent releasing polypeptide. Endocrine Rev. 16, 390–410 (1995).
Turton, M.D. et al. role for glucagon-like peptide-1 in the central regulation of feeding. Nature 379, 69–72 (1996).
Nauck, M.A. et al. Preserved incretin activity of glucagon-like peptide 1 [7–36 amide] but not of synthetic human gastric inhibitory polypeptide in patients with type-2 diabetes mellitus. J. Clin. Invest. 91, 301–307 (1993).
Nauck, M.A. et al> Normalization of fasting hyperglycaemi by exogenous glucagon-like peptide 1 (7–36 amide) in type 2 (non-insulin-dependent) dibetic patients. Diabetologi 36, 741–744 (1993).
Gutniak, M., Orskov, C., Holst, J.J.B. & Efendic, S. Antidiabetogenic effect of glucagon-like peptide-1 (7–36)amide in normal subjects and patients with diabetes mellitus. N. Engl.J. Med. 326, 1316–1322 (1992).
Dupre, J. et al> Glucagon-like peptide I reduces postprandial glycemic excursions in IDDM. Diabetes 44, 626–630 (1995).
Thorens, B. Expression cloning of the pancreatic B cell receptor for the gluco-in-cretin hormone glucagon-like peptide 1. Proc. Natl. Acad. Sci. USA 89, 8641–8645 (1992).
Willms, B. et al> Gastric emptying, glucose responses, and insulin secretion after liquid test meal: Effects of exogenous glucagon-like peptide-1 (GLP-1)-(7–36)amide in type 2 (non-insulin-dependent) diabetic patients. J. Clin. Endocrinol. Metab. 81, 327–332 (1996).
D' Alessio, D.A., Kahn, S.E., Leusner, C.R. & Ensinck, J.W. Glucagon-like peptide 1 enhances glucose tolerance both by stimulation of insulin release and by increasing insulin-independent glucose disposal. J. Clin. Invest. 93, 2263–2266 (1994).
D' Alessio, D.A., Prigeon, R.L. .& Ensinck, J.W. Enteral enhancement of glucose disposition by both insulin-dependent and insulin-independent processes — A physiological role of glucagon-like peptide I. Diabetes 44, 1433–1437 (1995).
Toft-Nielsen, M., Madsbad, S. & Holst, J.J. The effect of glucagon-like peptide 1 (GLP-1) on glucose elimination in healthy subjects depends on the pancreatic glucoregul tory hormones. Diabetes 45, 552–556 (1996).
Thorens, B. et al. Cloning and functional expression of the human islet GLP-1 receptor: Demonstration that exendin-4 is an agonist and exendin-(9–39) an antagonist of the receptor. Diabetes 42, 1678–1682 (1993).
Gremlich, S. et al. Cloning, functional expression, and chromosomal localization of the human pancreatic islet glucose-dependent insulinotropic polypeptide receptor. Diabetes 44, 1202–1208 (1995).
Drucker, D.J., Philippe, J., Mojsov, S., Chick, W.L. & Habener, J.F. Glucagon-like peptide I stimulates insulin gene expression and increases cyclic AMP levels in a rat islet cell line. Proc. Natl. Acad. Sci. USA 84, 3434–3438 (1987).
D' Alessio, D.A. et al. Elimination of the action of glucagon-like peptide 1 causes an impairment of glucose tolerance after nutrient ingestion by healthy ba boons. J. Clin. Invest. 97, 133–138 (1996).
Ritzel, R., Orskov, C., Holst, J.J. & Nauck, M.A., rmacokinetic, insulinotropic, and glucagonostatic properties of GLP-1 [7–36amide] after subcutaneous injection in he althy volunteers: Dose-response-relationships. Diabetologia 38, 720–725 (1995).
Hoosein, N.M. & Gurd, R.S. Human glucagon-like peptides 1 and 2 activate rat brain adenylate cyclase. FEBS Lett. 178, 83–86 (1984).
Kanse, S.M., Kreymann, B., Ghatei, M.A. & Bloom, S.R. Identification and characterization of glucagon-like peptide-1 7–36amide-binding sites in the rat brain and lung. FEBS Lett. 241, 209–212 (1988).
Campos, R.V., Lee, Y.C. & Drucker, D.J. Divergent tissue-specific and development al expression of receptors for glucagon and glucagon-like peptide-1 in the mouse. Endocrinology 134, 2156–2164 (1994).
Wei, Y. & Mojsov, S. Tissue-specific expression of the human receptor for glucagon-like peptide 1: Brain, heart and pancreatic forms have the same deduced amino acid sequences. FEBS Lett. 358, 219–224 (1995).
Erickson, J.C., Clegg, K.E. & Palmiter, R.D. Sensitivity to leptin and susceptibility to seizures of mice lacking neuropeptide Y. Nature 381, 415–418 (1996).
Egan, J.M. Montrose-Rafizadeh, C., Wang, Y., Bernier, M. & Roth, J. Glucagon-like peptide-1(7–36)amide (GLP-1) enhances insulin-stimulated glucose metabolism in 3T3-L1 adipocytes: One of several potential extrapancreatic sites of GLP-1 action. Endocrinology 135, 2070–2075 (1994).
Villanuev -Penacarrillo, M. L., Alcántar, A.I., Clemente, F., Delgado, E. & Valverde, I. Potent glycogenic effect of GLP-1 (7–36)amide in rat skeletal muscle. Diabetologi 37, 1163–1166 (1994).
Hvidberg Nielsen, M.T., Hilsted, J., Orskov, C. & Holst, J.J. Effect of glucagon-like peptide-1 (proglucgon 78–107amide) on hepatic glucose production in healthy man. Metabolism 43, 104–108 (1994).
Wheeler, M.B. et al. Functional expression of the rat glucagon-like peptide-I receptor: Evidence for coupling to both adenylyl cyclase and phospholipase-C. Endocrinology 133, 57–62 (1993).
Tybulewicz, V.L.J., Crawfor, C.E. Jackson, P.K., Bronson, R.T. & Mulligan, R.C. Neonatal lethality and lymphopenia in mice with a homozygous disruption of the c-ablproto-oncogene. Cell 65, 1153–1163 (1991).
Nagy Rossant, J., Nagy, R.W. & Roder, J.C. Derivation of completely cell culture-derived mice from early passage embryonic stem cells. Proc. Natl. Acad. Sci. USA 90, 8424–8428 (1993).
Nagy, A. & Rossant, J. Production of completely ES cell-derived fetuses. in Gene Ta rgeting: Practic lapproach, (ed. Joyner, A.L.) 147–178 (Oxford Univ. Press, Oxford, 1993).
Wurst, W. & Joyner, A.L. Production of targeted embryonic stem cell clones. in Gene Targeting: Practical Approach, (ed. Joyner, A.L.) 33–61 (Oxford Univ. Press, Oxford, 1993).
Brubaker, P.L., Lee, Y.C. & Drucker, D.J. Alterations in proglucagon processing and inhibition of proglucagon gene expression in glucagon-SV40 T antigen transgenic mice. J. Biol. Chem. 267, 20728–20733 (1992).
Akesson, T.R., Mantyh, P.W., Mantyh, C.R., Matt, D.W. & Micevych, P.E. Estrous cyclicity of 125I-cholecystokinin octapeptide binding in the ventromedial hypothalamic nucleus. Evidence for downmodulation by estrogen. Neuroendocrinology 45, 257–262 (1987).
Heinrichs, S.C. et al. Endogenous corticotropin releasing factor modulates feeding induced by neuropeptide Y or a tail-pinch stressor. Peptides 13, 879–884 (1992).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Scrocchi, L., Brown, T., Maclusky, N. et al. Glucose intolerance but normal satiety in mice with a null mutation in the glucagon–like peptide 1 receptor gene. Nat Med 2, 1254–1258 (1996). https://doi.org/10.1038/nm1196-1254
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm1196-1254
This article is cited by
-
GLP-1 metabolite GLP-1(9–36) is a systemic inhibitor of mouse and human pancreatic islet glucagon secretion
Diabetologia (2024)
-
The expanding incretin universe: from basic biology to clinical translation
Diabetologia (2023)
-
DPP-4 inhibitors and GLP-1RAs: cardiovascular safety and benefits
Military Medical Research (2022)
-
Activation of arcuate nucleus glucagon-like peptide-1 receptor-expressing neurons suppresses food intake
Cell & Bioscience (2022)
-
Signaling profiles in HEK 293T cells co-expressing GLP-1 and GIP receptors
Acta Pharmacologica Sinica (2022)