Abstract
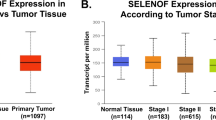
The sodium/iodide symporter mediates active iodide transport in both healthy and cancerous thyroid tissue. By exploiting this activity, radioiodide has been used for decades with considerable success in the detection and treatment of thyroid cancer. Here we show that a specialized form of the sodium/iodide symporter in the mammary gland mediates active iodide transport in healthy lactating (but not in nonlactating) mammary gland and in mammary tumors. In addition to characterizing the hormonal regulation of the mammary gland sodium/iodide symporter, we demonstrate by scintigraphy that mammary adenocarcinomas in transgenic mice bearing Ras or Neu oncogenes actively accumulate iodide by this symporter in vivo. Moreover, more than 80% of the human breast cancer samples we analyzed by immunohistochemistry expressed the symporter, compared with none of the normal (nonlactating) samples from reductive mammoplasties. These results indicate that the mammary gland sodium/iodide symporter may be an essential breast cancer marker and that radioiodide should be studied as a possible option in the diagnosis and treatment of breast cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Carrasco, N. Iodide transport in the thyroid gland. Biochim. Biophys. Acta 1154, 65–82 (1993).
Dai, G., Levy, O. & Carrasco, N. Cloning and characterization of the thyroid iodide transporter. Nature 379, 458–460 (1996).
Levy, O. et al. Characterization of the thyroid Na+/I− symporter with an anti-COOH terminus antibody. Proc. Natl. Acad. Sci. USA 94, 5568–5573 (1997).
Levy, O. et al. N-linked glycosylation of the thyroid Na+/I− Symporter (NIS): implications for its secondary structure model. J. Biol. Chem. 273, 22657–22663 (1998).
Eskandari, S. et al. Thyroid Na+/I− symporter: mechanism, stoichiometry, and specificity. J. Biol. Chem. 272, 27230–27238 (1997).
Stubbe, P., Schulte, F.J. & Heidenmann, P. Iodine deficiency and brain development. Biblthca. Nutr. Dieta 38, 206–208 (1986).
Mountford, P.J, Coakley, A.J., Fleet, I.R., Hamon, M. & Heap, R.B. Transfer of radioiodide to milk and its inhibition. Nature 322, 600 (1986).
DeGroot, L.J. in Endocrinology (ed. DeGroot, I.J.) 821–833 (Grune & Stratton Inc., Orlando, 1989).
Werner & Ingbar's The Thyroid 8th edn. (eds. Braverman, L.E. & Utiger, R.D.) 295–316 (Lippincott Williams & Wilkins, Philadelphia, 2000).
Nishizawa, K. et al. 131I in milk and rain after Chernobyl. Nature 324, 308 (1986).
Hill, C.R., Adam, I., Anderson, W., Ott, R.J. & Sowby, F.D. Iodine-131 in human thyroids in Britain following Chernobyl. Nature 321, 655–656 (1986).
Balter, M. Chernobyl's cancer toll. Science 270, 1758–1759 (1995).
Mazzaferri, E.L. NCCN thyroid carcinoma practice guidelines: NCCN proceedings. Oncology 13, 391–442 (1999).
Greenlee, R.T., Murray,T., Bolden, S. & Wingo, P. Cancer statistics 2000. A Cancer Journal for Clinicians 50, 7–33 (2000).
Socolow, E.L. & Ingbar, S.H. Metabolism of 99mpertechnetate by the thyroid gland of the rat. Endocrinology 80, 337–344 (1967).
Spitzweg, C., Joba, W., Eisenmenger, W. & Heufelder, A.E. Analysis of human sodium iodide symporter gene expression in extrathyroidal tissues and cloning of its complementary deoxyribonucleic acids from salivary gland, mammary gland, and gastric mucosa. J. Clin. Endocrinol. Metab. 83, 1746–1751 (1998).
Wakerley, J.B., O'Neill, D.S. & ter Haar, M.B. Relationship between the suckling-induced release of oxytocin and prolactin in the urethane-anaesthetized lactating rat. J. Endocrinol. 76, 493–500 (1978).
Higuschi, T., Honda, K., Fukuoka, T., Negoro, H. & Wakabayashi, K. Release of oxytocin during suckling and parturition in rat. J. Endocrinol. 105, 339–346 (1985).
McCormack, J.T. & Greenwald, G.S. Progesterone and oestradiol-17-_ concentrations in the peripheral plasma during pregnancy in the mouse. J. Endocrinol. 62, 101–107 (1974).
Rosenblatt, J. S., Mayer, A. D. & Giordano, A.L. Hormonal basis during pregnancy for the onset of maternal behaviour in the rat. Psychoneuroendocrinol. 13, 29–46 (1988).
Sinn, E. et al. Co-expression of MMTV/V-Ha-ras and MMTV/c-myc genes in transgenic mice: synergistic actions of oncogenes in vivo. Cell 49, 465–475 (1987).
Guy, C.T. et al. Expression of the neu protooncogene in the mammary epithelium of transgenic mice induces metastatic disease. Proc. Natl. Acad. Sci. USA 89, 10578–10582 (1992).
Hung, M. & Lau, Y. Basic science of HER-2/neu: a review. Semin. Oncol. 26, 51–59 (1999).
Slamon, D.J. et al. Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science 235, 177–182 (1987).
Paterson, M.C. et al. Correlation between c-erbB-2 amplification and risk of recurrent disease in node-negative breast cancer. Cancer Res. 51, 556–567 (1991).
Siegel, P.M., Ryan, E.D., Cardiff, R. D. & Muller, W. J. Elevated expression of activated forms of neu/ErbB-2 and ErbB-3 are involved in the induction of mammary tumors in transgenic mice: implications for human breast cancer. EMBO J. 18, 2149–2164 (1999).
DiGiovanna M.P. Clinical significance of HER-2/neu overexpression. Principles and Practice of Oncology 13, 1–10 (1999).
Zingg, H.H. et al. in Vasopressin and Oxytocin, Advances in Experimental Medicine and Biology (eds. H.H. Zingg, Bourque, C.W. & Bichet, D.G.) 287–297 (Plenum, New York, 1998).
Ohno, M., Zannini, M., Levy, O., Carrasco, N. & DiLauro, R. The paired domain transcription factor Pax8 binds to the upstream enhancer of the rat sodium/iodide symporter gene and participates in both thyroid-specific and cAMP dependent transcription. Mol. Cell. Biol. 19, 2051–2060 (1999).
Dorrington, J. & Gore-Langton, R.E. Prolactin inhibits oestrogen synthesis in the ovary. Nature 290, 600–602 (1981).
Gitay-Goren, H., Lindenbaum, E.S. & Kraiem, Z. Prolactin inhibits hCG-stimulated steroidogenesis and cAMP accumulation, possibly by increasing phosphodiesterase activity, in rat granulosa cell cultures. Mol. Cell. Endocrinol. 61, 69–76 (1989).
Krasnow, J.S., Hickey, G. J. & Richards, J.S. Regulation of aromatase mRNA and estradiol biosynthesis in rat ovarian granulosa and luteal cells by prolactin. Mol. Endocrinol. 4, 13–21 (1990).
Villanueva, L.A., Mendez, I., Ampuero, S. & Larrea, F. The prolactin inhibition of follicle-stimulating hormone-induced aromatase activity in cultured rat granulosa cells is in part tyrosine kinase and protein kinase-C dependent. Mol. Hum. Reprod. 2, 725–731 (1996).
Mattheij, J.A.M., Kuipers, M.A.G., Swarts, J.J.M. & Verstijnen, C.P.H.J. Intraperitonal infusion of EDTA in the rat blocks completely the prolactin rise in the plasma during suckling. Horm. Res. 16, 219–229 (1982).
Grazzini, E., Guillon, G., Mouillac, B. & Zingg, H.H. Inhibition of oxytocin receptor function by direct binding of progesterone. Nature 392, 509–512 (1998).
Fitzgerald, P.J. & Foote, F.W. Jr. The function of various types of thyroid carcinoma as revealed by the radioautographic demonstration of radioactive iodine. J. Clin. Endocrinol. 9, 1153–1170 (1949).
Pochin, E.E., Cunningham, R.M. & Hilton, G. Quantitative measurements of radioiodine retention in thyroid carcinoma. J. Clin. Endocrinol. Metab. 14, 1300–1308 (1954).
Valenta, L. Metastatic thyroid carcinoma in man concentrating iodide without organification. J. Clin. Endocrinol. 26, 1317–1324 (1966).
Cancroft, E.T. & Goldsmith, S.J. 99mTc-pertechnetate scintigraphy as an aid in the diagnosis of breast masses. Radiology 106, 441–444 (1973).
Smanik, P.A. et al. Cloning of the human sodium iodide symporter. Bioch. Biophys. Res. Commun. 226, 339–345 (1996).
Carrasco, N., Herzlinger, D., Danho, W. & Kaback, H.R. Preparation of monoclonal antibodies against the lac permease of Escherichia coli. Methods Enzymol. 125, 453–467 (1986).
Kaminsky, S.M., Levy, O., Salvador, C., Dai, G. & Carrasco, N. Na+/I− symport activity is present in membrane vesicles from thyrotropin-deprived non-I—transporting cultured thyroid cells. Proc. Natl. Acad. Sci. USA 91, 3789–3793 (1994).
Acknowledgements
We thank A. De la Vieja and C. Riedel for computer assistance, G. Orr for advice and T. Ciao and G. Dai for help with animal experiments. We also thank L. M. Amzel, T. Graf, and the members of the Carrasco laboratory for reviewing the manuscript. O.L. was supported by the National Institutes of Health Hepatology Research Training Grant DK-07218. R.G.P. is a recipient of the Irma T. Hirschl award. This work was supported in part by R29CA70897 and RO1CA75503 (to R.G.P.). Work at the Albert Einstein College of Medicine was supported by Cancer Center Core National Institutes of Health grant 5-P30-CA13330-26. This project was also supported by the National Institutes of Health DK-41544, the Susan G. Komen Breast Cancer Foundation, the American Cancer Society BE-79422, and the Irma T. Hirschl award (N.C.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tazebay, U., Wapnir, I., Levy, O. et al. The mammary gland iodide transporter is expressed during lactation and in breast cancer. Nat Med 6, 871–878 (2000). https://doi.org/10.1038/78630
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/78630
This article is cited by
-
Iodine Metabolism and Thyroid Function During the Perinatal Period: Maternal-Neonatal Correlation and Effects of Topical Povidone-Iodine Skin Disinfectants
Biological Trace Element Research (2023)
-
The sodium iodide symporter (NIS) as theranostic gene: potential role in pre-clinical therapy of extra-thyroidal malignancies
Clinical and Translational Imaging (2023)
-
Assessment and treatment of thyroid disorders in pregnancy and the postpartum period
Nature Reviews Endocrinology (2022)
-
Iodine in Disruption of Thyroid and Thyroid Hormone Receptive Systems
Proceedings of the Zoological Society (2021)
-
Causes of False-Positive Radioactive Iodine Uptake in Patients with Differentiated Thyroid Cancer
Current Radiology Reports (2021)