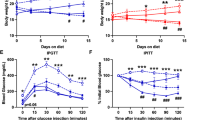
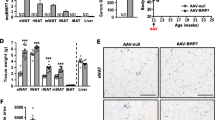
Abstract
BROWN adipose tissue, because of its capacity for uncoupled mitochondria! respiration1,2, has been implicated as an important site of facultative energy expenditure3–5. This has led to speculation that this tissue normally functions to prevent obesity3–5. Attempts to ablate or denervate brown adipose tissue surgically have been uninformative because it exists in diffuse depots and has substantial capacity for regeneration and hypertrophy6. Here we have used a transgenic toxigene approach7,8 to create two lines of transgenic mice with primary deficiency of brown adipose tissue. At 16 days, both lines have decreased brown fat and obesity. In one line, brown fat subsequently regenerates and obesity resolves. In the other line, the deficiency persists and obesity, with its morbid complications, advances. Obesity develops in the absence of hyperphagia, indicating that brown fat deficient mice have increased metabolic efficiency. As obesity progresses, transgenic animals develop hyperphagia. This study supports a critical role for brown adipose tissue in the nutritional homeostasis of mice.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Nicholls, D. G. & Locke, R. M. Physiol. Rev. 64, 1–64 (1984).
Klaus, S., Casteilla, L., Bouillaud, F. & Ricquier, D. Int. J. Biochem. 23, 791–801 (1991).
Rothwell, N. J. & Stock, M. J. Nature 281, 31–35 (1979).
Trayhurn, P. & Mercer, S. W. Biochem Soc. Trans. 14, 236–239 (1986).
Himms-Hagen, J. Prog. Lipid Res. 28, 67–115 (1989).
Rothwell, N. J. & Stock, M. J. Am. J. Physiol. 257, R253–R258 (1989).
Palmiter, R. D. et al. Cell 50, 435–443 (1987).
Breitman, M. L. et al. Science 238, 1563–1565 (1987).
Jacobsson, A., Stadler, U., Glotzer, M. A. & Kozak, L. P. J. biol. Chem. 260, 16250–16254 (1985).
Dubuc, P. U. Metabolism 25, 1567–1574 (1976).
Coleman, D. L. Diabetologia 14, 141–148 (1978).
Cameron, D. P., Cutbush, L. & Opat, F. Clin. exp. Pharmac. Physiol. 5, 41–51 (1978).
Breitman, M. L. & Bernstein, A. in Transgenic Animals (eds Grosveld, F. & Kollias, G.) 127–145 (Academic, Boston, 1992).
Bray, G. A. & York, D. A. Physiol. Rev. 59, 719–809 (1979).
Holloway, B. R. et al. Br. J. Pharmac. 104, 97–104 (1991).
Granneman, J. G., Lahners, K. N. & Chaudhry, A. Molec. Pharmac. 40, 895–899 (1991).
Muzzin, P. et al. J. biol. Chem. 266, 24053–24058 (1991).
Nahmias, C. et al. EMBO J. 10, 3721–3727 (1991).
Bloom, J. D. et al. J. med. Chem. 35, 3081–3084 (1992).
Foster, D. O. & Frydman, M. L. Can. J. Physiol. Pharmac. 57, 257–270 (1979).
Davis, T. R. A., Johnston, D. R., Bell, F. C. & Cremer, B. J. Am. J. Physiol. 198, 471–475 (1960).
Bergmeyer, H. U. Methods in Enzymatic Analysis 2nd edn (Academic, London, 1974).
Boyer, B. B. & Kozak, L. P. Molec. cell. Biol. 11, 4147–4156 (1991).
Maxwell, I. H., Maxwell, F. & Glode, L. M. Cancer Res. 46, 4660–4664 (1986).
Maxwell, F., Maxwell, I. H. & Glode, L. M. Molec. cell. Biol. 7, 1576–1579 (1987).
Breitman, M. L., Rombola, H., Maxwell, I. H., Klintworth, G. K. & Berstein, A. Molec. cell. Biol. 10, 474–479 (1990).
Salmon, D. M. W. & Flatt, J. P. Int. J. Obesity 9, 443–449 (1985).
Desautels, M., Zaror-Behrens, G. & Himms-Hagen, J. Can. J. Biochem. 56, 378–383 (1978).
Ma, S. W. Y. & Foster, D. O. Can. J. Physiol. Pharmac. 64, 1252–1258 (1986).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lowell, B., S-Susulic, V., Hamann, A. et al. Development of obesity in transgenic mice after genetic ablation of brown adipose tissue. Nature 366, 740–742 (1993). https://doi.org/10.1038/366740a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/366740a0
This article is cited by
-
Pulling the trigger: Noncoding RNAs in white adipose tissue browning
Reviews in Endocrine and Metabolic Disorders (2024)
-
Purinergic receptor: a crucial regulator of adipose tissue functions
Purinergic Signalling (2023)
-
Reduced secretion of neuronal growth regulator 1 contributes to impaired adipose-neuronal crosstalk in obesity
Nature Communications (2022)
-
Mitochondrial activity regulates the differentiation of skin-derived mesenchymal stem cells into brown adipocytes to contribute to hypertension
Stem Cell Research & Therapy (2021)
-
Overexpression of translationally controlled tumor protein ameliorates metabolic imbalance and increases energy expenditure in mice
International Journal of Obesity (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.