Abstract
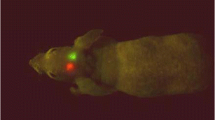
Currently, there is no mouse model of cervical cancer that allows for the study of the later stages of the disease, including metastasis. We report here the development of an orthotopic model of human cervical carcinoma in which tumor fragments are surgically implanted into the cervix of SCID mice. The human cervical carcinoma cell lines used in this study (CaSki, ME-180, and SiHa) have been engineered to stably express the fluorescent proteins enhanced green fluorescent protein (EGFP) or DsRed2, allowing for in vivo optical monitoring of tumor growth and metastasis. The cervical implants develop into large intraperitoneal masses involving the entire reproductive tract, with little local invasion of other abdominal structures. These tumors metastasize initially to local lymph nodes and later to lung, a pattern consistent with the clinical course of the disease. It was found that the use of the DsRed2 protein as a fluorescent marker has distinct advantages over EGFP due to the wavelength of its emission spectrum (575–625 nm vs 500–550 nm). Tissue penetration of light at this wavelength is greater, and the auto-fluorescence of mouse tissues is less intense, resulting in an enhanced signal to noise ratio compared to results obtained with EGFP. This model should allow for a more relevant investigation of the factors that affect the metastasis of cervical carcinoma and presents an opportunity to evaluate potential therapeutic strategies designed to prevent the spread of disease.
Abbreviations: SCID – severe combined immunodeficient; EGFP – enhanced green fluorescent protein; VEGF – vascular endothelial growth factor; PD-ECGF – platelet-derived endothelial cell growth factor; MMP – matrix metalloproteinase; HPV – human papilloma virus; ATCC – American type culture collection; α-MEM – alpha minimal essential medium; FBS – fetal bovine serum; CMV – cytomegalovirus; FACS – fluorescence activated cell sorting
Similar content being viewed by others
References
Jemal A, Murray T, Samuels A et al.Cancer statistics. CA Cancer J Clin 2003; 53: 5–26.
Bast RC, Kufe DW, Pollock RE et al.Cancer Medicine 5th edition. Hamilton, Canada: BC Decker 2000.
Graflund M, Sorbe B, Hussein A et al.The prognostic value of histopathologic grading parameters and microvessel density in patients with early squamous cell carcinoma of the uterine cervix. Int J Gynecol Cancer 2002; 12: 32–41.
Metcalf KS, Johnson N, Calvert S et al.Site specific lymph node metastasis in carcinoma of the cervix: Is there a sentinel node? Int J Gynecol Cancer 2000; 10: 411–6.
Loncaster JA, Cooper RA, Logue JP et al.Vascular endothelial growth factor (VEGF) expression is a prognostic factor for radiotherapy outcome in advanced carcinoma of the cervix. Br J Cancer 2000; 83: 620–5.
Hashimoto I, Kodama J, Seki N et al.Vascular endothelial growth factor-C expression and its relationship to pelvic lymph node status in invasive cervical cancer. Br J Cancer 2000; 85: 93–7.
Fujimoto J, Sakaguchi H, Aoki I et al.The value of platelet-derived endothelial cell growth factor as a novel predictor of advancement of uterine cervical cancers. Cancer Res 2000; 60: 3662–5.
Davidson B, Goldberg I, Kopolovic J et al.MMP-2 and TIMP-2 expression correlates with poor prognosis in cervical carcinoma–a clinicopathologic study using immunohistochemistry and mRNA in situhybridization. Gynecol Oncol 1999; 73: 372–82.
Speiser P, Kridelka F, Tempfer C et al.CD44v6 expression is an independent prognostic factor in node-negative FIGO stage IB cervical carcinoma. Int J Gynecol Cancer 1999; 9: 160–5.
Nishioka T, West CM, Gupta N et al.Prognostic significance of cerbB-2 protein expression in carcinoma of the cervix treated with radiotherapy. J Cancer Res Clin Oncol 1999; 125: 96–100.
Fyles A, Milosevic M, Hedley D et al.Tumor hypoxia has independent predictor impact only in patients with node-negative cervix cancer. J Clin Oncol 2002; 20: 680–7.
Pitson G, Fyles A, Milosevic M et al.Tumor size and oxygenation are independent predictors of nodal diseases in patients with cervix cancer. Int J Radiat Oncol Biol Phys 2001; 51: 699–703.
Hockel M, Schlenger K, Aral B et al.Association between tumor hypoxia and malignant progression in advanced cancer of the uterine cervix. Cancer Res 1996; 56: 4509–15.
Arbeit JM, Howley PM, Hanahan D. Chronic estrogen-induced cervical and vaginal squamous carcinogenesis in human papillomavirus type 16 transgenic mice. Proc Natl Acad Sci USA 1996; 93: 2930–5.
Killion JJ, Radinsky R, Fidler IJ. Orthotopic models are necessary to predict therapy of transplantable tumors in mice. Cancer Metast Rev 1998–99; 17: 279–84.
Kubota T.Metastatic models of human cancer xenografted in the nude mouse: The importance of orthotopic transplantation. J Cell Biochem 1994; 56: 4–8.
Bouvet M, Wang J, Nardin SR et al.Real-time optical imaging of primary tumor growth and multiple metastatic events in a pancreatic cancer orthotopic model. Cancer Res 2002; 62: 1534–40.
Yang M, Baranov E, Wang JW et al.Direct external imaging of nascent cancer, tumor progression, angiogenesis, and metastasis on internal organs in the fluorescent orthotopic model. Proc Natl Acad Sci USA 2002; 99: 3824–9.
Myers JN, Holsinger FC, Jasser SA et al.An orthotopic nude mouse model of oral tongue squamous cell carcinoma. Clin Cancer Res 2002; 8: 293–8.
Young SD, Hill RP. Dynamic heterogeneity: Isolation of murine tumor cell populations enriched for metastatic variants and quantification of the unstable expression of the phenotype. Clin Exp Metast 1986; 4: 153–76.
Rights and permissions
About this article
Cite this article
Cairns, R.A., Hill, R.P. A fluorescent orthotopic model of metastatic cervical carcinoma. Clin Exp Metastasis 21, 275–282 (2004). https://doi.org/10.1023/B:CLIN.0000037729.75981.9e
Issue Date:
DOI: https://doi.org/10.1023/B:CLIN.0000037729.75981.9e