Abstract
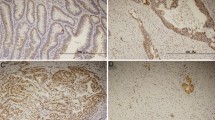
The objectives of this study are to investigate the apoptotic changes in gastric adenocarcinoma following neoadjuvant chemotherapy and to illuminate its correlation with response. One 67 gastric cancer patients with cT2-4 or TanyN1-3M0 between January 2006 and December 2007 were included. All patients had previous gastrectomy and D2 lymphadenectomy with curative intent performed. A total of 12 cycles of preoperative mFOLFOX7 chemotherapy was recommended for all patients, and 83 patients received only adjuvant chemotherapy. Resected specimens were subjected to in situ TUNEL assay and scanned with Applied Imaging Ariol SL-50. Apoptosis index (AI) was significantly higher in the patient received preoperative chemotherapy (CS group) than in the patient did not (S group). In the CS group, AI was found to have a strong positive correlation with the pathological response (rho = 0.403, P = 0.0002). A ROC curve presented a score of 49.4 as the AI cutoff value for response, dividing the CS group into two subgroups with significantly different prognoses (P = 0.003) and further allowing identification of 8 patients with significantly better prognosis out of 48 patients evaluated as grades 1a–b according to a pathological response evaluation (P = 0.022). In conclusion, AI is correlated with efficacy of preoperative chemotherapy and prognosis of gastric cancer patients following neoadjuvant chemotherapy. An AI cutoff value for response may be used as a complementary approach to current pathological response evaluations to help identify potential responders.




Similar content being viewed by others
References
Yang D, Hendifar A, Lenz C, Togawa K, Lenz F, Lurje G, et al. Survival of metastatic gastric cancer: significance of age, sex and race/ethnicity. J Gastrointest Oncol. 2011;2(2):77–84.
Cunningham D, Allum WH, Stenning SP, Thompson JN, Van de Velde CJ, Nicolson M, et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N Engl J Med. 2006;355(1):11–20.
Verlato G, Zanoni A, Tomezzoli A, Minicozzi A, Giacopuzzi S, Di Cosmo M, et al. Response to induction therapy in oesophageal and cardia carcinoma using Mandard tumour regression grade or size of residual foci. Br J Surg. 2010;97(5):719–25.
Ott K, Herrmann K, Lordick F, Wieder H, Weber WA, Becker K, et al. Early metabolic response evaluation by fluorine-18 fluorodeoxyglucose positron emission tomography allows in vivo testing of chemosensitivity in gastric cancer: long-term results of a prospective study. Clin Cancer Res. 2008;14(7):2012–8.
Lowy AM, Mansfield PF, Leach SD, Pazdur R, Dumas P, Ajani JA. Response to neoadjuvant chemotherapy best predicts survival after curative resection of gastric cancer. Ann Surg. 1999;229(3):303–8.
O’Dwyer PJ, Stevenson JP, Johnson SW. Clinical pharmacokinetics and administration of established platinum drugs. Drugs. 2000;59 Suppl 4:19–27.
McHugh PJ, Spanswick VJ, Hartley JA. Repair of DNA interstrand crosslinks: molecular mechanisms and clinical relevance. Lancet Oncol. 2001;2(8):483–90.
Ben-Izhak O, Laster Z, Araidy S, Nagler RM. TUNEL—an efficient prognosis predictor of salivary malignancies. Br J Cancer. 2007;96(7):1101–6.
JGCA. Japanese classification of gastric carcinoma: 3rd English edition. Gastric Cancer. 2011;14(2):101–12.
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45(2):228–47.
O’Connell GJ, Ryan E, Murphy KJ, Prefontaine M. Predictive value of CA 125 for ovarian carcinoma in patients presenting with pelvic masses. Obstet Gynecol. 1987;70(6):930–2.
Trillet-Lenoir V, Chapuis F, Touzet S, Barbier JY, Freyer G, Gaudin JL, et al. Any clinical benefit from the use of oncofoetal markers in the management of chemotherapy for patients with metastatic colorectal carcinomas? Clin Oncol (R Coll Radiol). 2004;16(3):196–203.
Mofidi R, Walsh R, Ridgway PF, Crotty T, McDermott EW, Keaveny TV, et al. Objective measurement of breast cancer oestrogen receptor status through digital image analysis. Eur J Surg Oncol. 2003;29(1):20–4.
Gokhale S, Rosen D, Sneige N, Diaz LK, Resetkova E, Sahin A, et al. Assessment of two automated imaging systems in evaluating estrogen receptor status in breast carcinoma. Appl Immunohistochem Mol Morphol. 2007;15(4):451–5.
Kasagi N, Ueno E, Hayashi H, Ito H. Terminal deoxynucleotidyl transferase-mediated dUTP-biotin nick end labeling (TUNEL) detects the initial phase of apoptosis in HL-60 cells induced by ultraviolet irradiation: comparison between biochemical findings and morphological appearance of DNA fragmentation (in Japanese). Trans Soc Pathol Jpn. 1995;84:173.
Kasagi N, Gomyo Y, Shirai H, Tsujitani S, Ito H. Apoptotic cell death in human gastric carcinoma: analysis by terminal deoxynucleotidyl transferase-mediated dUTP-biotin nick end labeling. Jpn J Cancer Res. 1994;85(9):939–45.
Moss SF, Calam J, Agarwal B, Wang S, Holt PR. Induction of gastric epithelial apoptosis by Helicobacter pylori. Gut. 1996;38(4):498–501.
Anti M, Armuzzi A, Iascone E, Valenti A, Lippi ME, Covino M, et al. Epithelial-cell apoptosis and proliferation in Helicobacter pylori-related chronic gastritis. Ital J Gastroenterol Hepatol. 1998;30(2):153–9.
Hahm KB, Lee KJ, Choi SY, Kim JH, Cho SW, Yim H, et al. Possibility of chemoprevention by the eradication of Helicobacter pylori: oxidative DNA damage and apoptosis in H. pylori infection. Am J Gastroenterol. 1997;92(10):1853–7.
Jones NL, Shannon PT, Cutz E, Yeger H, Sherman PM. Increase in proliferation and apoptosis of gastric epithelial cells early in the natural history of Helicobacter pylori infection. Am J Pathol. 1997;151(6):1695–703.
Mannick EE, Bravo LE, Zarama G, Realpe JL, Zhang XJ, Ruiz B, et al. Inducible nitric oxide synthase, nitrotyrosine, and apoptosis in Helicobacter pylori gastritis: effect of antibiotics and antioxidants. Cancer Res. 1996;56(14):3238–43.
Rudi J, Kuck D, Strand S, von Herbay A, Mariani SM, Krammer PH, et al. Involvement of the CD95 (APO-1/Fas) receptor and ligand system in Helicobacter pylori-induced gastric epithelial apoptosis. J Clin Invest. 1998;102(8):1506–14.
Steininger H, Faller G, Dewald E, Brabletz T, Jung A, Kirchner T. Apoptosis in chronic gastritis and its correlation with antigastric autoantibodies. Virchows Arch. 1998;433(1):13–8.
Imatani A, Sasano H, Yabuki N, Kato K, Ohara S, Asaki S, et al. In situ analysis of tissue dynamics and p53 expression in human gastric mucosa. J Pathol. 1996;179(1):39–42.
Ishida M, Gomyo Y, Tatebe S, Ohfuji S, Ito H. Apoptosis in human gastric mucosa, chronic gastritis, dysplasia and carcinoma: analysis by terminal deoxynucleotidyl transferase-mediated dUTP-biotin nick end labelling. Virchows Arch. 1996;428(4–5):229–35.
Satoh K, Kawata H, Tokumaru K, Kumakura Y, Ishino Y, Kawakami S, et al. Change in apoptosis in the gastric surface epithelium and glands after eradication of Helicobacter pylori. Dig Liver Dis. 2003;35(2):78–84.
Hirasawa R, Tatsuta M, Iishi H, Yano H, Baba M, Uedo N, et al. Increase in apoptosis and decrease in ornithine decarboxylase activity of the gastric mucosa in patients with atrophic gastritis and gastric ulcer after successful eradication of Helicobacter pylori. Am J Gastroenterol. 1999;94(9):2398–402.
Graeber TG, Osmanian C, Jacks T, Housman DE, Koch CJ, Lowe SW, et al. Hypoxia-mediated selection of cells with diminished apoptotic potential in solid tumours. Nature. 1996;379(6560):88–91.
Targa AC, Cesar AC, Cury PM, Silva AE. Apoptosis in different gastric lesions and gastric cancer: relationship with Helicobacter pylori, overexpression of p53 and aneuploidy. Genet Mol Res. 2007;6(3):554–65.
Forones NM, Carvalho AP, Giannotti-Filho O, Lourenco LG, Oshima CT. Cell proliferation and apoptosis in gastric cancer and intestinal metaplasia. Arq Gastroenterol. 2005;42(1):30–4.
Jensen K, Afroze S, Munshi MK, Guerrier M, Glaser SS. Mechanisms for nicotine in the development and progression of gastrointestinal cancers. Transl Gastrointest Cancer. 2011. doi:10.3978/j.issn.2224-4778.2011.12.01.
Acknowledgments
This study is supported by grants from Natural Science Foundation of China (81071983), Beijing Municipal Science & Technology Commission NOVA program (2007B-057).
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding authors
Additional information
The authors have no commercial, proprietary, or financial interest in the products or companies described in this article.
A correction to this article is available at https://content.iospress.com/articles/tumor-biology/tub219005
Rights and permissions
About this article
Cite this article
Jia, Y., Dong, B., Tang, L. et al. Apoptosis index correlates with chemotherapy efficacy and predicts the survival of patients with gastric cancer. Tumor Biol. 33, 1151–1158 (2012). https://doi.org/10.1007/s13277-012-0357-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-012-0357-8