Abstract
Objective
The usefulness of 2-deoxy-2-[F-18]fluoro-d-glucose (FDG)–positron emission tomography (PET) in monitoring breast cancer response to chemotherapy has previously been reported. Elevated uptake of FDG by treated tumors can persist however, particularly in the early period after treatment is initiated. 3′-[F-18]Fluoro-3′-deoxythymidine (FLT) has been developed as a marker for cellular proliferation and, in principle, could be a more accurate predictor of the long-term effect of chemotherapy on tumor viability. We examined side-by-side FDG and FLT imaging for monitoring and predicting tumor response to chemotherapy.
Methods
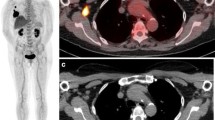
Fourteen patients with newly diagnosed primary or metastatic breast cancer, who were about to commence a new pharmacologic treatment regimen, were prospectively studied. Dynamic 3-D PET imaging of uptake into a field of view centered over tumor began immediately after administration of FDG or FLT (150 MBq). After 45 minutes of dynamic acquisition, a clinically standard whole-body PET scan was acquired. Patients were scanned with both tracers on two separate days within one week of each other (1) before beginning treatment, (2) two weeks following the end of the first cycle of the new regimen, and (3) following the final cycle of that regimen, or one year after the initial PET scans, whichever came first. (Median and mean times of early scans were 5.0 and 6.6 weeks after treatment initiation; median and mean times for late scans were 26.0 and 30.6 weeks after treatment initiation.) Scan data were analyzed on both tumor-by-tumor and patient-by-patient bases, and compared to each patient's clinical course.
Results
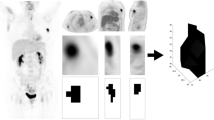
Mean change in FLT uptake in primary and metastatic tumors after the first course of chemotherapy showed a significant correlation with late (av. interval 5.8 months) changes in CA27.29 tumor marker levels (r = 0.79, P = 0.001). When comparing changes in tracer uptake after one chemotherapy course versus late changes in tumor size as measured by CT scans, FLT was again a good predictor of eventual tumor response (r = 0.74, P = 0.01). Tumor uptake of FLT was near-maximal by 10 minutes after injection. The time frame five to 10 minutes postinjection of FLT produced standardized uptake value (SUV) values highly correlated with SUV values obtained after 45-minute uptake (r = 0.83, P < 0.0001), and changes in these early SUVs after the first course of chemotherapy correlated with late changes in CA27.29 (r = 0.93, P = 0.003).
Conclusion
A 10-minute FLT-PET scan acquired two weeks after the end of the first course of chemotherapy is useful for predicting longer-term efficacy of chemotherapy regimens for women with breast cancer.




Similar content being viewed by others
References
Shields AF, Grierson JR, Dohmen BM, et al. (1998) Imaging proliferation in vivo with [F-18]FLT and positron emission tomography. Nat Med 4:1334–1336
Lu L, Samuelsson L, Bergstrom M, et al. (2002) Rat studies comparing 11C-FMAU, 18F-FLT, and 76Br-BFU as proliferation markers. J Nucl Med 43:1688–1698
Dittman H, Dohmen BM, Kehibach R, et al. (2002) Early changes in [18F]FLT uptake after chemotherapy: an experimental study. Eur J Nucl Med 29:1462–1469
Veselle H, Grierson J, Muzi M, et al. (2002) In vivo validation of 3′deoxy-3′-[18F]fluorothymidine ([18F]FLT) as a proliferation imaging tracer in humans: correlation of [18F]FLT uptake by positron emission tomography with Ki-67 immunohistochemistry and flow cytometry in human lung tumors. Clin Cancer Res 8:3315–3323
Wagner M, Seitz U, Buck A, et al. (2003) 3′-[18F]fluoro-3′-deoxythymidine ([18F]-FLT) as positron emission tomography tracer for imaging proliferation is a murine B-cell lymphoma model and in the human disease. Cancer Res 63:2681–2687
Smyczek-Gargya B, Fersis N, Dittman H, et al. (2004) PET with [18F]fluorothymidine for imaging of primary breast cancer: a pilot study. Eur J Nucl Med Mol Imaging 31:720–724
Dittman H, Dohman BM, Paulsen F, et al. (2003) [(18)F]FLT PET for diagnosis and staging of thoracic tumors. Eur J Nucl Med Mol Imaging 30:1407–1412
Satyamurthy N, Amarasekara B, Alvord CW, Barrio JR, Phelps ME (2002) Tantalum 18O-water target for the production of 18F-fluoride with high reactivity for the preparation of 2-desoxy-2-18F-fluoro-d-glucose. Mol Imaging Biol 4:65–70
Blocher A, Kuntzsch M, Wei R, Machulla H (2002) Synthesis and labeling of 5′-O-(4,4′-dimethoxytrityl)-2,3′-anhydrothymidine for [18F]FLT preparation. J Radioanal Nucl Chem 251:55–58
Machulla HJ, Blocher A, Kuntzsch M, Piert, M, Wei R, Grierson JR (2000) Simplified labeling approach for synthesizing 3′-deoxy-3′-[18F]fluorothymidine ([18F]FLT). J Radioanal Nucl Chem 243:843–846
Sturgeon C (2002) Practice guidelines for tumor marker use in the clinic. Clin Chem 48:1151–1159
Huovinen R, Leskinen-Kallio S, Nagren K, Lehikoinen P, Ruotsalainen U, Teras M (1993) Carbon-11-methionine and PET in evaluation of treatment response of breast cancer. Br J Cancer 67:787–791
Mortimer JE, Dehdashti F, Siegel BA, Katzenellenbogen JA, Fracasso P, Welch MJ (1996) Positron emission tomography with 2-[18F]fluoro-2-deoxy-D-glucose and 16alpha-[18F]fluoro-17beta-estradiol in breast cancer: correlation with estrogen receptor status and response to systemic therapy. Clin Cancer Res 2:933–939
Schirrmeister H, Guhlmann A, Kotzerke J, et al. (1999) Early detection and accurate description of extent of metastatic bone disease in breast cancer with fluoride ion and positron emission tomography. J Clin Oncol 17:2381–2389
Beaney RP, Lammertsma AA, Jones T, McKenzie CG, Halnan KE (1994) Positron emission tomography for in-vivo measurement of regional blood flow, oxygen utilisation, and blood volume in patients with breast carcinoma. Lancet 1:131–134
Wilson CB, Lammertsma AA, McKenzie CG, Sikora K, Jones T (1992) Measurements of blood flow and exchanging water space in breast tumors using positron emission tomography: a rapid and noninvasive dynamic method. Cancer Res 52:1592–1597
Lacan G, Kesner AL, Gangloff A, Zhang L, Czernin J, Melega W, Silverman DH (2005) Synthesis of 2-[(2-chloro-2′-[18F]fluoroethyl)amino]-2H-1,3,2-oxaphosphorinane-2-oxide (18F-fluorocyclophosphamide), a potential tracer for breast tumor prognostic imaging with PET. J Label Compd Radiopharm 48:635–643
Kesner AL, Townsend A, Hsueh WA, et al. (2004) A non-invasive assay to predict chemotherapy response to 5-fluorouracil using [18F]-5-FU and PET. J Nucl Med 45:331P, (Abstract)
Hsueh WA, Gangloff A, Kesner AL, et al. (2004) Predicting chemotherapy response to paclitaxel using [18F]-fluoropaclitaxel and PET. J Nucl Med 45:38P, (Abstract)
Hsueh WA, Gangloff A, Kesner AL, Pio BS, Beryt MM, Pegram M, Czernin J, Phelps ME, Silverman DH (2005) Added value of [18F]-fluoropaclitaxel beyond tumor size as a predictor for paclitaxel resistance. J Nucl Med 46(S2):398P, (Abstract)
Hsueh WA, Gangloff A, Kesner AL, Beryt MM, Pegram M, Czernin J, Phelps ME, Silverman DH (2005) FDG and FLT microPET predictors of tumor response and survival following chemotherapy. J Nucl Med 46(S2):155P, (Abstract)
Gangloff A, Hsueh WA, Kesner AL, Kiesewetter DO, Pio BS, Czernin J, Pegram MD, Beryt M, Phelps ME, Silverman DH (2005) Estimation of paclitaxel biodistribution and uptake in human-derived xenografts in vivo with [18F]-fluoropaclitaxel. J Nucl Med 46(11):1866–1871
Acknowledgements
The authors would like to thank the UCLA Cyclotron staff for PET tracer production, and the UCLA Ahmanson Biological Imaging Center Technologists for their support in PET acquisition.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pio, B.S., Park, C.K., Pietras, R. et al. Usefulness of 3′-[F-18]Fluoro-3′-deoxythymidine with Positron Emission Tomography in Predicting Breast Cancer Response to Therapy. Mol Imaging Biol 8, 36–42 (2006). https://doi.org/10.1007/s11307-005-0029-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-005-0029-9