Abstract
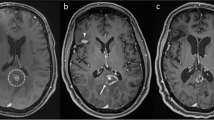
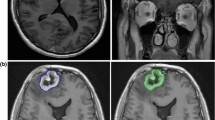
Distinguishing radiation necrosis (RN) from tumor recurrence after stereotactic radiosurgery (SRS) for brain metastases is challenging. This study assesses the sensitivity (SN) and specificity (SP) of an MRI-based parameter, the “lesion quotient” (LQ), in characterizing tumor progression from RN. Records of patients treated with SRS for brain metastases between 01/01/1999 and 12/31/2009 and with histopathologic analysis of a subsequent contrast enhancing enlarging lesion at the treated site at a single institution were examined. The LQ, the ratio of maximal nodular cross sectional area on T2-weighted imaging to the corresponding maximal cross sectional area of T1-contrast enhancement, was calculated by a neuroradiologist blinded to the histopathological outcome. Cutoffs of <0.3, 0.3–0.6, and >0.6 have been previously suggested to have correlated with RN, mixed findings and tumor recurrence, respectively. These cutoff values were evaluated for SN, SP, positive predictive value (PPV) and negative predictive value (NPV). Logistic regression analysis evaluated for associated clinical factors. For the 51 patients evaluated, the SN, SP, PPV and NPV for identifying RN (LQ < 0.3) were 8, 91, 25 and 73 %, respectively. For the combination of recurrent tumor and RN (LQ 0.3–0.6) the SN, SP, PPV and NPV were 0, 64, 0 and 83 %. The SN, SP, PPV and NPV of the LQ for recurrent tumor (LQ > 0.6) were 59, 41, 62 and 39 %, respectively. Standard MRI techniques do not reliably discriminate between tumor progression and RN after treatment with SRS for brain metastases. Additional imaging modalities are warranted to aid in distinguishing between these diagnoses.


Similar content being viewed by others
References
Aoyama H, Shirato H, Tago M, Nakagawa K, Toyoda T, Hatano K et al (2006) Stereotactic radiosurgery plus whole-brain radiation therapy vs stereotactic radiosurgery alone for treatment of brain metastases: a randomized controlled trial. JAMA 295:2483–2491
Truong MT, St Clair EG, Donahue BR, Rush SC, Miller DC, Formenti SC et al (2006) Results of surgical resection for progression of brain metastases previously treated by gamma knife radiosurgery. Neurosurgery 59:86–97; discussion 86–97
Blonigen BJ, Steinmetz RD, Levin L, Lamba MA, Warnick RE, Breneman JC (2010) Irradiated volume as a predictor of brain radionecrosis after linear accelerator stereotactic radiosurgery. Int J Radiat Oncol Biol Phys 77:996–1001
Kano H, Kondziolka D, Lobato-Polo J, Zorro O, Flickinger JC, Lunsford LD (2010) T1/T2 matching to differentiate tumor growth from radiation effects after stereotactic radiosurgery. Neurosurgery 66:486–491; discussion 491–482
Chao ST, Suh JH, Raja S, Lee SY, Barnett G (2001) The sensitivity and specificity of FDG PET in distinguishing recurrent brain tumor from radionecrosis in patients treated with stereotactic radiosurgery. Int J Cancer 96:191–197
Huang CF, Chiou SY, Wu MF, Tu HT, Liu WS, Chuang JC (2010) Apparent diffusion coefficients for evaluation of the response of brain tumors treated by Gamma Knife surgery. J Neurosurg 113(Suppl):97–104
Jain R, Narang J, Sundgren PM, Hearshen D, Saksena S, Rock JP et al (2010) Treatment induced necrosis versus recurrent/progressing brain tumor: going beyond the boundaries of conventional morphologic imaging. J Neurooncol 100:17–29
Mitsuya K, Nakasu Y, Horiguchi S, Harada H, Nishimura T, Bando E et al (2010) Perfusion weighted magnetic resonance imaging to distinguish the recurrence of metastatic brain tumors from radiation necrosis after stereotactic radiosurgery. J Neurooncol 99:81–88
Serizawa T, Saeki N, Higuchi Y, Ono J, Matsuda S, Sato M et al (2005) Diagnostic value of thallium-201 chloride single-photon emission computerized tomography in differentiating tumor recurrence from radiation injury after gamma knife surgery for metastatic brain tumors. J Neurosurg 102(Suppl):266–271
Kimura T, Sako K, Tohyama Y, Aizawa S, Yoshida H, Aburano T, et al. (2003) Diagnosis and treatment of progressive space-occupying radiation necrosis following stereotactic radiosurgery for brain metastasis: value of proton magnetic resonance spectroscopy. Acta Neurochir (Wien) 145:557–564; discussion 564
Hoefnagels FW, Lagerwaard FJ, Sanchez E, Haasbeek CJ, Knol DL, Slotman BJ et al (2009) Radiological progression of cerebral metastases after radiosurgery: assessment of perfusion MRI for differentiating between necrosis and recurrence. J Neurol 256:878–887
Dequesada IM, Quisling RG, Yachnis A, Friedman WA (2008) Can standard magnetic resonance imaging reliably distinguish recurrent tumor from radiation necrosis after radiosurgery for brain metastases? A radiographic-pathological study. Neurosurgery 63:898–903; discussion 904
Shaw E, Scott C, Souhami L, Dinapoli R, Kline R, Loeffler J et al (2000) Single dose radiosurgical treatment of recurrent previously irradiated primary brain tumors and brain metastases: final report of RTOG protocol 90-05. Int J Radiat Oncol Biol Phys 47:291–298
Chin LS, Ma L, DiBiase S (2001) Radiation necrosis following gamma knife surgery: a case-controlled comparison of treatment parameters and long-term clinical follow up. J Neurosurg 94:899–904
Suh JH (2010) Stereotactic radiosurgery for the management of brain metastases. N Engl J Med 362:1119–1127
Levin VA, Bidaut L, Hou P, Kumar AJ, Wefel JS, Bekele BN et al (2011) Randomized double-blind placebo-controlled trial of bevacizumab therapy for radiation necrosis of the central nervous system. Int J Radiat Oncol Biol Phys 79:1487–1495
Matuschek C, Bolke E, Nawatny J, Hoffmann TK, Peiper M, Orth K et al (2011) Bevacizumab as a treatment option for radiation-induced cerebral necrosis. Strahlenther Onkol 187:135–139
Perez-Espejo MA, Garcia-Fernandez R, Tobarra-Gonzalez BM, Palma-Copete JD, Gonzalez-Lopez A, De la Fuente-Munoz I et al (2009) Usefulness of hyperbaric oxygen in the treatment of radionecrosis and symptomatic brain edema after LINAC radiosurgery. Neurocirugia (Astur) 20:449–453
Vecil GG, Suki D, Maldaun MV, Lang FF, Sawaya R (2005) Resection of brain metastases previously treated with stereotactic radiosurgery. J Neurosurg 102:209–215
Yamanaka K, Iwai Y, Yasui T, Nakajima H, Komiyama M, Nishikawa M et al (1999) Gamma Knife radiosurgery for metastatic brain tumor: the usefulness of repeated Gamma Knife radiosurgery for recurrent cases. Stereotact Funct Neurosurg 72(Suppl 1):73–80
Dooms GC, Hecht S, Brant-Zawadzki M, Berthiaume Y, Norman D, Newton TH (1986) Brain radiation lesions: MR imaging. Radiology 158:149–155
Essig M, Waschkies M, Wenz F, Debus J, Hentrich HR, Knopp MV (2003) Assessment of brain metastases with dynamic susceptibility-weighted contrast-enhanced MR imaging: initial results. Radiology 228:193–199
Barajas RF, Chang JS, Sneed PK, Segal MR, McDermott MW, Cha S (2009) Distinguishing recurrent intra-axial metastatic tumor from radiation necrosis following gamma knife radiosurgery using dynamic susceptibility-weighted contrast-enhanced perfusion MR imaging. AJNR Am J Neuroradiol 30:367–372
Narang J, Jain R, Arbab AS, Mikkelsen T, Scarpace L, Rosenblum ML et al (2011) Differentiating treatment-induced necrosis from recurrent/progressive brain tumor using nonmodel-based semiquantitative indices derived from dynamic contrast-enhanced T1-weighted MR perfusion. Neuro-oncology 13:1037–1046
Acknowledgments
Thank you to Dr. David Chooljian for his review of the manuscript. Dr. Tievsky, Research grant from Bayer; Dr. Suh, Abbott Oncology; Dr. Vogelbaum, Honoraria from Merck.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Stockham, A.L., Tievsky, A.L., Koyfman, S.A. et al. Conventional MRI does not reliably distinguish radiation necrosis from tumor recurrence after stereotactic radiosurgery. J Neurooncol 109, 149–158 (2012). https://doi.org/10.1007/s11060-012-0881-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-012-0881-9