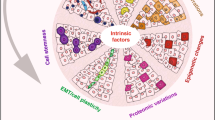
Abstract
Generation of intratumoral phenotypic and genetic heterogeneity has been attributed to clonal evolution and cancer stem cells that together give rise to a tumor with complex ecosystems. Each ecosystem contains various tumor cell subpopulations and stromal entities, which, depending upon their composition, can influence survival, therapy responses, and global growth of the tumor. Despite recent advances in breast cancer management, the disease has not been completely eradicated as tumors recur despite initial response to treatment. In this review, using data from clinically relevant breast cancer models, we show that the fates of tumor stem cells/progenitor cells in the individual tumor ecosystems comprising a tumor are predetermined to follow a limited (unipotent) and/or unlimited (multipotent) path of differentiation which create conditions for active generation and maintenance of heterogeneity. The resultant dynamic systems respond differently to treatments, thus disrupting the delicate stability maintained in the heterogeneous tumor. This raises the question whether it is better then to preserve stability by preventing takeover by otherwise dormant ecosystems in the tumor following therapy. The ultimate strategy for personalized therapy would require serial assessments of the patient’s tumor for biomarker validation during the entire course of treatment that is combined with their three-dimensional mapping to the tumor architecture and landscape.



Similar content being viewed by others
References
Fidler, I. J. (1978). Tumor heterogeneity and the biology of cancer invasion and metastasis. Cancer Research, 38, 2651–2660.
Heppner, G. H., & Miller, B. E. (1983). Tumor heterogeneity: biological implications and therapeutic consequences. Cancer Metastasis Reviews, 2, 5–23.
Dexter, D. L., Kowalski, H. M., Blazar, B. A., Fligiel, Z., Vogel, R., & Heppner, G. H. (1978). Heterogeneity of tumor cells from a single mouse mammary tumor. Cancer Research, 38, 3174–3181.
Marusyk, A., Almendro, V., & Polyak, K. (2012). Intra-tumour heterogeneity: a looking glass for cancer? Nature Reviews Cancer, 12, 323–334.
Nowell, P. C. (1976). The clonal evolution of tumor cell populations. Science, 194, 23–28.
Heppner, G. H., & Miller, F. R. (1998). The cellular basis of tumor progression. International Reviews in Cytology, 177, 1–56.
Merlo, L. M., Pepper, J. W., Reid, B. J., & Maley, C. C. (2006). Cancer as an evolutionary and ecological process. Nature Reviews Cancer, 6, 924–935.
Wolman, S. R., & Heppner, G. H. (1992). Genetic heterogeneity in breast cancer. Journal of National Cancer Institute, 84, 469–470.
Marusyk, A., & Polyak, K. (2010). Tumor heterogeneity: causes and consequences. Biochimica et Biophysica Acta, 1805, 105–117.
Janiszewska, M., & Polyak, K. (2015). Clonal evolution in cancer: a tale of twisted twines. Cell Stem Cell, 16, 11–12.
Bapat, S. A. (2007). Evolution of cancer stem cells. Seminars in Cancer Biology, 17, 204–213.
Bjerkvig, R., Tysnes, B. B., Aboody, K. S., Najbauer, J., & Terzis, A. J. (2005). Opinion: the origin of the cancer stem cell: current controversies and new insights. Nature Reviews Cancer, 5, 899–904.
Reya, T., Morrison, S. J., Clarke, M. F., & Weissman, I. L. (2001). Stem cells, cancer, and cancer stem cells. Nature, 414, 105–111.
Kreso, A., & Dick, J. E. (2014). Evolution of the cancer stem cell model. Cell Stem Cell, 14, 275–291.
Marjanovic, N. D., Weinberg, R. A., & Chaffer, C. L. (2013). Cell plasticity and heterogeneity in cancer. Clinical Chemistry, 59, 168–179.
Elshamy, W. M., & Duhe, R. J. (2013). Overview: cellular plasticity, cancer stem cells and metastasis. Cancer Letters, 341, 2–8.
Rhiannon, F., & Richard, C. (2012). The complex nature of breast cancer stem-like cells: heterogeneity and plasticity. Journal of Stem Cells Research and Therapy. doi:10.4172/2157-7633.S7-009.
Miller, F. R., Soule, H. D., Tait, L., Pauley, R. J., Wolman, S. R., Dawson, P. J., & Heppner, G. H. (1993). Xenograft model of progressive human proliferative breast disease. Journal of National Cancer Institute, 85, 1725–1732.
Miller, F. R. (2000). Xenograft models of premalignant breast disease. Journal of Mammary Gland Biology and Neoplasia, 5, 379–391.
Shekhar, M. P., Nangia-Makker, P., Wolman, S. R., Tait, L., Heppner, G. H., & Visscher, D. W. (1998). Direct action of estrogen on sequence of progression of human preneoplastic breast disease. American Journal of Pathology, 152, 1129–1132.
Shekhar, P. V., Chen, M. L., Werdell, J., Heppner, G. H., Miller, F. R., & Christman, J. K. (1998). Transcriptional activation of functional endogenous estrogen receptor gene expression in MCF10AT cells: a model for early breast cancer. International Journal of Oncology, 13, 907–915.
Visscher, D. W., Nanjia-Makker, P., Heppner, G., & Shekhar, P. V. (2001). Tamoxifen suppresses histologic progression to atypia and DCIS in MCFIOAT xenografts, a model of early human breast cancer. Breast Cancer Research & Treatment, 65, 41–47.
Al-Hajj, M., Wicha, M. S., Benito-Hernandez, A., Morrison, S. J., & Clarke, M. F. (2003). Prospective identification of tumorigenic breast cancer cells. Proceedings of National Academy of Sciences U S A, 100, 3983–3988.
Shekhar, M. P., & Tait, L. Breast cancer stem cell paradigm. In D. W. Parsons (Ed.), Stem cells and cancer (pp. 47–64). New York: Nova Science.
Liu, Y., Nenutil, R., Appleyard, M. V., Murray, K., Boylan, M., Thompson, A. M., & Coates, P. J. (2014). Lack of correlation of stem cell markers in breast cancer stem cells. British Journal of Cancer, 110, 2063–2071.
Miller, F. R., Santner, S. J., Tait, L., & Dawson, P. J. (2000). MCF10DCIS.com xenograft model of human comedo ductal carcinoma in situ. Journal of National Cancer Institute, 92, 1185–1186.
Santner, S. J., Dawson, P. J., Tait, L., Soule, H. D., Eliason, J., Mohamed, A. N., Wolman, S. R., Heppner, G. H., & Miller, F. R. (2001). Malignant MCF10CA1 cell lines derived from premalignant human breast epithelial MCF10AT cells. Breast Cancer Research & Treatment, 65, 101–110.
Guo, W., Keckesova, Z., Donaher, J. L., Shibue, T., Tischler, V., Reinhardt, F., Itzkovitz, S., Noske, A., Zürrer-Härdi, U., Bell, G., Tam, W. L., Mani, S. A., van Oudenaarden, A., & Weinberg, R. A. (2012). Slug and Sox9 cooperatively determine the mammary stem cell state. Cell, 148, 1015–1028.
Shekhar, M. P., Kato, I., Nangia-Makker, P., & Tait, L. (2013). Comedo-DCIS is a precursor lesion for basal-like breast carcinoma: identification of a novel p63/Her2/neu expressing subgroup. Oncotarget, 4, 231–241.
Hannemann, J., Velds, A., Halfwerk, J. B., Kreike, B., Peterse, J. L., & van de Vijver, M. J. (2006). Classification of ductal carcinoma in situ by gene expression profiling. Breast Cancer Research, 8, R61.
Yu, K., Lee, C. H., Tan, P. H., & Tan, P. (2004). Conservation of breast cancer molecular subtypes and transcriptional patterns of tumor progression across distinct ethnic populations. Clinical Cancer Research, 10, 5508–5517.
Bryan, B. B., Schnitt, S. J., & Collins, L. C. (2006). Ductal carcinoma in situ with basal-like phenotype: a possible precursor to invasive basal-like breast cancer. Modern Pathology, 19, 617–621.
Dabbs, D. J., Chivukula, M., Carter, G., & Bhargava, R. (2006). Basal phenotype of ductal carcinoma in situ: recognition and immunohistologic profile. Modern Pathology, 19, 1506–1511.
Livasy, C. A., Perou, C. M., Karaca, G., Cowan, D. W., Maia, D., Jackson, S., Tse, C. K., Nyante, S., & Millikan, R. C. (2007). Identification of a basal-like subtype of breast ductal carcinoma in situ. Human Pathology, 38, 197–204.
Paredes, J., Lopes, N., Milanezi, F., & Schmitt, F. C. (2007). P-cadherin and cytokeratin 5: useful adjunct markers to distinguish basal-like ductal carcinomas in situ. Virchows Archives, 450, 73–80.
Tang, P., Wang, X., Schiffhauer, L., Wang, J., Bourne, P., Yang, Q., Quinn, A., & Hajdu, S. I. (2006). Relationship between nuclear grade of ductal carcinoma in situ and cell origin markers. Annals of Clinical Laboratory Science, 36, 16–22.
Bertucci, F., Finetti, P., & Birnbaum, D. (2012). Basal breast cancer: a complex and deadly molecular subtype. Current Molecular Medicine, 12, 96–110.
Beachy, P. A., Karhadkar, S. S., & Berman, D. M. (2004). Tissue repair and stem cell renewal in carcinogenesis. Nature, 432, 324–331.
Hanley, C. J., Noble, F., Ward, M., Bullock, M., Drifka, C., Mellone, M., Manousopoulou, A., Johnston, H. E., Hayden, A., Thirdborough, S., Liu, Y., Smith, D. M., Mellows, T., Kao, W. J., Garbis, S. D., Mirnezami, A., Underwood, T. J., Eliceiri, K. W., & Thomas, G. J. (2016). A subset of myofibroblastic cancer-associated fibroblasts regulate collagen fiber elongation, which is prognostic in multiple cancers. Oncotarget, 7, 6159–6174.
He, K., Lv, W., Zheng, D., Cheng, F., Zhou, T., Ye, S., Ban, Q., Ying, Q., Huang, B., Chen, L., Wu, G., & Liu, D. (2015). The stromal genome heterogeneity between breast and prostate tumors revealed by a comparative transcriptomic analysis. Oncotarget, 6, 8687–8697.
Junk, D. J., Cipriano, R., Bryson, B. L., Gilmore, H. L., & Jackson, M. W. (2013). Tumor microenvironmental signaling elicits epithelial-mesenchymal plasticity through cooperation with transforming genetic events. Neoplasia, 15, 1100–1109.
Natrajan, R., Sailem, H., Mardakheh, F. K., Arias Garcia, M., Tape, C. J., Dowsett, M., Bakal, C., & Yuan, Y. (2016). Microenvironmental heterogeneity parallels breast cancer progression: a histology-genomic integration analysis. PLoS Medicine, 13, e1001961.
Roman-Perez, E., Casbas-Hernandez, P., Pirone, J. R., Rein, J., Carey, L. A., Lubet, R. A., Mani, S. A., Amos, K. D., & Troester, M. A. (2012). Gene expression in extratumoral microenvironment predicts clinical outcome in breast cancer patients. Breast Cancer Research, 14, R51.
Sugimoto, H., Mundel, T. M., Kieran, M. W., & Kalluri, R. (2006). Identification of fibroblast heterogeneity in the tumor microenvironment. Cancer Biology & Therapy, 5, 1640–1646.
Shekhar, M. P., Werdell, J., Santner, S. J., Pauley, R. J., & Tait, L. (2001). Breast stroma plays a dominant regulatory role in breast epithelial growth and differentiation: implications for tumor development and progression. Cancer Research, 61, 1320–1326.
Shekhar, M. P., Santner, S., Carolin, K. A., & Tait, L. (2007). Direct involvement of breast tumor fibroblasts in the modulation of tamoxifen sensitivity. American Journal Pathology, 170, 1546–1560.
Harahap, W.A., Daan Khambri, R., Haryono, S., & Nindrea, R.D. (2017). Outcomes of trastuzumab therapy for 6 and 12 months in Indonesian national health insurance system clients with operable HER2-positive breast cancer. Asian Pacific Journal of Cancer Prevention, 18, 1151–1156.
Cheng, Y. C., Shi, Y., Zhang, M. J., Brazauskas, R., Hemmer, M. T., Bishop, M. R., Nieto, Y., Stadtmauer, E., Ayash, L., Gale, R. P., Lazarus, H., Holmberg, L., Lill, M., Olsson, R. F., Wirk, B. M., Arora, M., Hari, P., & Ueno, N. (2017). Long-term outcome of inflammatory breast cancer compared to non-inflammatory breast cancer in the setting of high-dose chemotherapy with autologous hematopoietic cell transplantation. Journal of Cancer, 8, 1009–1017.
Nasir, A., Holzer, T. R., Chen, M., Man, M. Z., & Schade, A. E. (2017). Differential expression of VEGFR2 protein in HER2 positive primary human breast cancer: potential relevance to anti-angiogenic therapies. Cancer Cell International, 17, 56.
Rier, H. N., Levin, M. D., van Rosmalen, J., Bos, M., Drooger, J. C., de Jong, P., Portielje, J.E.A., Elsten, E.M.P., Ten Tije, A.J., Sleijfer, S., & Jager, A. (2017). First-line palliative HER2-targeted therapy in HER2-positive metastatic breast cancer is less effective after previous adjuvant trastuzumab-based therapy. Oncologist. doi:10.1634/the oncologist.2016-0448.
Yu, X., Wang, L., Shen, Y., Wang, C., Zhang, Y., Meng, Y., Yang, Y., Liang, B., Zhou, B., Wang, H., Wei, H., Lei, C., Hu, S., & Li, B. (2017). Targeting EGFR/HER2 heterodimerization with a novel anti-HER2 domain II/III antibody. Molecular Immunology, 87, 300–307.
Drakaki, A., & Hurvitz, S.A. (2015). Her2-positive breast cancer: Update on new and emerging agents. The American Journal of Hematology/Oncology, 11, 17-23.
Dickler, M. N., Tolaney, S., Rugo, H. S., Cortes, J., Dieras, V., Patt, D. A., Wildiers, H., Hudis, C. A., O, Shaughnessy, J. A., Zamora, E., Yardley, D., Frenzel, M., Koustenis, A. G., & Baselga, J. (2017). MONARCH 1, a phase 2 study of abemaciclib, a CDK4 and CDK6 inhibitor, as a single agent, in patients with refractory HR+/HER2− metastatic breast cancer. Clinical Cancer Research. doi:10.1158/1078-0432.CCR-17-0754 Epub ahead of print.
Dean, L. (2012). Pertuzumab therapy and ERBB2 (HER2) genotype. In V. Pratt, H. McLeod, L. Dean, A. Malheiro, & W. Rubinstein (Eds.), Medical genetics summaries. Bethesda: National Center for Biotechnology Information.
Dean, L. (2012). Trastuzumab (herceptin) therapy and ERBB2 (HER2) genotype. In V. Pratt, H. McLeod, L. Dean, A. Malheiro, & W. Rubinstein (Eds.), Medical genetics summaries. Bethesda: National Center for Biotechnology Information.
Dean, L. (2012). Tamoxifen therapy and CYP2D6 genotype. In V. Pratt, H. McLeod, L. Dean, A. Malheiro, & W. Rubinstein (Eds.), Medical genetics summaries. Bethesda: National Center for Biotechnology Information.
Gu, G., Dustin, D., & Fuqua, S. A. (2016). Targeted therapy for breast cancer and molecular mechanisms of resistance to treatment. Current Opinion in Pharmacology, 31, 97–103.
Yap, T. A., Omlin, A., & de Bono, J. S. (2013). Development of therapeutic combinations targeting major cancer signaling pathways. Journal of Clinical Oncology, 31, 1592–1605.
Tryfonidis, K., Senkus, E., Cardoso, M. J., & Cardoso, F. (2015). Management of locally advanced breast cancer—perspectives and future directions. Nature Reviews Clinical Oncology, 12, 147–162.
Cristofanilli, M., Turner, N. C., Bondarenko, I., Ro, J., Im, S. A., Masuda, N., Colleoni, M., DeMichele, A., Loi, S., Verma, S., Iwata, H., Harbeck, N., Zhang, K., Theall, K. P., Jiang, Y., Bartlett, C. H., Koehler, M., & Slamon, D. (2016). Fulvestrant plus palbociclib versus fulvestrant plus placebo for treatment of hormone-receptor-positive, HER2-negative metastatic breast cancer that progressed on previous endocrine therapy (PALOMA-3): final analysis of the multicentre, double-blind, phase 3 randomised controlled trial. Lancet Oncology, 17, 425–439.
Swain, S. M., Baselga, J., Kim, S. B., Ro, J., Semiglazov, V., Campone, M., Ciruelos, E., Ferrero, J. M., Schneeweiss, A., Heeson, S., Clark, E., Ross, G., Benyunes, M. C., Cortés, J., & CLEOPATRA Study Group. (2015). Pertuzumab, trastuzumab, and docetaxel in HER2-positive metastatic breast cancer. New England Journal of Medicine, 372, 724–734.
Ates, O., Sunar, V., Aslan, A., Karatas, F., Sahin, S., & Altundag, K. (2017). The short-term safety of adjuvant paclitaxel plus trastuzumab—a single centre experience. J Balkan Union of Oncology, 22, 320–324.
Liu, Z., He, K., Ma, Q., Yu, Q., Liu, C., Ndege, I., Wang, X., & Yu, Z. (2017). Autophagy inhibitor facilitates gefitinib sensitivity in vitro and in vivo by activating mitochondrial apoptosis in triple negative breast cancer. PLoS One, 12(5), e0177694.
Di Nicolantonio, F., Mercer, S. J., Knight, L. A., Gabriel, F. G., Whitehouse, P. A., Sharma, S., Fernando, A., Glaysher, S., Di Palma, S., Johnson, P., Somers, S. S., Toh, S., Higgins, B., Lamont, A., Gulliford, T., Hurren, J., Yiangou, C., & Cree, I. A. (2005). Cancer cell adaptation to chemotherapy. BMC Cancer, 5, 78.
Tan, S. H., Sapari, N. S., Miao, H., Hartman, M., Loh, M., Chng, W. J., Iau, P., Buhari, S. A., Soong, R., & Lee, S. C. (2015). High-throughput mutation profiling changes before and 3 weeks after chemotherapy in newly diagnosed breast cancer patients. PLoS One, 10, e0142466.
Murphy, C., & Dickler, M. (2016). Endocrine resistance in hormone responsive breast cancer: mechanisms and therapeutic strategies. Endocrine Related Cancer, 23, R337-352.
Jeselsohn, R., & Brown, M. (2016). How drug resistance takes shape. eLife, 5, e14973. doi:10.7554/eLife.14973.
Ter Brugge, P., Kristel, P., van der Burg, E., Boon, U., de Maaker, M., Lips, E., Mulder, L., de Ruiter, J., Moutinho, C., Gevensleben, H., Marangoni, E., Majewski, I., Józwiak, K., Kloosterman, W., van Roosmalen, M., Duran, K., Hogervorst, F., Turner, N., Esteller, M., Cuppen, E., Wesseling, J., & Jonkers, J. (2016). Mechanisms of therapy resistance in patient-derived xenograft models of BRCA1-deficient breast cancer. Journal of National Cancer Institute, 108, djw148. doi:10.1093/jnci/djw148.
Edwards, S. L., Brough, R., Lord, C. J., Natrajan, R., Vatcheva, R., Levine, D. A., Boyd, J., Reis-Filho, J. S., & Ashworth, A. (2008). Resistance to therapy caused by intragenic deletion in BRCA2. Nature, 451, 1111–1115.
Sakai, W., Swisher, E. M., Karlan, B. Y., Agarwal, M. K., Higgins, J., Friedman, C., Villegas, E., Jacquemont, C., Farrugia, D. J., Couch, F. J., Urban, N., & Taniguchi, T. (2008). Secondary mutations as a mechanism of cisplatin resistance in BRCA2-mutated cancers. Nature, 451, 1116–1120.
Creighton, C. J., Li, X., Landis, M., Dixon, J. M., Neumeister, V. M., Sjolund, A., Rimm, D. L., Wong, H., Rodriguez, A., Herschkowitz, J. I., Fan, C., Zhang, X., He, X., Pavlick, A., Gutierrez, M. C., Renshaw, L., Larionov, A. A., Faratian, D., Hilsenbeck, S. G., Perou, C. M., Lewis, M. T., Rosen, J. M., & Chang, J. C. (2009). Residual breast cancers after conventional therapy display mesenchymal as well as tumor-initiating features. Proceedings of National Academy of Sciences U S A, 106, 13820–13825.
Leder, K., Holland, E. C., & Michor, F. (2010). The therapeutic implications of plasticity of the cancer stem cell phenotype. PLoS One, 5, e14366.
Li, X., Lewis, M. T., Huang, J., Gutierrez, C., Osborne, C. K., Wu, M. F., Hilsenbeck, S. G., Pavlick, A., Zhang, X., Chamness, G. C., Wong, H., Rosen, J., & Chang, J. C. (2008). Intrinsic resistance of tumorigenic breast cancer cells to chemotherapy. Journal of National Cancer Institute, 100, 672–679.
Iwasa, Y., Nowak, M. A., & Michor, F. (2006). Evolution of resistance during clonal expansion. Genetics, 174, 2557–2566.
Zhou, X., Zhang, J., Yun, H., Shi, R., Wang, Y., Wang, W., Lagercrantz, S. B., & Mu, K. (2015). Alterations of biomarker profiles after neoadjuvant chemotherapy in breast cancer: tumor heterogeneity should be taken into consideration. Oncotarget, 6, 36894–36902.
Miller, B. E., Miller, F. R., Wilburn, D., & Heppner, G. H. (1988). Dominance of a tumor subpopulation line in mixed heterogeneous mouse mammary tumors. Cancer Research, 48, 5747–5753.
Crespi, B., Foster, K., & Ubeda, F. (2014). First principles of Hamiltonian medicine. Philosophical Transactions of the Royal Society of London Series B Biological Sciences, 369, 20130366.
Gerard, B., Tait, L., Nangia-Makker, P., & Shekhar, M. P. (2011). Rad6B acts downstream of Wnt signaling to stabilize beta-catenin: implications for a novel Wnt/beta-catenin target. Journal of Molecular Signaling, 6, 6.
Shekhar, M. P., Gerard, B., Pauley, R. J., Williams, B. O., & Tait, L. (2008). Rad6B is a positive regulator of beta-catenin stabilization. Cancer Research, 68, 1741–1750.
Marusyk, A., Tabassum, D. P., Altrock, P. M., Almendro, V., Michor, F., & Polyak, K. (2014). Non-cell-autonomous driving of tumour growth supports sub-clonal heterogeneity. Nature, 514, 54–58.
Parikh, J., Selmi, M., Charles-Edwards, G., Glendenning, J., Ganeshan, B., Verma, H., Mansi, J., Harries, M., Tutt, A., & Goh, V. (2014). Changes in primary breast cancer heterogeneity may augment midtreatment MR imaging assessment of response to neoadjuvant chemotherapy. Radiology, 272, 100–112.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Work related to the analysis of histologic origins and stromal contributions to therapy response was funded by grants DAMD-17-02-1-0618 and W81XWH-09-1-0608 from the Department of Defense. BH was supported by T32-CA009531 and a fellowship from the DeRoy Testamentary Foundation.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Haynes, B., Sarma, A., Nangia-Makker, P. et al. Breast cancer complexity: implications of intratumoral heterogeneity in clinical management. Cancer Metastasis Rev 36, 547–555 (2017). https://doi.org/10.1007/s10555-017-9684-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10555-017-9684-y