Abstract
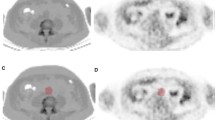
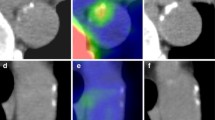
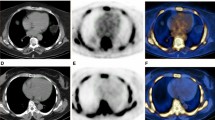
To compare regional vascular distribution and biological determinants of visible calcium load, as assessed by computed tomography, as well as of molecular calcium deposition as assessed by 18F–NaF positron emission tomography. Eighty oncologic patients undergoing 18F–NaF PET/CT scan were included in the study. Cardiovascular-risk stratification was performed according to a simplified version of the Framingham model [including age, diabetes, smoking, systolic blood pressure and body mass index (BMI)]. Arterial 18F–NaF uptake was measured by drawing regions of interest comprising the arteries on each slice of the transaxial PET/CT and normalized to blood 18F–NaF activity to obtain the arterial target-to-background ratio (TBR). The degree of arterial calcification (AC) was measured using a software program providing Agatston-like scores. Differences in mean values and regression analysis were tested. Predictors of AC and TBR were evaluated by univariate and multivariate analysis. p value of 0.05 was considered statistically significant. No correlation was documented between regional calcium load and regional TBR in any of the studied arterial segments. Visible calcium deposition was found to be dependent upon age while it was not influenced by all the remaining determinants of cardiovascular risk. By contrast, 18F–NaF uptake was significantly correlated with all descriptors of cardiovascular risk, with the exception of BMI. Vascular 18F–NaF uptake displays a different regional distribution, as well as different biological predictors, when compared to macroscopic AC. The tight dependency of tracer retention upon ongoing biological determinants of vascular damage suggests that this tool might provide an unexplored window on plaque pathophysiology.





Similar content being viewed by others
References
Hartiala O, Magnussen CG, Kajander S, Knuuti J, Ukkonen H, Saraste A, Rinta-Kiikka I, Kainulainen S, Kähönen M, Hutri-Kähönen N, Laitinen T, Lehtimäki T, Viikari JS, Hartiala J, Juonala M, Raitakari OT (2012) Adolescence risk factors are predictive of coronary artery calcification at middle age: the cardiovascular risk in young Finns study. J Am Coll Cardiol 60:1364–1370
Libby P (2002) Inflammation in atherosclerosis. Nature 420:868–874
Hayden MR, Tyagi SC, Kolb L (2005) Vascular ossification–calcification in metabolic syndrome, type 2 diabetes mellitus, chronic kidney disease, and calciphylaxis-calcific uremic arteriolopathy: the emerging role of sodium thiosulfate. Cardiovasc Diabetol 4:4
Demer LL, Tintut Y (2008) Vascular calcification: pathobiology of multifaceted disease. Circulation 117:2938–2948
Schoppet M, Shroff RC, Hofbauer LC, Shanahan CM (2008) Exploring the biology of vascular calcification in chronic kidney disease: what’s circulating? Kidney Int 73:384–390
Jeziorska M, McCollum C, Wooley DE (1998) Observations on bone formation and remodeling in advanced atherosclerotic lesions of human carotid arteries. Virchows Arch 433:559–565
Schmermund A, Baumgart D, Möhlenkamp S, Kriener P, Pump H, Grönemeyer D, Seibel R, Erbel R (2001) Natural history and topographic pattern of progression of coronary calcification in symptomatic patients: an electron-beam CT study. Arterioscler Thromb Vasc Biol 21:421–426
Bild DE, Folsom AR, Lowe LP, Sidney S, Kiefe C, Westfall AO, Zheng ZJ, Rumberger J (2001) Prevalence and correlates of coronary calcification in black and white young adults: the Coronary Artery Risk Development in Young Adults (CARDIA) study. Arterioscler Thromb Vasc Biol 21:852–857
Pohle K, Mäffert R, Ropers D, Moshage W, Stilianakis N, Daniel WG, Achenbach S (2001) Progression of aortic valve calcification: association with coronary atherosclerosis and cardiovascular risk factors. Circulation 104:1927–1932
Schmidt HH, Hill S, Makariou EV, Feuerstein IM, Dugi KA, Hoeg JM (1996) Relation of cholesterol-year score to severity of calcific atherosclerosis and tissue deposition in homozygous familial hypercholesterolemia. Am J Cardiol 77:575–580
Moura LM, Ramos SF, Zamorano JL, Barros IM, Azevedo LF, Rocha-Gonçalves F, Rajamannan NM (2007) Rosuvastatin affecting aortic valve endothelium to slow the progression of aortic stenosis. J Am Coll Cardiol 49:554–561
Cowell SJ, Newby DE, Prescott RJ et al (2005) A randomized trial of intensive lipid-lowering therapy in calcific aortic stenosis. N Engl J Med 352:2389–2397
Cowell SJ, Newby DE, Prescott RJ, Bloomfield P, Reid J, Northridge DB, Boon NA, Scottish Aortic Stenosis and Lipid Lowering Trial, Impact on Regression (SALTIRE) Investigators (2002) Computed tomography and magnetic resonance imaging for noninvasive coronary angiography and plaque imaging: current and potential future concepts. Circulation 106:2026–2034
Derlin T, Wisotzki C, Richter U, Apostolova I, Bannas P, Weber C, Mester J, Klutmann S (2011) In vivo imaging of mineral deposition in carotid plaque using 18F-sodium fluoride PET/CT: correlation with atherogenic risk factors. J Nucl Med 52:362–368
Derlin T, Richter U, Bannas P, Begemann P, Buchert R, Mester J, Klutmann S (2010) Feasibility of 18F-sodium fluoride PET/CT for imaging of atherosclerotic plaque. J Nucl Med 51:862–865
Derlin T, Tóth Z, Papp L, Wisotzki C, Apostolova I, Habermann CR, Mester J, Klutmann S (2011) Correlation of inflammation assessed by 18F-FDG PET, active mineral deposition assessed by 18F-fluoride PET, and vascular calcification in atherosclerotic plaque: a dual-tracer PET/CT study. J Nucl Med 52:1020–1027
Beheshti M, Saboury B, Mehta NN, Torigian DA, Werner T, Mohler E, Wilensky R, Newberg AB, Basu S, Langsteger W, Alavi A (2011) Detection and global quantification of cardiovascular molecular calcification by fluoro18-fluoride positron emission tomography/computed tomography—a novel concept. Hell J Nucl Med 14:114–120
Dweck MR, Chow MW, Joshi NV, Williams MC, Jones C, Fletcher AM, Richardson H, White A, McKillop G, van Beek EJ, Boon NA, Rudd JH, Newby DE (2012) Coronary arterial 18F-sodium fluoride uptake: a novel marker of plaque biology. J Am Coll Cardiol 59:1539–1548
Basu S, Høilund-Carlsen PF, Alavi A (2012) Assessing global cardiovascular molecular calcification with 18F-fluoride PET/CT: will this become a clinical reality and a challenge to CT calcification scoring? Eur J Nucl Med Mol Imaging 39:660–664
Blau M, Ganatra R, Bender M (1972) 18F-fluoride for bone imaging. Semin Nucl Med 2:31–37
Hawkins RA, Choi Y, Huang SC, Hoh CK, Dahlbom M, Schiepers C, Satyamurthy N, Barrio JR, Phelps ME (1992) Evaluation of the skeletal kinetics of fluorine-18-fluoride ion with PET. J Nucl Med 33:633–642
Pencina MJ, D’Agostino RB Sr, Larson MG, Massaro JM, Vasan RS (2009) Predicting the 30-year risk of cardiovascular disease: the Framingham heart study. Circulation 119:3078–3084
Tahara N, Kai H, Ishibashi M, Nakaura H, Kaida H, Baba K, Hayabuchi N, Imaizumi T (2006) Simvastatin attenuates plaque inflammation: evaluation by fluorodeoxyglucose positron emission tomography. J Am Coll Cardiol 48:1825–1831
ICRP Publication 80 (1999) Radiation dose to patients from radiopharmaceuticals. International Commission on Radiological Protection, Stockholm
Rudd JH, Myers KS, Bansilal S, Machac J, Pinto CA, Tong C, Rafique A, Hargeaves R, Farkouh M, Fuster V, Fayad ZA (2008) Atherosclerosis inflammation imaging with 18F-FDG PET: carotid, iliac, and femoral uptake reproducibility, quantification methods, and recommendations. J Nucl Med 49:871–878
Ellison RC, Zhang Y, Wagenknecht LE, Eckfeldt JH, Hopkins PN, Pankow JS, Djoussé L, Carr JJ (2005) Relation of the metabolic syndrome to calcified atherosclerotic plaque in the coronary arteries and aorta. Am J Cardiol 5:1180–1186
Czernin J, Satyamurthy N, Schiepers C (2010) Molecular mechanisms of bone 18F-NaF deposition. J Nucl Med 51:1826–1829
Kotze CW, Menezes LJ, Endozo R, Groves AM, Ell PJ, Yusuf SW (2009) Increased metabolic activity in abdominal aortic aneurysm detected by 18F-fluorodeoxyglucose (18F-FDG) positron emission tomography/computed tomography (PET/CT). Eur J Vasc Endovasc Surg 38:93–99
Sakalihasan N, Van Damme H, Gomez P, Rigo P, Lapiere CM, Nusgens B, Limet R (2002) Positron emission tomography (PET) evaluation of abdominal aortic aneurysm (AAA). Eur J Vasc Endovasc Surg 23:431–436
Eisen A, Tenenbaum A, Koren-Morag N, Tanne D, Shemesh J, Imazio M, Fisman EZ, Motro M, Schwammenthal E, Adler Y (2008) Calcification of the thoracic aorta as detected by spiral computed tomography among stable angina pectoris patients: association with cardiovascular events and death. Circulation 118:1328–1334
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
10554_2013_342_MOESM1_ESM.ppt
Fig.A: Correlation analysis between TBR and AC in the analyzed arterial districts. This analysis did not show any concordance between regional calcium load and TBR in any of the studied arterial segments. TBR = Target to Background Ratio. (PPT 224 kb)
10554_2013_342_MOESM2_ESM.ppt
Fig.B: Differences in average SUVmax of all the analyzed arterial districts values across different risk category groups. Significant difference in average SUVmax value was highlighted in high versus low risk groups, *p < 0.05). (PPT 83 kb)
Rights and permissions
About this article
Cite this article
Morbelli, S., Fiz, F., Piccardo, A. et al. Divergent determinants of 18F–NaF uptake and visible calcium deposition in large arteries: relationship with Framingham risk score. Int J Cardiovasc Imaging 30, 439–447 (2014). https://doi.org/10.1007/s10554-013-0342-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-013-0342-3