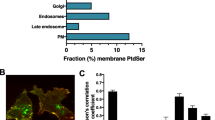
Abstract
Phosphatidylethanolamine (PE) is a major component in the mammalian plasma membrane. It is present mainly in the inner leaflet of the membrane bilayer in a viable, typical mammalian cell. However, accumulating evidence indicates that a number of biological events involve PE externalization. For instance, PE is concentrated at the surface of cleavage furrow between mitotic daughter cells and is correlated with the dynamics of contractile ring. In apoptotic cells, PE is exposed to the cell surface, thus providing a molecular marker for detection. In addition, PE is a cofactor in the anticoagulant mechanism, and a distinct distribution profile of PE has been documented at the blood–endothelium interface. These recent discoveries were made possible using PE-specific probes derived from duramycin and cinnamycin, which are members of type B lantibiotics. This review provides an account on the features of these PE-specific lantibiotics in the context of molecular probes for the characterization of PE on a cellular and tissue level. According to the existing data, PE is likely a versatile chemical species that plays a role in the regulation of defined biological and physiological activities. The utilities of lantibiotic-based molecular probes will help accelerate the characterization of PE as an abundant, yet elusive membrane component.







Similar content being viewed by others
References
Aoki Y, Uenaka T, Aoki J, Umeda M, Inoue K (1994) A novel peptide probe for studying the transbilayer movement of phosphatidylethanolamine. J Biochem 116:291–297
Berard M, Chantome R, Marcelli A, Boffa MC (1996) Antiphosphatidylethanolamine antibodies as the only antiphospholipid antibodies. I. Association with thrombosis and vascular cutaneous diseases. J Rheumatol 23:1369–1374
Bevers EM, Comfurius P, Zwaal RF (1996) Regulatory mechanisms in maintenance and modulation of transmembrane lipid asymmetry: pathophysiological implications. Lupus 5:480–487
Bevers EM, Comfurius P, Dekkers DW et al (1999) Lipid translocation across the plasma membrane of mammalian cells. Biochim Biophys Acta 1439:317–330
Bi EF, Lutkenhaus J (1991) FtsZ ring structure associated with division in Escherichia coli. Nature 354:161–164
Blankenberg FG, Katsikis PD, Tait JF, Davis RE, Naumovski L, Ohtsuki K, Kopiwoda S, Abrams MJ, Darkes M, Robbins RC, Maecker HT, Strauss HW (1998) In vivo detection and imaging of phosphatidylserine expression during programmed cell death. Proc Natl Acad Sci USA 95:6349–6354
Bleeker-Rovers CP, Boerman OC, Rennen HJ, Corstens FH, Oyen WJ (2004) Radiolabeled compounds in diagnosis of infectious and inflammatory disease. Curr Pharm Des 10:2935–2950
Brötz H, Josten M, Wiedemann I, Schneider U, Götz F, Bierbaum G, Sahl HG (1998) Role of lipid-bound peptidoglycan precursors in the formation of pores by nisin, epidermin and other lantibiotics. Mol Microbiol 30:317–3127
Chandran KB (1993) Flow dynamics in the human aorta. J Biomech Eng 115:611–616
Choung SY, Kobayashi T, Inoue J, Takemoto K, Ishitsuka H, Inoue K (1988) Hemolytic activity of a cyclic peptide Ro09–0198 isolated from Streptoverticillium. Biochim Biophys Acta 940:171–179
Devaux PF (1991) Static and dynamic lipid asymmetry in cell membranes. Biochemistry 30:1163–1173
Emoto K, Umeda M (2000) An essential role for a membrane lipid in cytokinesis. Regulation of contractile ring disassembly by redistribution of phosphatidylethanolamine. J Cell Biol 149:1215–1224
Emoto K, Kobayashi T, Yamaji A, Aizawa H, Yahara I, Inoue K, Umeda M (1996) Redistribution of phosphatidylethanolamine at the cleavage furrow of dividing cells during cytokinesis. Proc Natl Acad Sci USA 93:12867–12872
Emoto K, Toyama-Sorimachi N, Karasuyama H, Inoue K, Umeda M (1997) Exposure of phosphatidylethanolamine on the surface of apoptotic cells. Exp Cell Res 232:430–434
Emoto K, Inadome H, Kanaho Y, Narumiya S, Umeda M (2005) Local change in phospholipid composition at the cleavage furrow is essential for completion of cytokinesis. J Biol Chem 280:37901–37907
Esmon NL, Smirnov MD, Esmon CT (1997a) Lupus anticoagulants and thrombosis: the role of phospholipids. Haematologica 82:474–477
Esmon NL, Smirnov MD, Esmon CT (1997b) Thrombogenic mechanisms of antiphospholipid antibodies. Thromb Haemost 78:79–82
Esmon NL, Smirnov MD, Safa O, Esmon CT (1999) Lupus anticoagulants, thrombosis and the protein C system. Haematologica 84:446–451
Hachulla E, Harle JR, Sie P, Boffa MC (2007) Antiphosphatidylethanolamine antibodies are associated with an increased odds ratio for thrombosis. A multicenter study with the participation of the European Forum on antiphospholipid antibodies. Thromb Haemost 97:949–954
Hayashi F, Nagashima K, Terui Y, Kawamura Y, Matsumoto K, Itazaki H (1990) The structure of PA48009: the revised structure of duramycin. J Antibiot 43:1421–1430
Hosoda K, Ohya M, Kohno T, Maeda T, Endo S, Wakamatsu K (1996) Structure determination of an immunopotentiator peptide, cinnamycin, complexed with lysophosphatidylethanolamine by 1H-NMR1. J Biochem 119:226–230
Iwamoto K, Kobayashi S, Fukuda R, Umeda M, Kobayashi T, Ohta A (2004) Local exposure of phosphatidylethanolamine on the yeast plasma membrane is implicated in cell polarity. Genes Cells 9:891–903
Johnson LL, Schofield L, Donahay T, Narula N, Narula J (2005) 99mTc-annexin V imaging for in vivo detection of atherosclerotic lesions in porcine coronary arteries. J Nucl Med 46:1186–1193
Kaletta C, Entian KD, Jung G (1991) Prepeptide sequence of cinnamycin (Ro 09–0198): the first structural gene of a duramycin-type lantibiotic. Eur J Biochem 199:411–415
Kato U, Emoto K, Fredriksson C, Nakamura H, Ohta A, Kobayashi T, Murakami-Murofushi K, Kobayashi T, Umeda M (2002) A novel membrane protein, Ros3p, is required for phospholipid translocation across the plasma membrane in Saccharomyces cerevisiae. J Biol Chem 277:37855–37862
Kiestelaer BL, Reutelingsperger CPM, Heidendal GAK, Daemen MJAP, Mess WH, Hofstra L (2004) Noninvasive detection of plaque instability with use of radiolabeled annexin A5 in patients with carotid-artery atherosclerosis. N Engl J Med 350:1472–1473
Li Z, Wells CW, Esmon CT, Zhao M (2009) Phosphatidylethanolamine at the endothelial surface of aortic flow dividers. J Thromb Haemost 7:227–229
Li Z, Wells CW, North PE, Kumar S, Duris CB, McIntyre JA, Zhao M (2010) Phosphatidylethanolamine at the luminal endothelial surface—implications in hemostasis and thrombotic autoimmunity. Clin Appl Thromb Hem (in press)
Liu S, Edwards DS (1999) 99mTc-labeled small peptides as diagnostic radiopharmaceuticals. Chem Rev 99:2235–2268
Liu Z, Zhao M, Zhu X, Furenlid LR, Chen YC, Barrett HH (2007) In vivo dynamic imaging of myocardial cell death using 99mTc-labeled C2A domain of synaptotagmin I in a rat model of ischemia and reperfusion. Nucl Med Biol 34:907–915
Machaidze G, Seelig J (2003) Specific binding of cinnamycin (Ro 09–0198) to phosphatidylethanolamine. Comparison between micellar and membrane environments. Biochemistry 42:12570–12576
Marconescu A, Thorpe PE (2008) Coincident exposure of phosphatidylethanolamine and anionic phospholipids on the surface of irradiated cells. Biochim Biophys Acta 1778:2217–2224
Maulik N, Kagan VE, Tyurin VA, Das DK (1998) Redistribution of phosphatidylethanolamine and phosphatidylserine precedes reperfusion-induced apoptosis. Am J Physiol 274:H242–H248
Mileykovskaya E, Sun Q, Margolin W, Dowhan W (1998) Localization and function of early cell division proteins in filamentous Escherichia coli cells lacking phosphatidylethanolamine. J Bacteriol 180:4252–4257
Narula J, Acio ER, Narula N, Samuels LE, Fyfe B, Wood D, Fitzpatrick JM, Raghunath PN, Tomaszewski JE, Kelly C, Steinmetz N, Green A, Tait JF, Leppo J, Blankenberg FG, Jain D, Strauss HW (2001) Annexin-V imaging for noninvasive detection of cardiac allograft rejection. Nat Med 7:1347–1352
Pag U, Sahl HG (2002) Multiple activities in lantibiotics—models for the design of novel antibiotics? Curr Pharm Des 8:815–833
Rouser G, Yamamoto A, Kritchevsky G (1971) Cellular membranes. Structure and regulation of lipid class composition species differences, changes with age, and variations in some pathological states. Arch Intern Med 127:1105–1121
Sahl HG, Bierbaum G (1998) Lantibiotics: biosynthesis and biological activities of uniquely modified peptides from gram-positive bacteria. Annu Rev Microbiol 52:41–79
Sahu SK, Gummadi SN, Manoj N, Aradhyam GK (2007) Phospholipid scramblases: an overview. Arch Biochem Biophys 462:103–114
Sanmarco M, Alessi MC, Harle JR, Sapin C, Aillaud MF, Gentile S, Juhan-Vague I, Weiller PJ (2001) Antibodies to phosphatidylethanolamine as the only antiphospholipid antibodies found in patients with unexplained thromboses. Thromb Haemost 85:800–805
Seelig J (2004) Thermodynamics of lipid-peptide interactions. Biochim Biophys Acta 1666:40–50
Smirnov MD, Esmon CT (1994) Phosphatidylethanolamine incorporation into vesicles selectively enhances factor Va inactivation by activated protein C. J Biol Chem 269:816–819
Smirnov MD, Triplett DT, Comp PC, Esmon NL, Esmon CT (1995) On the role of phosphatidylethanolamine in the inhibition of activated protein C activity by antiphospholipid antibodies. J Clin Invest 95:309–316
Smirnov MD, Ford DA, Esmon CT, Esmon NL (1999) The effect of membrane composition on the hemostatic balance. Biochemistry 38:3591–3598
Song Z, Steller H (1999) Death by design: mechanism and control of apoptosis. Trends Cell Biol 9:M49–M52
Sosnovik DE, Schellenberger EA, Nahrendorf M, Novikov MS, Matsui T, Dai G, Reynolds F, Grazette L, Rosenzweig A, Weissleder R, Josephson L (2005) Magnetic resonance imaging of cardiomyocyte apoptosis with a novel magneto-optical nanoparticle. Magn Reson 54:718–724
Spector AA, Yorek MA (1985) Membrane lipid composition and cellular function. J Lipid Res 26:1015–1035
Sugi T, Katsunuma J, Izumi S, McIntyre JA, Makino T (1999) Prevalence and heterogeneity of antiphosphatidylethanolamine antibodies in patients with recurrent early pregnancy losses. Fertil Steril 71:1060–1065
Sugi T, Matsubayashi H, Inomo A, Dan L, Makino T (2004) Antiphosphatidylethanolamine antibodies in recurrent early pregnancy loss and mid-to-late pregnancy loss. J Obstet Gynaecol Res 30:326–332
Thimister PW, Hofstra L, Liem IH, Boersma HH, Kemerink G, Reutelingsperger CP, Heidendal GA (2003) In vivo detection of cell death in the area at risk in acute myocardial infarction. J Nucl Med 44:391–396
Tian Y, Jackson P, Gunter C, Wang J, Rock CO, Jackowski S (2006) Placental thrombosis and spontaneous fetal death in mice deficient in ethanolamine kinase 2. J Biol Chem 281:28438–28449
Umeda M, Emoto K (1999) Membrane phospholipid dynamics during cytokinesis: regulation of actin filament assembly by redistribution of membrane surface phospholipid. Chem Phys Lipids 101:81–91
Vinatier D, Dufour P, Cosson M, Houpeau JL (2001) Antiphospholipid syndrome and recurrent miscarriages. Eur J Obstet Gynecol Reprod Biol 96:37–50
Wakamatsu K, Choung SY, Kobayashi T, Inoue K, Higashijima T, Miyazawa T (1990) Complex formation of peptide antibiotic Ro09–0198 with lysophosphatidylethanolamine: 1H NMR analyses in dimethyl sulfoxide solution. Biochemistry 29:113–118
Widdick DA, Dodd HM, Barraille P, White J, Stein TH, Chater KF, Gasson MJ, Bibb MJ (2003) Cloning and engineering of the cinnamycin biosynthetic gene cluster from Streptomyces cinnamoneus cinnamoneus DSM 40005. Proc Natl Acad Sci USA 100:4316–4321
Williamson P, Schlegel RA (2002) Transbilayer phospholipid movement and the clearance of apoptotic cells. Biochim Biophys 1585:53–63
Wyllie AH (1997) Apoptosis: an overview. Br Med Bull 53:451–465
Zhao M, Beauregard DA, Loizou L, Davletov B, Brindle KM (2001) Non-invasive detection of apoptosis using magnetic resonance imaging and a targeted contrast agent. Nat Med 7:1241–1244
Zhao M, Zhu X, Ji S, Zhou J, Ozker KS, Fang W, Molthen RC, Hellman RS (2006) 99mTc-labeled C2A domain of synaptotagmin I as a target-specific molecular probe for noninvasive imaging of acute myocardial infarction. J Nucl Med 47:1367–1374
Zhao M, Li Z, Bugenhagen S (2008) 99mTc-labeled duramycin as a novel phosphatidylethanolamine-binding molecular probe. J Nucl Med 49:1345–1352
Zimmermann N, Freund S, Fredenhagen A, Jung G (1993) Solution structures of the lantibiotics duramycin B and C. Eur J Biochem 216:419–428
Acknowledgment
Funding support from the National Institutes of Health is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhao, M. Lantibiotics as probes for phosphatidylethanolamine. Amino Acids 41, 1071–1079 (2011). https://doi.org/10.1007/s00726-009-0386-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-009-0386-9