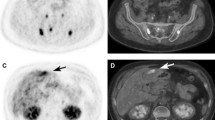
Abstract
The purpose of this prospective study was to compare the value of DOPA PET-CT with FDG PET-CT in the detection of malignant lesions in patients with medullary thyroid carcinoma (MTC). Twenty-six consecutive patients (10 men, 16 women, mean age 59 ± 14 years) with elevated calcitonin levels were evaluated in this prospective study. DOPA and FDG PET-CT modalities were performed within a maximum of 4 weeks (median 7 days) in all patients. The data were evaluated on a patient- and lesion-based analysis. The final diagnosis of positive PET lesions was based on histopathological findings and/or imaging follow-up studies (i.e., DOPA and/or FDG PET-CT) for at least 6 months (range 6–24 months). In 21 (21/26) patients at least one malignant lesion was detected by DOPA PET, while only 15 (15/26) patients showed abnormal FDG uptake. DOPA PET provided important additional information in the follow-up assessment in seven (27%) patients which changed the therapeutic management. The patient-based analysis of our data demonstrated a sensitivity of 81% for DOPA PET versus 58% for FDG PET, respectively. In four (4/26) postoperative patients DOPA and FDG PET-CT studies were negative in spite of elevated serum calcitonin and CEA levels as well as abnormal pentagastrin tests. Overall 59 pathological lesions with abnormal tracer uptake were seen on DOPA and/or FDG PET studies. In the final diagnosis 53 lesions proved to be malignant. DOPA PET correctly detected 94% (50/53) of malignant lesions, whereas only 62% (33/53) of malignant lesions were detected with FDG PET. DOPA PET-CT showed superior results to FDG PET-CT in the preoperative and follow-up assessment of MTC patients. Therefore, we recommend DOPA PET-CT as a one-stop diagnostic procedure to provide both functional and morphological data in order to select those patients who may benefit from (re-)operation with curative intent as well as guiding further surgical procedures.





Similar content being viewed by others
References
Lairmore TC, Wells SA Jr (1991) Medullary carcinoma of the thyroid: current diagnosis and management. Sem Surg Oncol 7:92–99
Saad MF, Ordonez NG, Rashid RK et al (1984) Medullary carcinoma of the thyroid. A study of the clinical features and prognostic factors in 161 patients. Medicine 63:319–342
Brierley J, Tsang R, Simpson WJ et al (1996) Medullary thyroid cancer: analyses of survival and prognostic factors and the role of radiation therapy in local control. Thyroid 6:305–310
Kebebew E, Clark OH (2000) Medullary thyroid cancer. Curr Treatm Opt Oncol 1:359–367
Vitale G, Caraglia M, Ciccarelli A et al (2001) Current approaches and perspectives in the therapy of medullary thyroid carcinoma. Cancer 91:1797–1808
Hoang-Vu C, Dralle H, Schroder S et al (1992) The nude rat is established as a model for endocrine studies: regulation of thyroid function and xenotransplantation of a thyroid tumor cell line. Lab Anim Sci 42:164–167
Orlandi F, Caraci P, Mussa A et al (2001) Treatment of medullary thyroid carcinoma: an update. Endocr Relat Cancer 8:135–147
Iten F, Muller B, Schindler C et al (2007) Response to [90yttrium-DOTA]-TOC treatment is associated with long-term survival benefit in metastasized medullary thyroid cancer: a phase II clinical trial. Clin Cancer Res 13:6696–6702
Cohen EG, Shaha AR, Rinaldo A et al (2004) Medullary thyroid carcinoma. Acta Otolaryngol 124:544–557
de Groot JW, Links TP, Sluiter WJ et al (2007) Locoregional control in patients with palpable medullary thyroid cancer: results of standardized compartment-oriented surgery. Head Neck 29:857–863
Moley JF, DeBenedetti MK (1999) Patterns of nodal metastases in palpable medullary thyroid carcinoma: recommendations for extent of node dissection. Ann Surg 229:880–887 discussion 887–888
Scollo C, Baudin E, Travagli JP et al (2003) Rationale for central and bilateral lymph node dissection in sporadic and hereditary medullary thyroid cancer. J Clin Endocrinol Metab 88:2070–2075
Modigliani E, Cohen R, Campos JM et al (1998) Prognostic factors for survival and for biochemical cure in medullary thyroid carcinoma: results in 899 patients. The GETC Study Group. Groupe d’etude des tumeurs a calcitonine. Clin Endocrinol 48:265–273
Machens A, Niccoli-Sire P, Hoegel J et al (2003) Early malignant progression of hereditary medullary thyroid cancer. N Engl J Med 349:1517–1525
Bollen EC, Goei R, van’t Hof-Grootenboer BE et al (1994) Interobserver variability and accuracy of computed tomographic assessment of nodal status in lung cancer. Ann Thorac Surg 58:158–162
Gdeedo A, Van Schil P, Corthouts B et al (1997) Comparison of imaging TNM [(i)TNM] and pathological TNM [pTNM] in staging of bronchogenic carcinoma. Eur J Cardiothorac Surg 12:224–227
Adams S, Baum RP, Hertel A et al (1998) Comparison of metabolic and receptor imaging in recurrent medullary thyroid carcinoma with histopathological findings. Eur J Nucl Med 25:1277–1283
Baulieu JL, Guilloteau D, Delisle MJ et al (1987) Radioiodinated meta-iodobenzylguanidine uptake in medullary thyroid cancer. A French cooperative study. Cancer 60:2189–2194
Berna L, Cabezas R, Mora J et al (1995) 111In-octreotide and 99mTc(V)-dimercaptosuccinic acid studies in the imaging of recurrent medullary thyroid carcinoma. J Endocrinol 144:339–345
Berna L, Chico A, Matias-Guiu X et al (1998) Use of somatostatin analogue scintigraphy in the localization of recurrent medullary thyroid carcinoma. Eur J Nucl Med. 25:1482–1488
Guerra U, Pizzocaro C, Terzi A et al (1988) The use of 99mTc(V)DMSA as imaging for the medullary thyroid carcinoma (MTC). J Nucl Med Allied Sci 32:242–247
Krausz Y, Rosler A, Guttmann H et al (1999) Somatostatin receptor scintigraphy for early detection of regional and distant metastases of medullary carcinoma of the thyroid. Clin Nucl Med 24:256–260
Mojiminiyi OA, Udelsman R, Soper ND et al (1991) Pentavalent Tc-99m DMSA scintigraphy. Prospective evaluation of its role in the management of patients with medullary carcinoma of the thyroid. Clin Nucl Med 16:259–262
Ugur O, Kostakglu L, Guler N et al (1996) Comparison of 99mTc(V)-DMSA, 201Tl and 99mTc-MIBI imaging in the follow-up of patients with medullary carcinoma of the thyroid. Eur J Nucl Med 23:1367–1371
Brandt-Mainz K, Muller SP, Gorges R et al (2000) The value of fluorine-18 fluorodeoxyglucose PET in patients with medullary thyroid cancer. Eur J Nucl Med 27:490–496
de Groot JW, Links TP, Jager PL et al (2004) Impact of 18F-fluoro-2-deoxy-D-glucose positron emission tomography (FDG-PET) in patients with biochemical evidence of recurrent or residual medullary thyroid cancer. Ann Surg Oncol 11:786–794
Diehl M, Risse JH, Brandt-Mainz K et al (2001) Fluorine-18 fluorodeoxyglucose positron emission tomography in medullary thyroid cancer: results of a multicentre study. Eur J Nucl Med 28:1671–1676
Gasparoni P, Rubello D, Ferlin G (1997) Potential role of fluorine-18-deoxyglucose (FDG) positron emission tomography (PET) in the staging of primitive and recurrent medullary thyroid carcinoma. J Endocrinol Investig 20:527–530
Musholt TJ, Musholt PB, Dehdashti F et al (1997) Evaluation of fluorodeoxyglucose-positron emission tomographic scanning and its association with glucose transporter expression in medullary thyroid carcinoma and pheochromocytoma: a clinical and molecular study. Surgery 122:1049–1060; discussion 1060–1041
Szakall S Jr, Esik O, Bajzik G et al (2002) 18F-FDG PET detection of lymph node metastases in medullary thyroid carcinoma. J Nucl Med 43:66–71
Simon GH, Nitzsche EU, Laubenberger JJ et al (1996) PET imaging of recurrent medullary thyroid cancer. Nuklearmedizin 35:102–104
Iagaru A, Masamed R, Singer PA et al (2007) Detection of occult medullary thyroid cancer recurrence with 2-deoxy-2-[F-18]fluoro-D-glucose-PET and PET/CT. Mol Imaging Biol 9:72–77
Beuthien-Baumann B, Strumpf A, Zessin J et al (2007) Diagnostic impact of PET with 18F-FDG, 18F-DOPA and 3-O-methyl-6-[18F]fluoro-DOPA in recurrent or metastatic medullary thyroid carcinoma. Eur J Nucl Med Mol Imaging 34:1604–1609
Hoegerle S, Altehoefer C, Ghanem N et al (2001) 18F-DOPA positron emission tomography for tumour detection in patients with medullary thyroid carcinoma and elevated calcitonin levels. Eur J Nucl Med 28:64–71
Nader M, Eichinger R, Frey A (2007) Development and validation of a new analytical HPLC- method for the quality control of 6-[18F]fluoro-L-dopa. J Nucl Med; 48(Supplement 2):138
Miyauchi A, Onishi T, Morimoto S et al (1984) Relation of doubling time of plasma calcitonin levels to prognosis and recurrence of medullary thyroid carcinoma. Ann Surg 199:461–466
Koopmans KP, de Groot JW, Plukker JT et al (2008) 18F-dihydroxyphenylalanine PET in patients with biochemical evidence of medullary thyroid cancer: relation to tumor differentiation. J Nucl Med 49:524–531
Jager PL, Chirakal R, Marriott CJ et al (2008) 6-L-18F-fluorodihydroxyphenylalanine PET in neuroendocrine tumors: basic aspects and emerging clinical applications. J Nucl Med 49:573–586
Ong SC, Schoder H, Patel SG et al (2007) Diagnostic accuracy of 18F-FDG PET in restaging patients with medullary thyroid carcinoma and elevated calcitonin levels. J Nucl Med 48:501–507
Li S, Beheshti M (2005) The radionuclide molecular imaging and therapy of neuroendocrine tumors. Current Cancer Drug Targets 5:139–148
Bockisch A, Brandt-Mainz K, Gorges R et al (2003) Diagnosis in medullary thyroid cancer with [18F]FDG-PET and improvement using a combined PET/CT scanner. Acta Med Austriaca 30:22–25
Gotthardt M, Battmann A, Hoffken H et al (2004) 18F-FDG PET, somatostatin receptor scintigraphy, and CT in metastatic medullary thyroid carcinoma: a clinical study and an analysis of the literature. Nucl Med Common 25:439–443
Wang Q, Takashima S, Fukuda H et al (1999) Detection of medullary thyroid carcinoma and regional lymph node metastases by magnetic resonance imaging. Arch Otolaryngol Head Neck Surg 125:842–848
Tung WS, Vesely TM, Moley JF (1995) Laparoscopic detection of hepatic metastases in patients with residual or recurrent medullary thyroid cancer. Surgery 118:1024–1029; discussion 1029–1030
Esik O, Szavcsur P, Szakall S Jr et al (2001) Angiography effectively supports the diagnosis of hepatic metastases in medullary thyroid carcinoma. Cancer 91:2084–2095
Adams S, Baum R, Rink T et al (1998) Limited value of fluorine-18 fluorodeoxyglucose positron emission tomography for the imaging of neuroendocrine tumours. Eur J Nucl Med 25:79–83
James C, Starks M, MacGillivray DC et al (1999) The use of imaging studies in the diagnosis and management of thyroid cancer and hyperparathyroidism. Surg Oncol Clin N Am 8:145–169
Raue F, Kotzerke J, Reinwein D et al (1993) Prognostic factors in medullary thyroid carcinoma: evaluation of 741 patients from the German Medullary Thyroid Carcinoma Register. Clin Investig 71:7–12
Samaan NA, Schultz PN, Hickey RC (1989) Medullary thyroid carcinoma: prognosis of familial versus nonfamilial disease and the role of radiotherapy. Horm Metab Res 21:21–25
Acknowledgment
We thank Aliasghar Khorsand, PhD from the Medical University of Vienna for his assistance with statistics and Bernhard Gruy, MS for his technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Beheshti, M., Pöcher, S., Vali, R. et al. The value of 18F-DOPA PET-CT in patients with medullary thyroid carcinoma: comparison with 18F-FDG PET-CT. Eur Radiol 19, 1425–1434 (2009). https://doi.org/10.1007/s00330-008-1280-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-008-1280-7