Abstract
Background
In oncological imaging using PET/CT, the standardized uptake value has become the most common parameter used to measure tracer accumulation. The aim of this analysis was to evaluate ultra high definition (UHD) and ordered subset expectation maximization (OSEM) PET/CT reconstructions for their potential impact on quantification.
Patients and methods
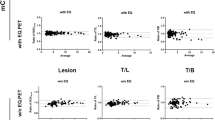
We analyzed 40 PET/CT scans of lung cancer patients who had undergone PET/CT. Standardized uptake values corrected for body weight (SUV) and lean body mass (SUL) were determined in the single hottest lesion in the lung and normalized to the liver for UHD and OSEM reconstruction. Quantitative uptake values and their normalized ratios for the two reconstruction settings were compared using the Wilcoxon test. The distribution of quantitative uptake values and their ratios in relation to the reconstruction method used were demonstrated in the form of frequency distribution curves, box-plots and scatter plots. The agreement between OSEM and UHD reconstructions was assessed through Bland-Altman analysis.
Results
A significant difference was observed after OSEM and UHD reconstruction for SUV and SUL data tested (p < 0.0005 in all cases). The mean values of the ratios after OSEM and UHD reconstruction showed equally significant differences (p < 0.0005 in all cases). Bland-Altman analysis showed that the SUV and SUL and their normalized values were, on average, up to 60 % higher after UHD reconstruction as compared to OSEM reconstruction.
Conclusion
OSEM and HD reconstruction brought a significant difference for SUV and SUL, which remained constantly high after normalization to the liver, indicating that standardization of reconstruction and the use of comparable SUV measurements are crucial when using PET/CT.






Similar content being viewed by others
References
Kobe C, Dietlein M, Franklin J, Markova J, Lohri A, Amthauer H, et al. Positron emission tomography has a high negative predictive value for progression or early relapse for patients with residual disease after first-line chemotherapy in advanced-stage Hodgkin lymphoma. Blood. 2008;112:3989–94.
Barrington SF, Mikhaeel NG, Kostakoglu L, Meignan M, Hutchings M, Mueller SP, et al. Role of imaging in the staging and response assessment of lymphoma: consensus of the international conference on malignant lymphomas imaging working group. J Clin Oncol. 2014;32:3048–58.
Ito T, Wan CN, Reivich M, Kuhl DE, Casella J, Fowler JS, et al. Labeled 2-deoxy-D-glucose analogs. 18F-labeled 2-deoxy- 2-fluoro-D-glucose, 2-deoxy- 2-fluoro-D-mannose and 14C–2-deoxy- 2-fluoro-D-glucose. J Label Compd Radiopharm. 1978;14:175–83.
Warburg O, Posener K, Negelein E. Über den Stoffwechsel der Carcinomzelle. Biochem Z. 1924;152:309–35.
Boellaard R, Delgado-Bolten R, Oyen WJG, Giammarile F, Tatsch K, Eschner W, et al. FDG PET and PET/CT: EANM procedure guidelines for tumour PET imaging: version 2.0. Eur J Nucl Med Mol Imaging. 2015;42:328–54.
Boellaard R, Oyen WJ, Hoekstra CJ, Hoekstra OS, Visser EP, Willemsen AT, et al. The Netherlands protocol for standardisation and quantification of FDG whole body PET studies in multi-centre trials. Eur J Nucl Med Mol Imaging. 2008;35:2320–33.
Benz MR, Evilevitch V, Allen-Auerbach MS, Eilber FC, Phelps ME, Czernin J, et al. Treatment monitoring by 18F-FDG PET/CT in patients with sarcomas: interobserver variability of quantitative parameters in treatment-induced changes in histopathologically responding and nonresponding tumors. J Nucl Med. 2008;49:1038–46.
Wahl RL, Jacene H, Kasamon Y, Lodge MA. From RECIST to PERCIST: evolving considerations for PET response criteria in solid tumors. J Nucl Med. 2009;50 Suppl 1:122S–50.
Kahraman D, Scheffler M, Zander T, Nogova L, Lammertsma AA, Boellaard R, et al. Quantitative analysis of response to treatment with erlotinib in advanced non–small cell lung cancer using 18F-FDG and 3’-Deoxy-3’-18F-Fluorothymidine PET. J Nucl Med. 2011;52:1871–7.
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45:228–47.
Chiaravalloti A, Danieli R, Abbatiello P, Di Pietro B, Travascio L, Cantonetti M, et al. Factors affecting intrapatient liver and mediastinal blood pool 18F-FDG standardized uptake value changes during ABVD chemotherapy in Hodgkin’s lymphoma. Eur J Nucl Med Mol Imaging. 2014;41:1123–32.
Shankar LK, Hoffman JM, Bacharach S, Graham MM, Karp J, Lammertsma AA, et al. Consensus recommendations for the use of 18F-FDG PET as an indicator of therapeutic response in patients in national cancer institute trials. J Nucl Med. 2006;47:1059–66.
Adams MC, Turkington TG, Wilson JM, Wong TZ. A systematic review of the factors affecting accuracy of SUV measurements. AJR. 2010;195:310–20.
Lasnon C, Desmonts C, Quak E, Gervais R, Do P, Dubos-Arvis C, et al. Harmonizing SUVs in multicentre trials when using different generation PET systems: prospective validation in non-small cell lung cancer patients. Eur J Nucl Med Mol Imaging. 2013;40:985–96.
Rubello D, Gordien P, Morliere C, Guyot M, Bordenave L, Colletti PM, et al. Variability of hepatic 18F-FDG uptake at interim PET in patients with Hodgkin lymphoma. Clin Nucl Med. 2015;40:e405–10.
Hasenclever D, Kurch L, Mauz-Körholz C, Elsner A, Georgi T, Wallace H, et al. qPET – a quantitative extension of the Deauville scale to assess response in interim FDG-PET scans in lymphoma. Eur J Nucl Med Mol Imaging. 2014;41:1301–8.
Paquet N, Albert A, Foidart J, Hustinx R. Within-patient Variability of 18F-FDG: standardized uptake values in normal tissues. J Nucl Med. 2004;45:784–8.
Keyes Jr JW. SUV: standard uptake or silly useless value? J Nucl Med. 1995;36:1836–9.
Visser EP, Boerman OC, Oyen WJG. SUV: from silly useless value to smart uptake value. J Nucl Med. 2010;51:173–5.
Tylski P, Stute S, Grotus N, Doyeux K, Hapdey S, Gardin I, et al. Comparative assessment of methods for estimating tumor volume and standardized uptake value in 18F-FDG PET. J Nucl Med. 2010;51:268–76.
Hristova I, Boellaard R, Vogel W, Mottaghy F, Marreaud S, Collette S, et al. Retrospective quality control review of FDG scans in the imaging sub-study of PALETTE EORTC 62072/VEG110727: a randomized, double-blind, placebo-controlled phase III trial. Eur J Nucl Med Mol Imaging. 2015;42:848–57.
Lodge MA, Chaudhry MA, Wahl RL. Noise considerations for PET quantification using maximum and peak standardized uptake value. J Nucl Med. 2012;53:1041–7.
Brendle C, Kupferschläger J, Nikolaou K, la Fougère C, Gatidis S, Pfannenberg C. Is the standard uptake value (SUV) appropriate for quantification in clinical PET imaging? - Variability induced by different SUV measurements and varying reconstruction methods. Eur J Radiol. 2015;84:158–62.
Boellaard R, Krak NC, Hoekstra OS, Lammertsma AA. Effects of noise, image resolution, and ROI definition on the accuracy of standard uptake values: a simulation study. J Nucl Med. 2004;45:1519–27.
Akamatsu G, Mitsumoto K, Taniguchi T, Tsutsui Y, Baba S, Sasaki M. Influences of point-spread function and time-of-flight reconstructions on standardized uptake value of lymph node metastases in FDG-PET. Eur J Radiol. 2014;83:226–30.
Akamatsu G, Ishikawa K, Mitsumoto K, Taniguchi T, Ohya N, Baba S, et al. Improvement in PET/CT image quality with a combination of Point-Spread Function and Time-of-Flight in relation to reconstruction parameters. J Nucl Med. 2012;53:1716–22.
Lasnon C, Hicks RJ, Beauregard JM, Milner A, Paciencia M, Guizard AV, et al. Impact of point spread function reconstruction on thoracic lymph node staging with 18F-FDG PET/CT in non-small cell lung cancer. Clin Nucl Med. 2012;37:971–6.
Barrington SF, MacKewn JE, Schleyer P, Marsden PK, Mikhaeel NG, Qian W, et al. Establishment of a UK-wide network to facilitate the acquisition of quality assured FDG–PET data for clinical trials in lymphoma. Ann Oncol. 2011;22:739–45.
Compliance with Ethical Standards
All procedures performed in our analysis were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For our type of study formal consent is not required.
Conflict of interests
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kuhnert, G., Boellaard, R., Sterzer, S. et al. Impact of PET/CT image reconstruction methods and liver uptake normalization strategies on quantitative image analysis. Eur J Nucl Med Mol Imaging 43, 249–258 (2016). https://doi.org/10.1007/s00259-015-3165-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-015-3165-8