Abstract
ᅟ
Imaging of inflammation early after myocardial infarction (MI) is a promising approach to the guidance of novel molecular interventions that support endogenous healing processes. 18F-FDG PET has been used, but may be complicated by physiological myocyte uptake. We evaluated the potential of two alternative imaging targets: lactoferrin binding by 68Ga-citrate and somatostatin receptor binding by 68Ga-DOTATATE.
Methods
C57Bl/6 mice underwent permanent coronary artery ligation. Serial PET imaging was performed 3 – 7 days after MI using 68Ga-citrate, 68Ga-DOTATATE, or 18F-FDG with ketamine/xylazine suppression of myocyte glucose uptake. Myocardial perfusion was evaluated by 13N-ammonia PET and cardiac geometry by contrast-enhanced ECG-gated CT.
Results
Mice exhibited a perfusion defect of 30 – 40 % (of the total left ventricle) with apical anterolateral wall akinesia and thinning on day 7 after MI. 18F-FDG with ketamine/xylazine suppression demonstrated distinct uptake in the infarct region, as well as in the border zone and remote myocardium. The myocardial standardized uptake value in MI mice was significantly higher than in healthy mice under ketamine/xylazine anaesthesia (1.9 ± 0.4 vs. 1.0 ± 0.1). 68Ga images exhibited high blood pool activity with no specific myocardial uptake up to 90 min after injection (tissue-to-blood contrast 0.9). 68Ga-DOTATATE was rapidly cleared from the blood, but myocardial SUV was very low (0.10 ± 0.03).
Conclusion
Neither 68Ga nor 68Ga-DOTATATE is a useful alternative to 18F-FDG for PET imaging of myocardial inflammation after MI in mice. Among the three tested approaches, 18F-FDG with ketamine/xylazine suppression of cardiomyocyte uptake remains the most practical imaging marker of post-infarct inflammation.






Similar content being viewed by others
References
Mezzaroma E, Toldo S, Farkas D, Seropian IM, Van Tassell BW, Salloum FN, et al. The inflammasome promotes adverse cardiac remodeling following acute myocardial infarction in the mouse. Proc Natl Acad Sci U S A. 2011;108:19725–30. doi:10.1073/pnas.1108586108.
Swirski FK, Nahrendorf M. Leukocyte behavior in atherosclerosis, myocardial infarction, and heart failure. Science. 2013;339:161–6. doi:10.1126/science.1230719.
Kempf T, Zarbock A, Vestweber D, Wollert KC. Anti-inflammatory mechanisms and therapeutic opportunities in myocardial infarct healing. J Mol Med. 2012;90:361–9. doi:10.1007/s00109-011-0847-y.
Bengel FM, George RT, Schuleri KH, Lardo AC, Wollert KC. Image-guided therapies for myocardial repair: concepts and practical implementation. Eur Heart J Cardiovasc Imaging. 2013;14:741–51. doi:10.1093/ehjci/jet038.
Basu S, Zhuang H, Torigian DA, Rosenbaum J, Chen W, Alavi A. Functional imaging of inflammatory diseases using nuclear medicine techniques. Semin Nucl Med. 2009;39:124–45. doi:10.1053/j.semnuclmed.2008.10.006.
Rudd JH, Warburton EA, Fryer TD, Jones HA, Clark JC, Antoun N, et al. Imaging atherosclerotic plaque inflammation with [18F]-fluorodeoxyglucose positron emission tomography. Circulation. 2002;105:2708–11.
Lee WW, Marinelli B, van der Laan AM, Sena BF, Gorbatov R, Leuschner F, et al. PET/MRI of inflammation in myocardial infarction. J Am Coll Cardiol. 2012;59:153–63. doi:10.1016/j.jacc.2011.08.066.
Campian ME, Hardziyenka M, de Bruin K, van Eck-Smit BL, de Bakker JM, Verberne HJ, et al. Early inflammatory response during the development of right ventricular heart failure in a rat model. Eur J Heart Fail. 2010;12:653–8. doi:10.1093/eurjhf/hfq066.
Campian ME, Verberne HJ, Hardziyenka M, de Groot EA, van Moerkerken AF, van Eck-Smit BL, et al. Assessment of inflammation in patients with arrhythmogenic right ventricular cardiomyopathy/dysplasia. Eur J Nucl Med Mol Imaging. 2010;37:2079–85. doi:10.1007/s00259-010-1525-y.
Chen WC, Tsai KD, Chen CH, Lin MS, Chen CM, Shih CM, et al. Role of gallium-67 scintigraphy in the evaluation of occult sepsis in the medical ICU. Intern Emerg Med. 2012;7:53–8. doi:10.1007/s11739-011-0739-8.
Hofman MS, Kong G, Neels OC, Eu P, Hong E, Hicks RJ. High management impact of Ga-68 DOTATATE (GaTate) PET/CT for imaging neuroendocrine and other somatostatin expressing tumours. J Med Imaging Radiat Oncol. 2012;56:40–7. doi:10.1111/j.1754-9485.2011.02327.x.
Silvola JM, Laitinen I, Sipila HJ, Laine VJ, Leppanen P, Yla-Herttuala S, et al. Uptake of 68gallium in atherosclerotic plaques in LDLR-/-ApoB100/100 mice. EJNMMI Res. 2011;1:14. doi:10.1186/2191-219X-1-14.
Kayani I, Conry BG, Groves AM, Win T, Dickson J, Caplin M, et al. A comparison of 68Ga-DOTATATE and 18F-FDG PET/CT in pulmonary neuroendocrine tumors. J Nucl Med. 2009;50:1927–32. doi:10.2967/jnumed.109.066639.
Armani C, Catalani E, Balbarini A, Bagnoli P, Cervia D. Expression, pharmacology, and functional role of somatostatin receptor subtypes 1 and 2 in human macrophages. J Leukoc Biol. 2007;81:845–55. doi:10.1189/jlb.0606417.
Schatka I, Wollenweber T, Haense C, Brunz F, Gratz KF, Bengel FM. Peptide receptor targeted radionuclide therapy alters inflammation in atherosclerotic plaques. J Am Coll Cardiol. 2013;62:2344–5. doi:10.1016/j.jacc.2013.08.1624.
Kempf T, Zarbock A, Widera C, Butz S, Stadtmann A, Rossaint J, et al. GDF-15 is an inhibitor of leukocyte integrin activation required for survival after myocardial infarction in mice. Nat Med. 2011;17:581–8. doi:10.1038/nm.2354.
Korf-Klingebiel M, Kempf T, Schluter KD, Willenbockel C, Brod T, Heineke J, et al. Conditional transgenic expression of fibroblast growth factor 9 in the adult mouse heart reduces heart failure mortality after myocardial infarction. Circulation. 2011;123:504–14. doi:10.1161/CIRCULATIONAHA.110.989665.
White DA, Su Y, Kanellakis P, Kiriazis H, Morand EF, Bucala R, et al. Differential roles of cardiac and leukocyte derived macrophage migration inhibitory factor in inflammatory responses and cardiac remodelling post myocardial infarction. J Mol Cell Cardiol. 2014;69:32–42. doi:10.1016/j.yjmcc.2014.01.015.
Guo N, Lang L, Li W, Kiesewetter DO, Gao H, Niu G, et al. Quantitative analysis and comparison study of [18F]AlF-NOTA-PRGD2, [18F]FPPRGD2 and [68Ga]Ga-NOTA-PRGD2 using a reference tissue model. PLoS One. 2012;7:e37506. doi:10.1371/journal.pone.0037506.
Perez J, Viollet C, Doublier S, Videau C, Epelbaum J, Baud L. Somatostatin binds to murine macrophages through two distinct subsets of receptors. J Neuroimmunol. 2003;138:38–44.
Li X, Samnick S, Lapa C, Israel I, Buck AK, Kreissl MC, et al. 68Ga-DOTATATE PET/CT for the detection of inflammation of large arteries: correlation with 18F-FDG, calcium burden and risk factors. EJNMMI Res. 2012;2:52. doi:10.1186/2191-219X-2-52.
Rominger A, Saam T, Vogl E, Ubleis C, la Fougere C, Forster S, et al. In vivo imaging of macrophage activity in the coronary arteries using 68Ga-DOTATATE PET/CT: correlation with coronary calcium burden and risk factors. J Nucl Med. 2010;51:193–7. doi:10.2967/jnumed.109.070672.
Cotugno G, Aurilio M, Annunziata P, Capalbo A, Faella A, Rinaldi V, et al. Noninvasive repetitive imaging of somatostatin receptor 2 gene transfer with positron emission tomography. Hum Gene Ther. 2011;22:189–96. doi:10.1089/hum.2010.098.
Saha JK, Xia J, Grondin JM, Engle SK, Jakubowski JA. Acute hyperglycemia induced by ketamine/xylazine anesthesia in rats: mechanisms and implications for preclinical models. Exp Biol Med (Maywood). 2005;230:777–84.
Ahmed N, Kansara M, Berridge MV. Acute regulation of glucose transport in a monocyte-macrophage cell line: Glut-3 affinity for glucose is enhanced during the respiratory burst. Biochem J. 1997;327(Pt 2):369–75.
Lumeng CN, Deyoung SM, Saltiel AR. Macrophages block insulin action in adipocytes by altering expression of signaling and glucose transport proteins. Am J Physiol Endocrinol Metab. 2007;292:E166–74. doi:10.1152/ajpendo.00284.2006.
Cheng VY, Slomka PJ, Le Meunier L, Tamarappoo BK, Nakazato R, Dey D, et al. Coronary arterial 18F-FDG uptake by fusion of PET and coronary CT angiography at sites of percutaneous stenting for acute myocardial infarction and stable coronary artery disease. J Nucl Med. 2012;53:575–83. doi:10.2967/jnumed.111.097550.
Harisankar CN, Mittal BR, Agrawal KL, Abrar ML, Bhattacharya A. Utility of high fat and low carbohydrate diet in suppressing myocardial FDG uptake. J Nucl Cardiol. 2011;18:926–36. doi:10.1007/s12350-011-9422-8.
Roivainen A, Jalkanen S, Nanni C. Gallium-labelled peptides for imaging of inflammation. Eur J Nucl Med Mol Imaging. 2012;39 Suppl 1:S68–77. doi:10.1007/s00259-011-1987-6.
Saraste A, Laitinen I, Weidl E, Wildgruber M, Weber AW, Nekolla SG, et al. Diet intervention reduces uptake of alphavbeta3 integrin-targeted PET tracer 18F-galacto-RGD in mouse atherosclerotic plaques. J Nucl Cardiol. 2012;19:775–84. doi:10.1007/s12350-012-9554-5.
Razavian M, Tavakoli S, Zhang J, Nie L, Dobrucki LW, Sinusas AJ, et al. Atherosclerosis plaque heterogeneity and response to therapy detected by in vivo molecular imaging of matrix metalloproteinase activation. J Nucl Med. 2011;52:1795–802. doi:10.2967/jnumed.111.092379.
Acknowledgments
The authors thank the preclinical molecular imaging and radiochemistry staff of the Department of Nuclear Medicine for their technical assistance. J.T.T. is supported by fellowships from the German Academic Exchange Service (DAAD) and the Canadian Institutes of Health Research. This project was partially funded by the REBIRTH-2 Cluster of Excellence and by EU FP7 grant PIRG08-GA-2010-276889 (F.M.B.).
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Suppl Fig 1
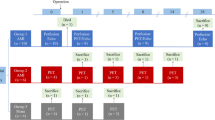
Experimental design and imaging protocols. (a) Experimental timeline with MI at day 0 followed by PET imaging at 3, 5 and 7 days post-MI. Mice were randomized and serially imaged with different sequential tracers as indicated horizontally (i.e. a different tracer used in each individual subject for each time point). (b) Imaging protocols for 68Ga-citrate and 68Ga-DOTATATE and (c) 18F-FDG acquisitions (acq) and 57Co transmission scans (Tx) under isoflurane (isofl) or ketamine/xylazine (KX) anaesthesia. 13N-Ammonia (NH3) PET scans and contrast CT imaging was performed only on day 7 post-MI. (PPTX 69 kb).
Suppl Fig 2
Late frame myocardial 68Ga-citrate distribution. (a) Representative parametric SUV-scaled 68Ga-citrate transaxial, coronal, and sagittal myocardial images obtained at 60-90 min, 120-150 min, and 180-210 min after injection in healthy control mice. (b) Semi-quantitative analysis of whole left ventricle myocardium and left atrial cavity (blood pool) SUV in late frames demonstrates persistence in blood pool over 3h after injection. (PPTX 1386 kb)
Suppl Fig 3
Early frame myocardial 68Ga-DOTATATE distribution. (a) Representative transaxial, coronal, and sagittal myocardial image at 7-10 min after injection demonstrates low myocardial activity early after tracer injection in a mouse 3 days post-MI. (b) Semi-quantitative analysis of whole myocardium and blood pool activity at 7-10 min after injection. (c) Early frame time-activity curves of kidney and urinary bladder demonstrate rapid renal clearance of 68Ga-DOTATATE in post-MI mice. (PPTX 478 kb)
Suppl Fig 4
Whole body distribution of 18F-FDG in healthy and post-infarct mice under ketamine/xylazine anaesthesia. Representative 18F-FDG images; left, anterior 3D PET/CT maximum intensity projection in healthy control mice under isoflurane or ketamine-xylazine (KX) anaesthesia; right, parametric myocardial images in healthy mice under isoflurane or with suppression of cardiomyocyte uptake by KX. (PPTX 1380 kb)
Rights and permissions
About this article
Cite this article
Thackeray, J.T., Bankstahl, J.P., Wang, Y. et al. Targeting post-infarct inflammation by PET imaging: comparison of 68Ga-citrate and 68Ga-DOTATATE with 18F-FDG in a mouse model. Eur J Nucl Med Mol Imaging 42, 317–327 (2015). https://doi.org/10.1007/s00259-014-2884-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-014-2884-6