Abstract
Purpose
The aim of the present study was to compare amyloid-β (Aβ) deposition, translocator protein (TSPO) activity, regional cerebral metabolic rate of glucose (rCMRglc), and mitochondrial complex I (MC-I) activity in the brain of aged monkeys.
Methods
PET scans with 11C-PIB (Aβ), 18F-BCPP-EF (MC-I), 11C-DPA-713 (TSPO), and 18F-FDG (rCMRglc) were performed in aged monkeys (Macaca mulatta) in the conscious state and under isoflurane anaesthesia. 11C-PIB binding to Aβ and 11C-DPA-713 binding to TSPO were evaluated in terms of standard uptake values (SUV). The total volume of distribution (V T) of 18F-BCPP-EF and rCMRglc with 18F-FDG were calculated using arterial blood sampling.
Results
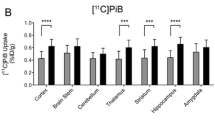
Isoflurane did not affect MC-I activity measured in terms of 18F-BCPP-EF uptake in living brain. There was a significant negative correlation between 18F-BCPP-EF binding (V T) and 11C-PIB uptake (SUVR), and there was a significant positive correlation between 11C-DPA-713 uptake (SUV) and 11C-PIB uptake. In contrast, there was no significant correlation between rCMRglc ratio and 11C-PIB uptake.
Conclusion
18F-BCPP-EF could be a potential PET probe for quantitative imaging of impaired MC-I activity that is correlated with Aβ deposition in the living brain.




Similar content being viewed by others
References
Bratic A, Larsson NG. The role of mitochondria in aging. J Clin Invest. 2013;123:952–7.
Ankarcrona M, Dypbukt JM, Bonfoco E, Zhivotovsky B, Orrenius S, Lipton SA, et al. Glutamate-induced neuronal death: a succession of necrosis or apoptosis depending on mitochondrial function. Neuron. 1995;15:961–73.
Baek BS, Kwon HJ, Lee KH, Yoo MA, Kim KW, Ikeno Y, et al. Regional difference of ROS generation, lipid peroxidation, and antioxidant enzyme activity in rat brain and their dietary modulation. Arch Pharm Res. 1999;22:361–6.
Kannurpatti SS, Sanganahalli BG, Mishra S, Joshi PG, Joshi NB. Glutamate-induced differential mitochondrial response in young and adult rats. Neurochem Int. 2004;44:361–9.
Choi BH. Oxidative stress and Alzheimer's disease. Neurobiol Aging. 1995;16:675–8.
Schon EA, Przedborski S. Mitochondria: the next (neurode)generation. Neuron. 2011;70:1033–53.
Lenaz G, Cavazzoni M, Genova ML, D’Aurelio M, Pich MM, Pallotti F, et al. Oxidative stress, antioxidant defences and aging. Biofactors. 1998;8:195–204.
Robinson BH. Human complex I deficiency: clinical spectrum and involvement of oxygen free radicals in the pathogenicity of the defect. Biochim Biophys Acta. 1998;1364:271–86.
Hardy J, Selkoe DJ. The amyloid hypothesis of Alzheimer’s disease: progress and problems on the road to therapeutics. Science. 2002;297:353–6.
Grundke-Iqbal I, Iqbal K, Tung YC, Quinlan M, Wisniewski HM, Binder LI. Abnormal phosphorylation of the microtubule-associated protein tau (tau) in Alzheimer cytoskeletal pathology. Proc Natl Acad Sci U S A. 1986;83:4913–7.
Monson NL, Ireland SJ, Ligocki AJ, Chen D, Rounds WH, Li M, et al. Elevated CNS inflammation in patients with preclinical Alzheimer’s disease. J Cereb Blood Flow Metab. 2014;34:30–3.
Silva DF, Selfridge JE, Lu J, Lezi E, Cardoso SM, Swerdlow RH. Mitochondrial abnormalities in Alzheimer’s disease: possible targets for therapeutic intervention. Adv Pharmacol. 2012;64:83–126.
Swomley AM, Förster S, Keeney JT, Triplett J, Zhang Z, Sultana R, et al. Abeta, oxidative stress in Alzheimer disease: evidence based on proteomics studies. Biochim Biophys Acta. 2013. doi:10.1016/j.bbadis.2013.09.015
Harada N, Nishiyama S, Kanazawa M, Tsukada H. Development of novel PET probes, [18F]BCPP-EF, [18F]BCPP-BF, and [11C]BCPP-EM for mitochondrial complex I imaging in the living brain. J Labelled Comp Radiopharm. 2013;56:553–61.
Tsukada H, Nishiyama S, Fukumoto D, Kanazawa M, Harada N. Novel PET probes 18F-BCPP-EF and 18F-BCPP-BF for mitochondrial complex I: a PET study by comparison with 18F-BMS-747158-02 in rat brain. J Nucl Med. 2014;55:473–80.
Tsukada H, Ohba H, Kanazawa M, Kakiuchi T, Harada N. Evaluation of 18F-BCPP-EF for mitochondrial complex I imaging in conscious monkey brain using PET. Eur J Nucl Med Mol Imaging. 2014;41:755–63.
Tsukada H, Ohba H, Nishiyama S, Kanazawa M, Kakiuchi T, Harada N. PET imaging of ischemia-induced impairment of mitochondrial complex I function in monkey brain. J Cereb Blood Flow Metab. 2014;34:708–14.
Tsukada H, Miyasato K, Kakiuchi T, Nishiyama S, Harada N, Domino EF. Comparative effects of methamphetamine and nicotine on the striatal [11C]raclopride binding in unanesthetized monkeys. Synapse. 2002;45:207–12.
Noda A, Takamatsu H, Minoshima S, Tsukada H, Nishimura S. Determination of kinetic rate constants for FDG and partition coefficient of water in conscious macaque and alterations in aging or anesthesia examined on parametric images with an anatomic standardization technique. J Cereb Blood Flow Metab. 2003;23:1441–7.
Klunk WE, Engler H, Nordberg A, Wang Y, Blomqvist G, Holt DP, et al. Imaging brain amyloid in Alzheimer’s disease with Pittsburgh Compound-B. Ann Neurol. 2004;55:306–19.
Boutin H, Chauveau F, Thominiaux C, Gregoire MC, James ML, Trebossen R, et al. Receptor PET ligand for in vivo imaging of neuroinflammation. J Nucl Med. 2007;48:573–81.
Oberdorfer F, Hull WE, Traving BC, Maier-Borst W. Synthesis and purification of 2-deoxy-2-[18F]fluoro-D-glucose and 2-deoxy-2-[18F]fluoro-D-mannose: characterization of products by 1H- and 19F-NMR spectroscopy. Int J Rad Appl Instrum A. 1986;37:695–701.
Noda A, Murakami Y, Nishiyama S, Fukumoto D, Miyoshi S, Tsukada H, et al. Amyloid imaging in aged and young macaques with [11C]PIB and [18F]FDDNP. Synapse. 2008;62:472–5.
Jones EG, Stone JM, Karten HJ. High-resolution digital brain atlases: a Hubble telescope for the brain. Ann NY Acad Sci. 2011;1225 Suppl 1:E147–59.
Sokoloff L, Reivich M, Kennedy C, Des Rosiers MH, Patlak CS, Pettigrew KD, et al. The [14C]deoxyglucose method for the measurement of local cerebral glucose utilization: theory, procedure, and normal values in the conscious and anesthetized albino rat. J Neurochem. 1977;28:897–916.
Reivich M, Alavi A, Wolf A, Fowler J, Russell J, Arnett C, et al. Glucose metabolic rate kinetic model parameter determination in humans: the lumped constants and rate constants for [18F]fluorodeoxyglucose and [11C]deoxyglucose. J Cereb Blood Flow Metab. 1985;5:179–92.
Furst AJ, Rabinovici GD, Rostomian AH, Steed T, Alkalay A, Racine C, et al. Cognition, glucose metabolism and amyloid burden in Alzheimer’s disease. Neurobiol Aging. 2010;49:1490–5.
Dukart J, Mueller K, Horstmann A, Vogt B, Frisch S, Barthel H, et al. Differential effects of global and cerebellar normalization on detection and differentiation of dementia in FDG-PET studies. J Cereb Blood Flow Metab. 2005;25:1528–47.
Kumar A, Muzik O, Shandal V, Chugani D, Chakraborty P, Chugani HT. Evaluation of age-related changes in translocator protein (TSPO) in human brain using 11C-[R]-PK11195 PET. J Neuroinflammation. 2012;9:232.
Hanley PJ, Ray J, Brandt U, Daut J. Halothane, isoflurane and sevoflurane inhibit NADH:ubiquinone oxidoreductase (complex I) of cardiac mitochondria. J Physiol. 2002;544(3):687–93.
Bains R, Moe MC, Vinje ML, Berg-Johnsen J. Isoflurane-induced depolarization of neural mitochondria increases with age. Acta Anaesthesiol Scand. 2009;53:85–92.
Nicholls DG, Budd SL. Mitochondria and neuronal survival. Physiol Rev. 2000;80:315–60.
Logan J, Volkow ND, Fowler JS, Wang G-J, Dewey SL, MacGregor R, et al. Effects of blood flow on [11C]raclopride binding in the brain: model simulations and kinetic analysis of PET data. J Cereb Blood Flow Metab. 1994;14:995–1010.
Finch CE, Austad SN. Primate aging in the mammalian scheme: the puzzle of extreme variation in brain aging. Age. 2012;34:1075–91.
Kemppainen NM, Aalto S, Wilson LA, Nagren K, Helin S, Bruck A, et al. PET amyloid ligand [11C]PIB uptake is increased in mild cognitive impairment. Neurology. 2007;68:1603–6.
Mosconi L, Andrews RD, Matthews DC. Comparing brain amyloid deposition, glucose metabolism, and atrophy in mild cognitive impairment with and without a family history of dementia. J Alzheimers Dis. 2013;35:509–24.
Edison P, Archer HA, Hinz R, Hammers A, Pavese N, Tai YF, et al. Amyloid, hypometabolism, and cognition in Alzheimer disease: an [11C]PIB and [18F]FDG PET study. Neurology. 2007;68:501–8.
Warburg O. On respiratory impairment in cancer cells. Science. 1956;124:269–70.
Winkeler A, Boisgard R, Martin A, Tavitian B. Radioisotopic imaging of neuroinflammation. J Nucl Med. 2010;51:1–4.
Navarro A, Boveris A. The mitochondrial energy transduction system and the aging process. Am J Physiol Cell Physiol. 2007;292:C670–86.
Manczak M, Jung Y, Park BS, Partovi D, Reddy PH. Time-course of mitochondrial gene expressions in mice brains: implications for mitochondrial dysfunction, oxidative damage, and cytochrome c in aging. J Neurochem. 2005;92:494–504.
Acknowledgements
We gratefully acknowledge the technical assistance provided by Kaori Suzuki and Aiko Iwazaki.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tsukada, H., Nishiyama, S., Ohba, H. et al. Comparing amyloid-β deposition, neuroinflammation, glucose metabolism, and mitochondrial complex I activity in brain: a PET study in aged monkeys. Eur J Nucl Med Mol Imaging 41, 2127–2136 (2014). https://doi.org/10.1007/s00259-014-2821-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-014-2821-8