Abstract
Purpose
Over recent decades interest in diagnosis and treatment of neuroendocrine tumours (NET) has steadily grown. The basis for diagnosis and therapy of NET with radiolabelled somatostatin (hsst) analogues is the variable overexpression of hsst receptors (hsst1–5 receptors). We hypothesized that radiometal derivatives of DOTA-iodo-Tyr3-octreotide analogues might be excellent candidates for somatostatin receptor imaging. We therefore explored the diagnostic potential of 68Ga-DOTA-iodo-Tyr3-octreotate [68Ga-DOTA,3-iodo-Tyr3,Thr8]octreotide (68Ga-HA-DOTATATE; HA, high-affinity) compared to the established 68Ga-DOTA-Tyr3-octreotate (68Ga-DOTATATE) in vivo.
Methods
The study included 23 patients with known somatostatin receptor-positive metastases from NETs, thyroid cancer or glomus tumours who were investigated with both 68Ga-HA-DOTATATE and 68Ga-DOTATATE. A patient-based and a lesion-based comparative analysis was carried out of normal tissue distribution and lesion detectability in a qualitative and a semiquantitative manner.
Results
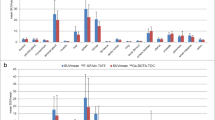
68Ga-HA-DOTATATE and 68Ga-DOTATATE showed comparable uptake in the liver (SUVmean 8.9 ± 2.2 vs. 9.3 ± 2.5, n.s.), renal cortex (SUVmean 13.3 ± 3.9 vs. 14.5 ± 3.7, n.s.) and spleen (SUVmean 24.0 ± 6.7 vs. 22.9 ± 7.3, n.s.). A somewhat higher pituitary uptake was found with 68Ga-HA-DOTATATE (SUVmean 6.3 ± 1.8 vs. 5.4 ± 2.1, p < 0.05). On a lesion-by-lesion basis a total of 344 lesions were detected. 68Ga-HA-DOTATATE demonstrated 328 lesions (95.3 % of total lesions seen), and 68Ga-DOTATATE demonstrated 332 lesions (96.4 %). The mean SUVmax of all lesions was not significantly different between 68Ga-HA-DOTATATE and 68Ga-DOTATATE (17.8 ± 11.4 vs. 16.7 ± 10.7, n.s.).
Conclusion
Our analysis demonstrated very good concordance between 68Ga-HA-DOTATATE and 68Ga-DOTATATE PET data. As the availability and use of 68Ga-HA-DOTATATE is not governed by patent restrictions it may be an attractive alternative to other 68Ga-labelled hsst analogues.





Similar content being viewed by others
References
Atkinson H, England JA, Rafferty A, Jesudason V, Bedford K, Karsai L, et al. Somatostatin receptor expression in thyroid disease. Int J Exp Pathol. 2013;94:226–9. doi:10.1111/iep.12024.
Kaemmerer D, Peter L, Lupp A, Schulz S, Sanger J, Prasad V, et al. Molecular imaging with 68Ga-SSTR PET/CT and correlation to immunohistochemistry of somatostatin receptors in neuroendocrine tumours. Eur J Nucl Med Mol Imaging. 2011;38:1659–68. doi:10.1007/s00259-011-1846-5.
Lamberts SW, Bakker WH, Reubi JC, Krenning EP. Somatostatin-receptor imaging in the localization of endocrine tumors. N Engl J Med. 1990;323:1246–9.
Kwekkeboom DJ, Bakker WH, Kam BL, Teunissen JJ, Kooij PP, de Herder WW, et al. Treatment of patients with gastro-entero-pancreatic (GEP) tumours with the novel radiolabelled somatostatin analogue [177Lu-DOTA(0), Tyr3]octreotate. Eur J Nucl Med Mol Imaging. 2003;30:417–22. doi:10.1007/s00259-002-1050-8.
Krenning EP, Bakker WH, Breeman WA, Koper JW, Kooij PP, Ausema L, et al. Localisation of endocrine-related tumours with radioiodinated analogue of somatostatin. Lancet. 1989;1:242–4.
Virgolini I, Ambrosini V, Bomanji JB, Baum RP, Fanti S, Gabriel M, et al. Procedure guidelines for PET/CT tumour imaging with 68Ga-DOTA-conjugated peptides: 68Ga-DOTA-TOC, 68Ga-DOTA-NOC, 68Ga-DOTA-TATE. Eur J Nucl Med Mol Imaging. 2010;37:2004–10. doi:10.1007/s00259-010-1512-3.
Reubi JC, Schar JC, Waser B, Wenger S, Heppeler A, Schmitt JS, et al. Affinity profiles for human somatostatin receptor subtypes SST1-SST5 of somatostatin radiotracers selected for scintigraphic and radiotherapeutic use. Eur J Nucl Med. 2000;27:273–82.
Ginj M, Schmitt JS, Chen J, Waser B, Reubi JC, de Jong M, et al. Design, synthesis, and biological evaluation of somatostatin-based radiopeptides. Chem Biol. 2006;13:1081–90. doi:10.1016/j.chembiol.2006.08.012.
Brogsitter C, Schottelius M, Zophel K, Kotzerke J, Wester HJ. Twins in spirit: DOTATATE and high-affinity DOTATATE. Eur J Nucl Med Mol Imaging. 2013;40:1789. doi:10.1007/s00259-013-2497-5.
Krenning EP, Bakker WH, Kooij PP, Breeman WA, Oei HY, de Jong M, et al. Somatostatin receptor scintigraphy with indium-111-DTPA-D-Phe-1-octreotide in man: metabolism, dosimetry and comparison with iodine-123-Tyr-3-octreotide. J Nucl Med. 1992;33:652–8.
Buchmann I, Henze M, Engelbrecht S, Eisenhut M, Runz A, Schafer M, et al. Comparison of 68Ga-DOTATOC PET and 111In-DTPAOC (Octreoscan) SPECT in patients with neuroendocrine tumours. Eur J Nucl Med Mol Imaging. 2007;34:1617–26. doi:10.1007/s00259-007-0450-1.
Hartmann H, Zophel K, Freudenberg R, Oehme L, Andreeff M, Wunderlich G, et al. Radiation exposure of patients during 68Ga-DOTATOC PET/CT examinations. Nuklearmedizin. 2009;48:201–7. doi:10.3413/nukmed-0214.
Kowalski J, Henze M, Schuhmacher J, Macke HR, Hofmann M, Haberkorn U. Evaluation of positron emission tomography imaging using [68Ga]-DOTA-D Phe(1)-Tyr(3)-Octreotide in comparison to [111In]-DTPAOC SPECT. First results in patients with neuroendocrine tumors. Mol Imaging Biol. 2003;5:42–8.
Virgolini IJ, Gabriel M, von Guggenberg E, Putzer D, Kendler D, Decristoforo C. Role of radiopharmaceuticals in the diagnosis and treatment of neuroendocrine tumours. Eur J Cancer. 2009;45 Suppl 1:274–91. doi:10.1016/S0959-8049(09)70042-5.
Ben-Shlomo A, Melmed S. Pituitary somatostatin receptor signaling. Trends Endocrinol Metab. 2010;21:123–33. doi:10.1016/j.tem.2009.12.003.
Hofland LJ, Feelders RA, de Herder WW, Lamberts SW. Pituitary tumours: the sst/D2 receptors as molecular targets. Mol Cell Endocrinol. 2010;326:89–98. doi:10.1016/j.mce.2010.04.020.
Kabasakal L, Demirci E, Ocak M, Decristoforo C, Araman A, Ozsoy Y, et al. Comparison of 68Ga-DOTATATE and 68Ga-DOTANOC PET/CT imaging in the same patient group with neuroendocrine tumours. Eur J Nucl Med Mol Imaging. 2012;39:1271–7.
Poeppel TD, Binse I, Petersenn S, Lahner H, Schott M, Antoch G, et al. 68Ga-DOTATOC versus 68Ga-DOTATATE PET/CT in functional imaging of neuroendocrine tumors. J Nucl Med. 2011;52:1864–70.
Wild D, Bomanji JB, Benkert P, Maecke H, Ell PJ, Reubi JC, et al. Comparison of 68Ga-DOTANOC and 68Ga-DOTATATE PET/CT within patients with gastroenteropancreatic neuroendocrine tumors. J Nucl Med. 2013;54:364–72.
Putzer D, Kroiss A, Waitz D, Gabriel M, Traub-Weidinger T, Uprimny C, et al. Somatostatin receptor PET in neuroendocrine tumours: 68Ga-DOTA0, Tyr3-octreotide versus 68Ga-DOTA0-lanreotide. Eur J Nucl Med Mol Imaging. 2013;40:364–72. doi:10.1007/s00259-012-2286-6.
Demirci E, Ocak M, Kabasakal L, Araman A, Ozsoy Y, Kanmaz B. Comparison of Ga-68 DOTA-TATE and Ga-68 DOTA-LAN PET/CT imaging in the same patient group with neuroendocrine tumours: preliminary results. Nucl Med Commun. 2013;34:727–32. doi:10.1097/MNM.0b013e328362cca6.
Naswa N, Sharma P, Suman Kc S, Lata S, Kumar R, Malhotra A, et al. Prospective evaluation of 68Ga-DOTA-NOC PET-CT in patients with recurrent medullary thyroid carcinoma: comparison with 18F-FDG PET-CT. Nucl Med Commun. 2012;33:766–74. doi:10.1097/MNM.0b013e3283541157.
Shastry M, Kayani I, Wild D, Caplin M, Visvikis D, Gacinovic S, et al. Distribution pattern of 68Ga-DOTATATE in disease-free patients. Nucl Med Commun. 2010;31:1025–32.
Vanhagen PM, Krenning EP, Reubi JC, Kwekkeboom DJ, Bakker WH, Mulder AH, et al. Somatostatin analogue scintigraphy in granulomatous diseases. Eur J Nucl Med. 1994;21:497–502.
Mussig K, Oksuz MO, Pfannenberg C, Adam P, Zustin J, Beckert S, et al. Somatostatin receptor expression in an epitheloid hemangioma causing oncogenic osteomalacia. J Clin Endocrinol Metab. 2009;94:4123–4.
Winter PF, Lapke J, Winek R. Subcutaneous cavernous hemangioma visualized on an indium-111-octreotide scan. J Nucl Med. 1996;37:1516–7.
Conflicts of interest
H.J. Wester is CEO of SCINTOMICS, Germany, but without any conflicts of interest. The other authors declare no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Brogsitter, C., Zöphel, K., Hartmann, H. et al. Twins in spirit part II: DOTATATE and high-affinity DOTATATE—the clinical experience. Eur J Nucl Med Mol Imaging 41, 1158–1165 (2014). https://doi.org/10.1007/s00259-014-2690-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-014-2690-1