Abstract
Purpose
Current understanding of cerebral FDG uptake during childhood originates from a small number of studies in patients with neurological abnormalities. Our aim was to describe cerebral FDG uptake in a dataset of FDG PET scans in children more likely to represent a normal population.
Methods
We reviewed cerebral FDG PET scans in children up to 16 years of age with suspected/proven extracranial malignancies and the following exclusions: central nervous system metastases, previous malignancies, previous chemotherapy or radiotherapy, development of cerebral metastases during therapy, neurological conditions, taking antiepileptic medication or medications likely to interfere with cerebral metabolism, and general anaesthesia within 24 h. White matter, basal ganglia, thalamus and the cerebellar cortex were analysed using regional SUVmax, and the cerebral cortex, basal ganglia, thalamus and cerebellum were analysed using a regional relative uptake analysis in comparison to maximal cortical uptake.
Results
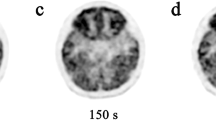
Scans from 30 patients (age range 11 months to 16 years, mean age 10 years 5 months) were included. All regions showed increasing SUVmax with age. The parietal, occipital, lateral temporal and medial temporal lobes showed lower rates of increasing FDG uptake causing changing patterns of regional FDG uptake during childhood. The cortical regions showing the most intense uptake in early childhood were the parietal and occipital lobes. At approximately 7 years of age these regions had relatively less uptake than the frontal lobes and at approximately 10 years of age these regions had relatively less uptake than the thalamus.
Conclusion
Relative FDG uptake in the brain has not reached an adult pattern by 1 year of age, but continues to change up to 16 years of age. The changing pattern is due to different regional rates of increasing cortical FDG uptake, which is less rapid in the parietal, occipital and temporal lobes than in the frontal lobes.







Similar content being viewed by others
References
Van Bogaert P, Wikler D, Damhaut P, Szliwowski HB, Goldman S. Regional changes in glucose metabolism during brain development from the age of 6 years. Neuroimage. 1998;8(1):62–8.
Wang GJ, Volkow ND, Wolf AP, Brodie JD, Hitzemann RJ. Intersubject variability of brain glucose metabolic measurements in young normal males. J Nucl Med. 1994;35(9):1457–66.
Loessner A, Alavi A, Lewandrowski KU, Mozley D, Souder E, Gur RE. Regional cerebral function determined by FDG-PET in healthy volunteers: normal patterns and changes with age. J Nucl Med. 1995;36(7):1141–9.
Chugani HT, Phelps ME. Maturational changes in cerebral function in infants determined by 18FDG positron emission tomography. Science. 1986;231(4740):840–3.
Chugani HT, Phelps ME, Mazziotta JC. Positron emission tomography study of human brain functional development. Ann Neurol. 1987;22(4):487–97.
Kinnala A, Suhonen-Polvi H, Aarimaa T, Kero P, Korvenranta H, Ruotsalainen U, et al. Cerebral metabolic rate for glucose during the first six months of life: an FDG positron emission tomography study. Arch Dis Child Fetal Neonatal Ed. 1996;74(3):F153–7.
Shi Y, Jin RB, Zhao JN, Tang SF, Li HQ, Li TY, et al. Brain positron emission tomography in preterm and term newborn infants. Early Hum Dev. 2009;85(7):429–32.
Gaillard WD, Zeffiro T, Fazilat S, DeCarli C, Theodore WH. Effect of valproate on cerebral metabolism and blood flow: an 18F-2-deoxyglucose and 15O water positron emission tomography study. Epilepsia. 1996;37(6):515–21.
Theodore WH, Bromfield E, Onorati L. The effect of carbamazepine on cerebral glucose metabolism. Ann Neurol. 1989;25(5):516–20.
Theodore WH, DiChiro G, Margolin R, Fishbein D, Porter RJ, Brooks RA. Barbiturates reduce human cerebral glucose metabolism. Neurology. 1986;36(1):60–4.
Ramani R, Wardhan R. Understanding anesthesia through functional imaging. Curr Opin Anaesthesiol. 2008;21(5):530–6. doi:10.1097/ACO.0b013e32830edbf3.
Takahashi T, Shirane R, Sato S, Yoshimoto T. Developmental changes of cerebral blood flow and oxygen metabolism in children. AJNR Am J Neuroradiol. 1999;20(5):917–22.
Nariai T, Matsushima Y, Imae S, Tanaka Y, Ishii K, Senda M, et al. Severe haemodynamic stress in selected subtypes of patients with moyamoya disease: a positron emission tomography study. J Neurol Neurosurg Psychiatry. 2005;76(5):663–9.
Chiron C, Raynaud C, Maziere B, Zilbovicius M, Laflamme L, Masure MC, et al. Changes in regional cerebral blood flow during brain maturation in children and adolescents. J Nucl Med. 1992;33(5):696–703.
Ogawa A, Nakamura N, Sugita K, Sakurai Y, Kayama T, Wada T, et al. [Regional cerebral blood flow in children––normal value and regional distribution of cerebral blood flow in childhood]. No To Shinkei. 1987;39(2):113–8.
Powers WJ, Rosenbaum JL, Dence CS, Markham J, Videen TO. Cerebral glucose transport and metabolism in preterm human infants. J Cereb Blood Flow Metab. 1998;18(6):632–8.
Rubinstein M, Denays R, Ham HR, Piepsz A, VanPachterbeke T, Haumont D, et al. Functional imaging of brain maturation in humans using iodine-123 iodoamphetamine and SPECT. J Nucl Med. 1989;30(12):1982–5.
Schiepers C, Verbruggen A, Casaer P, De Roo M. Normal brain perfusion pattern of technetium-99m-ethylcysteinate dimer in children. J Nucl Med. 1997;38(7):1115–20.
Chugani HT. A critical period of brain development: studies of cerebral glucose utilization with PET. Prev Med. 1998;27(2):184–8.
Conklin HM, Luciana M, Hooper CJ, Yarger RS. Working memory performance in typically developing children and adolescents: behavioral evidence of protracted frontal lobe development. Dev Neuropsychol. 2007;31(1):103–28.
Gogtay N, Giedd J, Rapoport JL. Brain development in healthy, hyperactive, and psychotic children. Arch Neurol. 2002;59(8):1244–8.
Wilke M, Krageloh-Mann I, Holland SK. Global and local development of gray and white matter volume in normal children and adolescents. Exp Brain Res. 2007;178(3):296–307.
Huttenlocher PR, de Courten C. The development of synapses in striate cortex of man. Hum Neurobiol. 1987;6(1):1–9.
Paolicelli RC, Bolasco G, Pagani F, Maggi L, Scianni M, Panzanelli P, et al. Synaptic pruning by microglia is necessary for normal brain development. Science. 2011;333(6048):1456–8.
Giedd JN, Blumenthal J, Jeffries NO, Castellanos FX, Liu H, Zijdenbos A, et al. Brain development during childhood and adolescence: a longitudinal MRI study. Nat Neurosci. 1999;2(10):861–3.
Keyes Jr JW. SUV: standard uptake or silly useless value? J Nucl Med. 1995;36(10):1836–9.
Acknowledgments
The authors would like to acknowledge the vital assistance of Prof. Roger Uren and Dr. David Chung in the blinded reading of the scans for the relative uptake analysis. We would also acknowledge the funding assistance of an Australian National Health and Medical Research Council Biomedical Scholarship, and a New South Wales Cancer Institute Research Scholar Award.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
London, K., Howman-Giles, R. Normal cerebral FDG uptake during childhood. Eur J Nucl Med Mol Imaging 41, 723–735 (2014). https://doi.org/10.1007/s00259-013-2639-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-013-2639-9