Abstract
Purpose
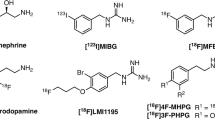
Both 131I- and 123I-labeled meta-iodobenzylguanidine (MIBG) have been widely used in the clinic for targeted imaging of the norepinephrine transporter (NET). The human NET (hNET) gene has been imaged successfully with 124I-MIBG positron emission tomography (PET) at time points of >24 h post-injection (p.i.). 18F-labeled MIBG analogs may be ideal to image hNET expression at time points of <8 h p.i. We developed improved methods for the synthesis of known MIBG analogs, [18F]MFBG and [18F]PFBG and evaluated them in hNET reporter gene-transduced C6 rat glioma cells and xenografts.
Methods
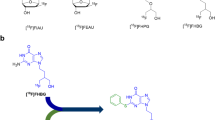
[18F]MFBG and [18F]PFBG were synthesized manually using a three-step synthetic scheme. Wild-type and hNET reporter gene-transduced C6 rat glioma cells and xenografts were used to comparatively evaluate the 18F-labeled analogs with [123I]/[124I]MIBG.
Results
The fluorination efficacy on benzonitrile was predominantly determined by the position of the trimethylammonium group. The para-isomer afforded higher yields (75 ± 7 %) than meta-isomer (21 ± 5 %). The reaction of [18F]fluorobenzylamine with 1H-pyrazole-1-carboximidamide was more efficient than with 2-methyl-2-thiopseudourea. The overall radiochemical yields (decay-corrected) were 11 ± 2 % (n = 12) for [18F]MFBG and 41 ± 12 % (n = 5) for [18F]PFBG, respectively. The specific uptakes of [18F]MFBG and [18F]PFBG were similar in C6-hNET cells, but 4-fold less than that of [123I]/[124I]MIBG. However, in vivo [18F]MFBG accumulation in C6-hNET tumors was 1.6-fold higher than that of [18F]PFBG at 1 h p.i., whereas their uptakes were similar at 4 h. Despite [18F]MFBG having a 2.8-fold lower affinity to hNET and approximately 4-fold lower cell uptake in vitro compared to [123I]/[124I]MIBG, PET imaging demonstrated that [18F]MFBG was able to visualize C6-hNET xenografts better than [124I]MIBG. Biodistribution studies showed [18F]MFBG and 123I-MIBG had a similar tumor accumulation, which was lower than that of no-carrier-added [124I]MIBG, but [18F]MFBG showed a significantly more rapid body clearance and lower uptake in most non-targeting organs.
Conclusion
[18F]MFBG and [18F]PFBG were synthesized in reasonable radiochemical yields under milder conditions. [18F]MFBG is a better PET ligand to image hNET expression in vivo at 1–4 h p.i. than both [18F]PFBG and [123I]/[124I]MIBG.




Similar content being viewed by others
Abbreviations
- NET:
-
Norepinephrine transporter
- hNET:
-
Human norepinephrine transporter
- MIBG:
-
Meta-iodobenzylguanidine
- [18F]MFBG:
-
Meta-[18F]fluorobenzylguanidine
- [18F]PFBG:
-
Para-[18F]fluorobenzylguanidine
- SPECT:
-
Single photon emission computed tomography
- PET:
-
Positron emission tomography
- [76Br]MBBG:
-
Meta-[76Br]bromobenzylguanidine
- [18F]FIBG:
-
4-[18F]fluoro-3-iodobenzylguanidine
- LMI1195:
-
N-[3-bromo-4-(3-(18)F-fluoro-propoxy)-benzyl]-guanidine
- C6-WT:
-
C6 wild-type rat glioma cells
- C6-hNET:
-
hNET gene stably transduced C6 cells
- PIBG:
-
Para-iodobenzylguanidine
- TBA[18F]F:
-
Tetrabutylammonium [18F]fluoride
- MeCN:
-
acetonitrile
- TFA:
-
Trifluoroacetic acid
- PBS:
-
Phosphate-buffered saline
- ROI:
-
Region of interest
References
Pacholczyk T, Blakely RD, Amara SG. Expression cloning of a cocaine- and antidepressant-sensitive human noradrenaline transporter. Nature 1991;350:350–4.
Axelrod J, Kopin IJ. The uptake, storage, release and metabolism of noradrenaline in sympathetic nerves. Prog Brain Res 1969;31:21–32.
Anton M, Wagner B, Haubner R, Bodenstein C, Essien BE, Bönisch H, et al. Use of the norepinephrine transporter as a reporter gene for non-invasive imaging of genetically modified cells. J Gene Med 2004;6:119–26.
Moroz MA, Serganova I, Zanzonico P, Ageyeva L, Beresten T, Dyomina E, et al. Imaging hNET reporter gene expression with 124I-MIBG. J Nucl Med 2007;48:827–36.
Grünwald F, Ezziddin S. 131I-metaiodobenzylguanidine therapy of neuroblastoma and other neuroendocrine tumors. Semin Nucl Med 2010;40:153–63.
Brisse HJ, McCarville MB, Granata C, Krug KB, Wootton-Gorges SL, Kanegawa K, et al. Guidelines for imaging and staging of neuroblastic tumors: consensus report from the International Neuroblastoma Risk Group Project. Radiology 2011;261:243–57.
Pryma D, Divgi C. Meta-iodobenzyl guanidine for detection and staging of neuroendocrine tumors. Nucl Med Biol 2008;35:5.
Jacobson AF, Deng H, Lombard J, Lessig HJ, Black RR. 123I-meta-iodobenzylguanidine scintigraphy for the detection of neuroblastoma and pheochromocytoma: results of a meta-analysis. J Clin Endocrinol Metab 2010;95:2596–606.
Fonte JS, Robles JF, Chen CC, Reynolds J, Whatley M, Ling A, et al. False-negative 123I-MIBG SPECT is most commonly found in SDHB-related pheochromocytoma or paraganglioma with high frequency to develop metastatic disease. Endocr Relat Cancer 2012;19:83–93.
Vaidyanathan G. Meta-iodobenzylguanidine and analogues: chemistry and biology. Q J Nucl Med Mol Imaging 2008;52:351–68.
Lee CL, Wahnishe H, Sayre GA, Cho HM, Kim HJ, Hernandez-Pampaloni M, et al. Radiation dose estimation using preclinical imaging with 124I-metaiodobenzylguanidine (MIBG) PET. Med Phys 2010;37:4861–7.
Watanabe S, Hanaoka H, Liang JX, Iida Y, Endo K, Ishioka NS. PET imaging of norepinephrine transporter-expressing tumors using 76Br-meta-bromobenzylguanidine. J Nucl Med 2010;51:1472–9.
Garg PK, Garg S, Zalutsky MR. Synthesis and preliminary evaluation of para- and meta-[18F]fluorobenzylguanidine. Nucl Med Biol 1994;21:97–103.
Berry CR, Garg PK, Zalutsky MR, Coleman RE, DeGrado TR. Uptake and retention kinetics of para-fluorine-18-fluorobenzylguanidine in isolated rat heart. J Nucl Med 1996;37:2011–6.
Berry CR, DeGrado TR, Nutter F, Garg PK, Breitschwerdt EB, Spaulding K, et al. Imaging of pheochromocytoma in 2 dogs using p-[18F] fluorobenzylguanidine. Vet Radiol Ultrasound 2002;43:183–6.
Vaidyanathan G, Affleck DJ, Zalutsky MR. (4-[18F]fluoro-3-iodobenzyl)guanidine, a potential MIBG analogue for positron emission tomography. J Med Chem 1994;37:3655–62.
Vaidyanathan G, Affleck DJ, Zalutsky MR. Validation of 4-[fluorine-18]fluoro-3-iodobenzylguanidine as a positron-emitting analog of MIBG. J Nucl Med 1995;36:644–50.
Lee BC, Paik JY, Chi DY, Lee KH, Choe YS. Potential and practical adrenomedullary PET radiopharmaceuticals as an alternative to m-iodobenzylguanidine: m-(omega-[18F]fluoroalkyl)benzylguanidines. Bioconjug Chem 2004;15:104–11.
Rischpler C, Fukushima K, Isoda T, Javadi MS, Dannals RF, Abraham R, et al. Discrepant uptake of the radiolabeled norepinephrine analogues hydroxyephedrine (HED) and metaiodobenzylguanidine (MIBG) in rat hearts. Eur J Nucl Med Mol Imaging 2013;40:1077–83.
Yu M, Bozek J, Lamoy M, Guaraldi M, Silva P, Kagan M, et al. Evaluation of LMI1195, a novel 18F-labeled cardiac neuronal PET imaging agent, in cells and animal models. Circ Cardiovasc Imaging 2011;4:435–43.
Vaidyanathan G, Affleck DJ, Alston KL, Zalutsky MR. A tin precursor for the synthesis of no-carrier-added [*I]MIBG and [211At]MABG. J Labelled Comp Radiopharm 2007;50:177–82.
Ohara K, Vasseur JJ, Smietana M. NIS-promoted guanylation of amines. Tetrahedron Lett 2009;50:1463–5.
Zhang H, Huang R, Lewis JS, Blasberg RG. Imaging human norepinephrine transporter (hNET) expressing reporter cells and tumors with 4-18F-fluorobenzylguanidine. J Nucl Med 2012;53:1584.
Howman-Giles R, Shaw PJ, Uren RF, Chung DK. Neuroblastoma and other neuroendocrine tumors. Semin Nucl Med 2007;37:286–302.
Travin MI. Cardiac neuronal imaging at the edge of clinical application. Cardiol Clin 2009;27:311–27.
Vaidyanathan G, Zalutsky MR. No-carrier-added meta-[123I]iodobenzylguanidine: synthesis and preliminary evaluation. Nucl Med Biol 1995;22:61–4.
Acknowledgments
This work was supported by NIH grant P50 CA84638. We appreciate the assistance of staff from the MSKCC Small Animal Imaging Core Facility for the PET imaging and the MSKCC Radiochemistry & Molecular Imaging Probe Core for 18F production. These MSKCC Cores were supported by the NIH Center grant P30 CA08748. DLJT was supported by the R25T Molecular Imaging Fellowship: Molecular Imaging Training in Oncology (5R25CA096945-07; Principal investigator H. Hricak). We also thank Dr. Diane Abou for PET imaging assistance and valuable discussion. Dedicated to Prof. H. Maecke on the occasion of his 70th birthday.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 167 kb)
Rights and permissions
About this article
Cite this article
Zhang, H., Huang, R., Pillarsetty, N. et al. Synthesis and evaluation of 18F-labeled benzylguanidine analogs for targeting the human norepinephrine transporter. Eur J Nucl Med Mol Imaging 41, 322–332 (2014). https://doi.org/10.1007/s00259-013-2558-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-013-2558-9