Abstract
Purpose
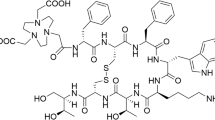
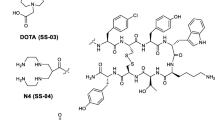
Targeting of tumours positive for somatostatin receptors (sst) with radiolabelled peptides is of interest for tumour localization, staging, therapy follow-up and targeted radionuclide therapy. The peptides used clinically are exclusively agonists, but recently we have shown that the radiolabelled somatostatin-based antagonist 111In-DOTA-sst2-ANT may be preferable to agonists. However, a comprehensive study of this radiolabelled antagonist to determine its significance was lacking. The present report describes the evaluation of this novel antagonist labelled with 111In and 177Lu in three different tumour models.
Methods
Radiopeptide binding, internalization and dissociation studies were performed using cells expressing HEK293-rsst2. Biodistribution studies were performed in HEK293-rsst2, HEK293-hsst2 and HEK293-rsst3 xenografted mice.
Results
Saturation binding analysis confirmed earlier IC50 data for 111/natIn-DOTA-sst2-ANT and showed similar affinity of 177/natLu-DOTA-sst2-ANT for the sst2. Only low internalization was found in cell culture (6.68 ± 0.06 % at 4 h), which was not unexpected for an antagonist, and this could be further reduced by the addition of sucrose. No internalization was observed in HEK293 cells not expressing sst. Both results indicate that the internalization was specific. 111In-DOTA-sst2-ANT and 177Lu-DOTA-sst2-ANT were shown to target tumour xenografts expressing the rat and the human sst2 receptor with no differences in their uptake or pharmacokinetics. The uptake in rsst2 and hsst2 was high (about 30 %IA/g 4 h after injection) and surprisingly long-lasting (about 20–23 %IA/g 24 h after injection). Kidney uptake was blocked by approximately 50 % by lysine or Gelofusine.
Conclusion
These results indicate that radiolabelled somatostatin-based antagonists may be superior to corresponding agonists. The long tumour retention time of 177Lu-DOTA-sst2-ANT indicates that this new class of compounds is of relevance not only in diagnostic imaging but also in targeted radionuclide therapy of sst-positive tumours.





Similar content being viewed by others
Abbreviations
- HEK:
-
Human embryonic kidney
- DOTA:
-
1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid
- CB-TE2A:
-
4,11-bis(carboxymethyl)-1,4,8,11-tetraazabicyclo[6.6.2]hexadecane
- DMEM:
-
Dulbecco’s modified Eagle’s medium
- BSA:
-
Bovine serum albumin
- FBS:
-
Fetal bovine serum
- PBS:
-
Phosphate-buffered saline
- HEPES:
-
2-[4-(2-Hydroxyethyl)piperazin-1-yl]ethanesulphonic acid
References
Reubi JC, Maecke HR. Peptide-based probes for cancer imaging. J Nucl Med. 2008;49:1735–8.
Reubi JC. Peptide receptors as molecular targets for cancer diagnosis and therapy. Endocr Rev. 2003;24:389–427.
Storch D, Behe M, Walter MA, Chen J, Powell P, Mikolajczak R, et al. Evaluation of [99mTc/EDDA/HYNIC0]octreotide derivatives compared with [111In-DOTA0, Tyr3, Thr8]octreotide and [111In-DTPA0]octreotide: does tumor or pancreas uptake correlate with the rate of internalization? J Nucl Med. 2005;46:1561–9.
Fani M, Mueller A, Tamma ML, Nicolas G, Rink HR, Cescato R, et al. Radiolabeled bicyclic somatostatin-based analogs: a novel class of potential radiotracers for SPECT/PET of neuroendocrine tumors. J Nucl Med. 2010;51:1771–9.
Ginj M, Zhang H, Eisenwiener KP, Wild D, Schulz S, Rink H, et al. New pansomatostatin ligands and their chelated versions: affinity profile, agonist activity, internalization, and tumor targeting. Clin Cancer Res. 2008;14:2019–27.
Ginj M, Zhang H, Waser B, Cescato R, Wild D, Wang X, et al. Radiolabeled somatostatin receptor antagonists are preferable to agonists for in vivo peptide receptor targeting of tumors. Proc Natl Acad Sci U S A. 2006;103:16436–41.
Wadas TJ, Eiblmaier M, Zheleznyak A, Sherman CD, Ferdani R, Liang K, et al. Preparation and biological evaluation of 64Cu-CB-TE2A-sst2-ANT, a somatostatin antagonist for PET imaging of somatostatin receptor-positive tumors. J Nucl Med. 2008;49:1819–27.
Maina T, Nock BA, Zhang H, Nikolopoulou A, Waser B, Reubi JC, et al. Species differences of bombesin analog interactions with GRP-R define the choice of animal models in the development of GRP-R-targeting drugs. J Nucl Med. 2005;46:823–30.
Cescato R, Erchegyi J, Waser B, Piccand V, Maecke HR, Rivier JE, et al. Design and in vitro characterization of highly sst2-selective somatostatin antagonists suitable for radiotargeting. J Med Chem. 2008;51:4030–7.
Ginj M, Schmitt JS, Chen J, Waser B, Reubi JC, de Jong M, et al. Design, synthesis, and biological evaluation of somatostatin-based radiopeptides. Chem Biol. 2006;13:1081–90.
Maecke HR, Reubi JC. Somatostatin receptors as targets for nuclear medicine imaging and radionuclide treatment. J Nucl Med. 2011;52:841–4.
Imhof A, Brunner P, Marincek N, Briel M, Schindler C, Rasch H, et al. Response, survival, and long-term toxicity after therapy with the radiolabeled somatostatin analogue [90Y-DOTA]-TOC in metastasized neuroendocrine cancers. J Clin Oncol. 2011;29:2416–23.
Kwekkeboom DJ, de Herder WW, Kam BL, van Eijck CH, van Essen M, Kooij PP, et al. Treatment with the radiolabeled somatostatin analog [177Lu-DOTA0,Tyr3]octreotate: toxicity, efficacy, and survival. J Clin Oncol. 2008;26:2124–30.
Weckbecker G, Lewis I, Albert R, Schmid HA, Hoyer D, Bruns C. Opportunities in somatostatin research: biological, chemical and therapeutic aspects. Nat Rev Drug Discov. 2003;2:999–1017.
Meyerhof W. The elucidation of somatostatin receptor functions: a current view. Rev Physiol Biochem Pharmacol. 1998;133:55–108.
Fani M, Del Pozzo L, Abiraj K, Mansi R, Tamma ML, Cescato R, et al. PET of somatostatin receptor-positive tumors using 64Cu- and 68Ga-somatostatin antagonists: the chelate makes the difference. J Nucl Med. 2011;52:1110–8.
van Eerd JE, Vegt E, Wetzels JF, Russel FG, Masereeuw R, Corstens FH, et al. Gelatin-based plasma expander effectively reduces renal uptake of 111In-octreotide in mice and rats. J Nucl Med. 2006;47:528–33.
Vegt E, Wetzels JF, Russel FG, Masereeuw R, Boerman OC, van Eerd JE, et al. Renal uptake of radiolabeled octreotide in human subjects is efficiently inhibited by succinylated gelatin. J Nucl Med. 2006;47:432–6.
Nguyen K, Parry JJ, Rogers BE, Anderson CJ. Evaluation of copper-64-labeled somatostatin agonists and antagonist in SSTr2-transfected cell lines that are positive and negative for p53: implications for cancer therapy. Nucl Med Biol. 2012;39:187–97.
Heuser JE, Anderson RG. Hypertonic media inhibit receptor-mediated endocytosis by blocking clathrin-coated pit formation. J Cell Biol. 1989;108:389–400.
Wild D, Fani M, Behe M, Brink I, Rivier JE, Reubi JC, et al. First clinical evidence that imaging with somatostatin receptor antagonists is feasible. J Nucl Med. 2011;52:1412–7.
Acknowledgments
We thank the Swiss National Science Foundation and the FP7 project TARCC for financial support, and also the COST action D38.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, X., Fani, M., Schulz, S. et al. Comprehensive evaluation of a somatostatin-based radiolabelled antagonist for diagnostic imaging and radionuclide therapy. Eur J Nucl Med Mol Imaging 39, 1876–1885 (2012). https://doi.org/10.1007/s00259-012-2231-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-012-2231-8