Abstract
Purpose
[11C]DASB is currently the most frequently used highly selective radiotracer for visualization and quantification of central SERT. Its use, however, is hampered by the short half-life of 11C, the moderate cortical test–retest reliability, and the lack of quantifying endogenous serotonin. Labelling with 18F allows in principle longer acquisition times for kinetic analysis in brain tissue and may provide higher sensitivity. The aim of our study was to firstly use the new highly SERT-selective 18F-labelled fluoromethyl analogue of (+)-McN5652 ((+)-[18F]FMe-McN5652) in humans and to evaluate its potential for SERT quantification.
Methods
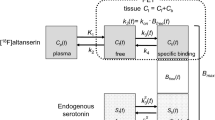
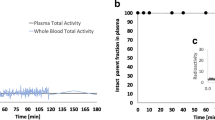
The PET data from five healthy volunteers (three men, two women, age 39 ± 10 years) coregistered with individual MRI scans were semiquantitatively assessed by volume-of-interest analysis using the software package PMOD. Rate constants and total distribution volumes (V T) were calculated using a two-tissue compartment model and arterial input function measurements were corrected for metabolite/plasma data. Standardized uptake region-to-cerebellum ratios as a measure of specific radiotracer accumulation were compared with those of a [11C]DASB PET dataset from 21 healthy subjects (10 men, 11 women, age 38 ± 8 years).
Results
The two-tissue compartment model provided adequate fits to the data. Estimates of total distribution volume (V T) demonstrated good identifiability based on the coefficients of variation (COV) for the volumes of interest in SERT-rich and cortical areas (COV V T <10%). Compared with [11C]DASB PET, there was a tendency to lower mean uptake values in (+)-[18F]FMe-McN5652 PET; however, the standard deviation was also somewhat lower. Altogether, cerebral (+)-[18F]FMe-McN5652 uptake corresponded well with the known SERT distribution in humans.
Conclusion
The results showed that (+)-[18F]FMe-McN5652 is also suitable for in vivo quantification of SERT with PET. Because of the long half-life of 18F, the widespread use within a satellite concept seems feasible.








Similar content being viewed by others
References
D'Amato RJ, Largent BL, Snowman AM, Snyder SH. Selective labeling of serotonin uptake sites in rat brain by [3H]citalopram contrasted to labeling of multiple sites by [3H]imipramine. J Pharmacol Exp Ther. 1987;242:364–71.
Hesse S, Barthel H, Schwarz J, Sabri O, Müller U. Advances in in vivo imaging of serotonergic neurons in neuropsychiatric disorders. Neurosci Biobehav Rev. 2004;28:547–63.
Meyer JH. Imaging the serotonin transporter during major depressive disorder and antidepressant treatment. J Psychiatry Neurosci. 2007;32:86–102.
Hesse S, Müller U, Lincke T, Barthel H, Villmann T, Angermeyer MC, et al. Serotonin and dopamine transporter imaging in patients with obsessive-compulsive disorder. Psychiatry Res. 2005;140:63–72.
Bailer UF, Frank GK, Henry SE, Price JC, Meltzer CC, Becker C, et al. Serotonin transporter binding after recovery from eating disorders. Psychopharmacol (Berl). 2007;195:315–24.
Szabo Z, Owonikoko T, Peyrot M, Varga J, Mathews WB, Ravert HT, et al. Positron emission tomography imaging of the serotonin transporter in subjects with a history of alcoholism. Biol Psychiatry. 2004;55:766–71.
McCann UD, Szabo Z, Vranesic M, Palermo M, Mathews WB, Ravert HT. Positron emission tomographic studies of brain dopamine and serotonin transporters in abstinent (±)3,4-methylenedioxymethamphetamine ("ecstasy") users: relationship to cognitive performance. Psychopharmacology (Berl). 2008;200:439–50.
Suehiro M, Scheffel U, Ravert HT, Dannals RF, Wagner Jr HN. [11C](+)McN5652 as a radiotracer for imaging serotonin uptake sites with PET. Life Sci. 1993;53:883–92.
Wilson AA, Ginovart N, Schmidt M, Meyer JH, Threlkeld PG, Houle S. Novel radiotracers for imaging the serotonin transporter by positron emission tomography: synthesis, radiosynthesis, and in vitro and ex vivo evaluation of 11C-labeled 2-(phenylthio)araalkylamines. J Med Chem. 2000;43:3103–10.
Ogden RT, Ojha A, Erlandsson K, Oquendo MA, Mann JJ, Parsey RV. In vivo quantification of serotonin transporters using [11C]DASB and positron emission tomography in humans: modeling considerations. J Cereb Blood Flow Metab. 2007;27:205–17.
Talbot PS, Frankle WG, Hwang DR, Huang Y, Suckow RF, Slifstein M, et al. Effects of reduced endogenous 5-HT on the in vivo binding of the serotonin transporter radioligand 11C-DASB in healthy humans. Synapse. 2005;55:164–75.
Huang YY, Ma KH, Tseng TW, Chou TK, Ng H, Mirsalis JC, et al. Biodistribution, toxicity and radiation dosimetry studies of the serotonin transporter radioligand 4-[18F]-ADAM in rats and monkeys. Eur J Nucl Med Mol Imaging. 2010;37:545–55.
Stehouwer JS, Goodman MM. Fluorine-18 radiolabeled PET tracers for imaging monoamine transporters: dopamine, serotonin, and norepinephrine. PET Clin 2009;4:101–28.
Huang Y, Zheng MQ, Gerdes JM. Development of effective PET and SPECT imaging agents for the serotonin transporter: has a twenty-year journey reached its destination? Curr Top Med Chem. 2010;10:1499–526.
Zessin J, Eskola O, Brust P, Bergman J, Steinbach J, Lehikoinen P, et al. Synthesis of S-([18F]fluoromethyl)-(+)-McN5652 as a potential PET radioligand for the serotonin transporter. Nucl Med Biol. 2001;28:857–63.
Kretzschmar M, Brust P, Zessin J, Cumming P, Bergmann R, Johannsen B. Autoradiographic imaging of the serotonin transporter in the brain of rats and pigs using S-([18F]fluoromethyl)-(+)-McN5652. Eur Neuropsychopharmacol. 2003;13:387–97.
Marjamäki P, Zessin J, Eskola O, Grönroos T, Haaparanta M, Bergman J, et al. S-[18F]fluoromethyl-(+)-McN5652, a PET tracer for the serotonin transporter: evaluation in rats. Synapse. 2003;47:45–53.
Brust P, Zessin J, Kuwabara H, Pawelke B, Kretzschmar M, Hinz R, et al. Positron emission tomography imaging of the serotonin transporter in the pig brain using [11C](+)-McN5652 and S-([18F]fluoromethyl)-(+)-McN5652. Synapse. 2003;47:143–51.
Brust P, Hinz R, Kuwabara H, Hesse S, Zessin J, Pawelke B, et al. In vivo measurement of the serotonin transporter with (S)-([18F]fluoromethyl)-(+)-McN5652. Neuropsychopharmacology. 2003;28:2010–9.
Hesse S, Stengler K, Regenthal R, Patt M, Becker GA, Franke A, et al. The serotonin transporter availability in untreated early-onset and late-onset patients with obsessive-compulsive disorder. Int J Neuropsychopharmacol. 2011;14:606–17.
Bertoldo A, Cobelli C. Physiological modelling of positron emission tomography images. In: Carson E, Cobelli C, editors. Modelling methodology for physiology and medicine. San Diego: Academic Press; 2001. p. 179–211.
Carson RE. Parameter estimation in positron emission tomography. In: Phelps ME, Mazziotta JC, Schelbert HR, editors. Positron emission tomography and autoradiography: principles and applications for the brain and heart. New York: Raven Press; 1986. p. 347–90.
Buck A, Gucker PM, Schönbächler RD, Arigoni M, Kneifel S, Vollenweider FX, et al. Evaluation of serotonergic transporters using PET and [11C](+)McN-5652: assessment of methods. J Cereb Blood Flow Metab. 2000;20:253–62.
Parsey RV, Kegeles LS, Hwang DR, Simpson N, Abi-Dargham A, Mawlawi O, et al. In vivo quantification of brain serotonin transporters in humans using [11C]McN 5652. J Nucl Med. 2000;41:1465–77.
Ibaraki M, Miura S, Shimosegawa E, Sugawara S, Mizuta T, Ishikawa A, et al. Quantification of cerebral blood flow and oxygen metabolism with 3-dimensional PET and 15O: validation by comparison with 2-dimensional PET. J Nucl Med. 2008;49:50–9.
Acknowledgments
We would like to thank all the volunteers who participated in this trial. This work was partially supported by the German Federal Ministry of Education and Research (BMBF), Germany, FKZ: 01EO1001. We also thank the anonymous reviewers for their helpful criticisms and comments that significantly improved the manuscript.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hesse, S., Brust, P., Mäding, P. et al. Imaging of the brain serotonin transporters (SERT) with 18F-labelled fluoromethyl-McN5652 and PET in humans. Eur J Nucl Med Mol Imaging 39, 1001–1011 (2012). https://doi.org/10.1007/s00259-012-2078-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-012-2078-z