Abstract
Purpose
Recombinant antibodies isolated from human antibody libraries have excellent affinities and high target specificity. As full-length IgGs are cleared inadequately slowly from the circulation, the aim of this work was to figure out which kind of recombinant antibody fragment proves to be appropriate for imaging epithelial cell adhesion molecule (EpCAM)-expressing tumours with the short-living radioisotope 68Ga.
Methods
In order to combine the promising tumour targeting properties of antibodies with 68Ga, four antibody variants with the same specificity and origin only differing in molecular weight were constructed for comparison. Therefore, the binding domains of a single-chain fragment variable (scFv) isolated from a human naïve antibody library were modified genetically to construct the respective full-length IgG, the tria- and diabody variants. These molecules were conjugated with the bifunctional chelating agent N,N′-bis[2-hydroxy-5-(carboxyethyl)benzyl]ethylenediamine-N,N′-diacetic acid (HBED-CC) to enable 68Ga labelling at ambient temperature and compared in biodistribution and immuno-PET imaging experiments.
Results
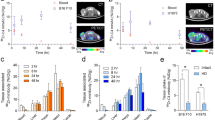
The antibody variants with identical specificity proved to have the correct molecular weight, high binding affinity and specificity to their antigen, EpCAM. Radiometal complexation was efficiently performed at room temperature leading to 68Ga-labelled antibodies with unchanged binding properties compared to the original antibody variants. The best targeting properties were obtained with the scFv and especially with the diabody. The triabody showed higher absolute tumour uptake but only moderate clearance from circulation.
Conclusion
The antibody variants differed considerably in normal organ uptake, clearance from circulation and tumour accumulation. The data demonstrate the feasibility of imaging solid tumours with the 68Ga-labelled diabody format. This type of recombinant protein might be a promising carrier even for the short-lived radiometal 68Ga to support e.g. the management of immunotherapy which may provide important information regarding receptor expression of solid tumours.







Similar content being viewed by others
References
Boswell CA, Brechbiel MW. Development of radioimmunotherapeutic and diagnostic antibodies: an inside-out view. Nucl Med Biol 2007;34:757–78.
Harris M. Monoclonal antibodies as therapeutic agents for cancer. Lancet Oncol 2004;5:292–302.
Oriuchi N, Higuchi T, Hanaoka H, Iida Y, Endo K. Current status of cancer therapy with radiolabeled monoclonal antibody. Ann Nucl Med 2005;19:355–65.
Nayak TK, Brechbiel MW. Radioimmunoimaging with longer-lived positron-emitting radionuclides: potentials and challenges. Bioconjug Chem 2009.
Olafsen T, Kenanova VE, Sundaresan G, Anderson AL, Crow D, Yazaki PJ, et al. Optimizing radiolabeled engineered anti-p185HER2 antibody fragments for in vivo imaging. Cancer Res 2005;65:5907–16.
Robinson MK, Doss M, Shaller C, Narayanan D, Marks JD, Adler LP, et al. Quantitative immuno-positron emission tomography imaging of HER2-positive tumor xenografts with an iodine-124 labeled anti-HER2 diabody. Cancer Res 2005;65:1471–8.
Wong JY, Chu DZ, Williams LE, Yamauchi DM, Ikle DN, Kwok CS, et al. Pilot trial evaluating an 123I-labeled 80-kilodalton engineered anticarcinoembryonic antigen antibody fragment (cT84.66 minibody) in patients with colorectal cancer. Clin Cancer Res 2004;10:5014–21.
Santimaria M, Moscatelli G, Viale GL, Giovannoni L, Neri G, Viti F, et al. Immunoscintigraphic detection of the ED-B domain of fibronectin, a marker of angiogenesis, in patients with cancer. Clin Cancer Res 2003;9:571–9.
Sundaresan G, Yazaki PJ, Shively JE, Finn RD, Larson SM, Raubitschek AA, et al. 124I-labeled engineered anti-CEA minibodies and diabodies allow high-contrast, antigen-specific small-animal PET imaging of xenografts in athymic mice. J Nucl Med 2003;44:1962–9.
Todorovska A, Roovers RC, Dolezal O, Kortt AA, Hoogenboom HR, Hudson PJ. Design and application of diabodies, triabodies and tetrabodies for cancer targeting. J Immunol Methods 2001;248:47–66.
Hudson PJ, Kortt AA. High avidity scFv multimers; diabodies and triabodies. J Immunol Methods 1999;231:177–89.
Holliger P, Prospero T, Winter G. “Diabodies”: small bivalent and bispecific antibody fragments. Proc Natl Acad Sci U S A 1993;90:6444–8.
Cai W, Olafsen T, Zhang X, Cao Q, Gambhir SS, Williams LE, et al. PET imaging of colorectal cancer in xenograft-bearing mice by use of an 18F-labeled T84.66 anti-carcinoembryonic antigen diabody. J Nucl Med 2007;48:304–10.
Birchler MT, Thuerl C, Schmid D, Neri D, Waibel R, Schubiger A, et al. Immunoscintigraphy of patients with head and neck carcinomas, with an anti-angiogenetic antibody fragment. Otolaryngol Head Neck Surg 2007;136:543–8.
Kortt AA, Lah M, Oddie GW, Gruen CL, Burns JE, Pearce LA, et al. Single-chain Fv fragments of anti-neuraminidase antibody NC10 containing five- and ten-residue linkers form dimers and with zero-residue linker a trimer. Protein Eng 1997;10:423–33.
Fani M, André JP, Maecke HR. 68 Ga-PET: a powerful generator-based alternative to cyclotron-based PET radiopharmaceuticals. Contrast Media Mol Imaging 2008;3:67–77.
Velikyan I, Beyer GJ, Långström B. Microwave-supported preparation of (68)Ga bioconjugates with high specific radioactivity. Bioconjug Chem 2004;15:554–60.
Maecke HR, Hofmann M, Haberkorn U. (68)Ga-labeled peptides in tumor imaging. J Nucl Med 2005;46 Suppl 1:172S–8S.
Eder M, Wängler B, Knackmuss S, LeGall F, Little M, Haberkorn U, et al. Tetrafluorophenolate of HBED-CC: a versatile conjugation agent for (68)Ga-labeled small recombinant antibodies. Eur J Nucl Med Mol Imaging 2008;35:1878–86.
Velikyan I, Maecke H, Langstrom B. Convenient preparation of 68Ga-based PET-radiopharmaceuticals at room temperature. Bioconjug Chem 2008;19:569–73.
Chaudry MA, Sales K, Ruf P, Lindhofer H, Winslet MC. EpCAM an immunotherapeutic target for gastrointestinal malignancy: current experience and future challenges. Br J Cancer 2007;96:1013–9.
Baeuerle PA, Gires O. EpCAM (CD326) finding its role in cancer. Br J Cancer 2007;96:417–23.
de Bono JS, Tolcher AW, Forero A, Vanhove GF, Takimoto C, Bauer RJ, et al. ING-1, a monoclonal antibody targeting Ep-CAM in patients with advanced adenocarcinomas. Clin Cancer Res 2004;10:7555–65.
Balzar M, Winter MJ, de Boer CJ, Litvinov SV. The biology of the 17-1A antigen (Ep-CAM). J Mol Med 1999;77:699–712.
Schuhmacher J, Maier-Borst W. A new Ge-68/Ga-68 radioisotope generator system for production of Ga-68 in dilute HCl. Int J Appl Radiat Isot 1981;32:31–6.
Kipriyanov SM, Moldenhauer G, Little M. High level production of soluble single chain antibodies in small-scale Escherichia coli cultures. J Immunol Methods 1997;200:69–77.
Kipriyanov SM, Kupriyanova OA, Little M, Moldenhauer G. Rapid detection of recombinant antibody fragments directed against cell-surface antigens by flow cytometry. J Immunol Methods 1996;196:51–62.
Kipriyanov SM. High-level periplasmic expression and purification of scFvs. Methods Mol Biol 2002;178:333–41.
Schuck P. Size-distribution analysis of macromolecules by sedimentation velocity ultracentrifugation and lamm equation modeling. Biophys J 2000;78:1606–19.
Laue TM, Shah BD, Ridgeway TM, Pelletier SL. Analytical ultracentrifugation in biochemistry and polymer science. Cambridge: Royal Society of Chemistry; 1992; p. 90–125.
Gonzales NR, De Pascalis R, Schlom J, Kashmiri SV. Minimizing the immunogenicity of antibodies for clinical application. Tumour Biol 2005;26:31–43.
Yazaki PJ, Wu AM, Tsai SW, Williams LE, Ikler DN, Wong JY, et al. Tumor targeting of radiometal labeled anti-CEA recombinant T84.66 diabody and t84.66 minibody: comparison to radioiodinated fragments. Bioconjug Chem 2001;12:220–8.
Adams GP, Shaller CC, Dadachova E, Simmons HH, Horak EM, Tesfaye A, et al. A single treatment of yttrium-90-labeled CHX-A″-C6.5 diabody inhibits the growth of established human tumor xenografts in immunodeficient mice. Cancer Res 2004;64:6200–6.
Khawli LA, Biela B, Hu P, Epstein AL. Comparison of recombinant derivatives of chimeric TNT-3 antibody for the radioimaging of solid tumors. Hybrid Hybridomics 2003;22:1–9.
Holliger P, Hudson PJ. Engineered antibody fragments and the rise of single domains. Nat Biotechnol 2005;23:1126–36.
Wu AM, Senter PD. Arming antibodies: prospects and challenges for immunoconjugates. Nat Biotechnol 2005;23:1137–46.
Wu AM. Antibodies and antimatter: the resurgence of immuno-PET. J Nucl Med 2009;50:2–5.
Behr TM, Goldenberg DM, Becker W. Reducing the renal uptake of radiolabeled antibody fragments and peptides for diagnosis and therapy: present status, future prospects and limitations. Eur J Nucl Med 1998;25:201–12.
Li L, Olafsen T, Anderson AL, Wu A, Raubitschek AA, Shively JE. Reduction of kidney uptake in radiometal labeled peptide linkers conjugated to recombinant antibody fragments. Site-specific conjugation of DOTA-peptides to a Cys-diabody. Bioconjug Chem 2002;13:985–95.
Balzar M, Briaire-de Bruijn IH, Rees-Bakker HA, Prins FA, Helfrich W, de Leij L, et al. Epidermal growth factor-like repeats mediate lateral and reciprocal interactions of Ep-CAM molecules in homophilic adhesions. Mol Cell Biol 2001;21:2570–80.
Jain RK. Physiological barriers to delivery of monoclonal antibodies and other macromolecules in tumors. Cancer Res 1990;50:814s–19s.
Orlova A, Magnusson M, Eriksson TL, Nilsson M, Larsson B, Höidén-Guthenberg I, et al. Tumor imaging using a picomolar affinity HER2 binding affibody molecule. Cancer Res 2006;66:4339–48.
Acknowledgements
A research grant from the Deutsche Krebshilfe for M. Eder is gratefully acknowledged. There is no other financial relationship of the author as stated above. Biodistribution and PET imaging were kindly performed by U. Schierbaum, K. Leotta and U. Bauder-Wüst (all German Cancer Research Center, Heidelberg, Germany). Dr. Moldenhauer (German Cancer Research Center, Heidelberg, Germany) and Prof. Diehl (Cologne, Germany) are gratefully acknowledged for providing cell lines for cell binding analysis. All animal experiments complied with the current laws of the Federal Republic of Germany.
Author information
Authors and Affiliations
Corresponding author
Additional information
Financial support
Research grant from the Deutsche Krebshilfe for M. Eder
Rights and permissions
About this article
Cite this article
Eder, M., Knackmuss, S., Le Gall, F. et al. 68Ga-labelled recombinant antibody variants for immuno-PET imaging of solid tumours. Eur J Nucl Med Mol Imaging 37, 1397–1407 (2010). https://doi.org/10.1007/s00259-010-1392-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-010-1392-6