Abstract
Purpose
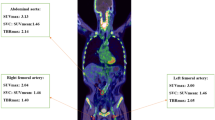
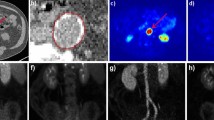
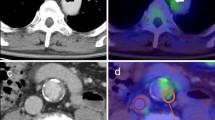
Inflamed atherosclerotic plaques may rupture and cause acute myocardial infarction, stroke and other thrombotic events. Early detection of these unstable plaques could, in many cases, prevent such potentially fatal events. 11C-choline or 18F-labelled choline derivatives for visualizing the synthesis of phospholipids, are promising markers of plaque inflammation with potential advantages over 18F-FDG. Their potential for plaque characterization in humans is, however, unclear. In this study the prevalence and distribution of 11C-choline uptake in the aortic and common carotid arterial walls of elderly male patients was evaluated with combined PET/CT. Additionally, the localization of radiotracer uptake and calcification was correlated in various vessel segments.
Methods
Image data from 93 consecutive male patients between 60 and 80 years old who had undergone whole-body 11C-choline PET/CT assessment for prostate cancer were evaluated retrospectively. 11C-choline uptake and calcification were analysed qualitatively and semiquantitatively and compared.
Results
11C-choline uptake was found in 95% of patients, calcification in 94% throughout all vessel segments. In 6% of the patients radiotracer uptake was colocalized with calcifications, whereas less than 1% of calcification sites showed increased radiotracer uptake.
Conclusion
Both 11C-choline uptake and calcification in the aortic and common carotid arterial walls are common in elderly men. Radiotracer uptake and calcification are, however, only rarely colocalized. 11C-choline has the potential to provide information about atherosclerotic plaques independent of calcification measurement.



Similar content being viewed by others
References
Tahara N, Kai H, Ishibashi M, Nakaura H, Kaida H, Baba K, et al. Simvastatin attenuates plaque inflammation: evaluation by fluorodeoxyglucose positron emission tomography. J Am Coll Cardiol 2006;48:1825–31.
Tatsumi M, Cohade C, Nakamoto Y, Wahl RL. Fluorodeoxyglucose uptake in the aortic wall at PET/CT: possible finding for active atherosclerosis. Radiology 2003;229:831–7.
Ben-Haim S, Kupzov E, Tamir A, Israel O. Evaluation of 18F-FDG uptake and arterial wall calcifications using 18F-FDG PET/CT. J Nucl Med 2004;45:1816–21.
Dunphy MP, Freiman A, Larson SM, Strauss HW. Association of vascular 18F-FDG uptake with vascular calcification. J Nucl Med 2005;46:1278–84.
Okane K, Ibaraki M, Toyoshima H, Sugawara S, et al. 18F-FDG accumulation in atherosclerosis: use of CT and MR co-registration of thoracic and carotid arteries. Eur J Nucl Med Mol Imaging 2006;33:589–94.
Hara M, Goodman PC, Leder RA. FDG-PET finding in early phase Takayasu arteritis. J Comput Assist Tomogr 1999;23:16–8.
Blockmans D, Stroobants S, Males A, Mortelmans L. Positron emission tomography in giant cell arteritis and polymyalgia rheumatica: evidence for inflammation of the aortic arch. Am J Med 2000;108:246–9.
De Winter F, Petrovic M, Van de Wiele C, et al. Imaging of giant cell arteritis: evidence of splenic involvement using FDG positron emission tomography. Clin Nucl Med 2000;25:633–4.
Turlakow A, Yeung HW, Pui J, Macapinlac H, et al. Fludeoxyglucose positron emission tomography in the diagnosis of giant cell arteritis. Arch Intern Med 2001;161:1003–7.
Yun M, Yeh D, Araujo LI, Jang S, et al. F-18 FDG uptake in the large arteries: a new observation. Clin Nucl Med 2001;26:314–9.
Yun M, Jang S, Cucchiara A, Newberg AB, et al. 18F-FDG uptake in the large arteries: a correlation study with the atherogenic risk factors. Semin Nucl Med 2002;32:70–6.
Rudd JH, Warburton EA, Fryer TD, Jones HA, et al. Imaging atherosclerotic plaque inflammation with [18F]-fluorodeoxyglucose positron emission tomography. Circulation 2002;105:2708–11.
Meller J, Strutz F, Siefker U, Scheel A, et al. Early diagnosis and follow-up of aortitis with [(18)F]FDG PET and MRI. Eur J Nucl Med Mol Imaging 2003;30:730–6.
Vallabhajosula S, Fuster V. Atherosclerosis: imaging techniques and the evolving role of nuclear medicine. J Nucl Med 1997;38:1788–96.
Ross R. Atherosclerosis, an inflammatory disease. N Engl J Med 1999;340:115–26.
Rittner HL, Kaiser M, Brack A, Szweda LI, et al. Tissue-destructive macrophages in giant cell arteritis. Circ Res 1999;84:1050–8.
Reape TJ, Groot PH. Chemokines and atherosclerosis. Atherosclerosis 1999;147:213–25.
Meller J, Strutz F, Siefker U, Scheel A, et al. Early diagnosis and follow-up of aortitis with [(18)F]FDG PET and MRI. Eur J Nucl Med Mol Imaging 2003;30:730–6.
Sakalihasan N, Van Damme H, Gomez P, Rigo P, et al. Positron emission tomography (PET) evaluation of abdominal aortic aneurysm (AAA). Eur J Vasc Endovasc Surg 2002;23:431–6.
Hara T, Kosaka N, Kishi H. PET imaging of prostate cancer using carbon-11-choline. J Nucl Med 1998;39:990–5.
DeGrado TR, Coleman RE, Wang S, Baldwin SW, et al. Synthesis and evaluation of 18F-labeled choline as an oncologic tracer for positron emission tomography: initial findings in prostate cancer. Cancer Res 2001;61:110–7.
Hara T, Kosaka N, Kishi H. Development of (18)F-fluoroethylcholine for cancer imaging with PET: synthesis, biochemistry, and prostate cancer imaging. J Nucl Med 2002;43:187–99.
Haeffner EW. Studies on choline permeation through the plasma membrane and its incorporation into phosphatidyl choline of Ehrlich-Lettre-ascites tumor cells in vitro. Eur J Biochem 1975;51:219–28.
Boggs KP, Rock CO, Jackowski S. Lysophosphatidylcholine and 1-O-octadecyl-2-O-methyl-rac-glycero-3-phosphocholine inhibit the CDP-choline pathway of phosphatidylcholine synthesis at the CTP: phosphocholine cytidylyltransferase step. J Biol Chem 1995;270:7757–64.
Katz-Brull R, Degani H. Kinetics of choline transport and phosphorylation in human breast cancer cells; NMR application of the zero trans method. Anticancer Res 1996;16:1375–80.
de Molina Ramirez A, Gutierrez R, Ramos MA, et al. Increased choline kinase activity in human breast carcinomas: clinical evidence for a potential novel antitumor strategy. Oncogene 2002;21:4317–22.
Wyss MT, Weber B, Honer M, Spath N, et al. 18F-choline in experimental soft tissue infection assessed with autoradiography and high-resolution PET. Eur J Nucl Med Mol Imaging 2004;31:312–6.
Matter CM, Wyss MT, Meier P, Spath N, et al. 18F-choline images murine atherosclerotic plaques ex vivo. Arterioscler Thromb Vasc Biol 2006;26:584–9.
Bucerius J, Schmaljohann J, Böhm I, Palmedo H, et al. Feasibility of 18F-fluoromethylcholine PET/CT for imaging of vessel wall alterations in humans – first results. Eur J Nucl Med Mol Imaging 2008;35:815–20.
Diamond GA, Forrester JS. Analysis of probability as an aid in the clinical diagnosis of coronary-artery disease. N Engl J Med 1979;300:1350–8.
Roivainen A, Yli-Kerttula T. Whole-body distribution of (11)C-choline and uptake in knee synovitis. Eur J Nucl Med Mol Imaging 2006;33:1372–3.
Rudd JH, Myers KS, Bansilal S, et al. Atherosclerosis inflammation imaging with 18F-FDG PET: carotid, iliac, and femoral uptake reproducibility, quantification methods, and recommendations. J Nucl Med 2008;49:871–8.
Rudd JH, Myers KS, Bansilal S, et al. Relationships among regional arterial inflammation, calcification, risk factors, and biomarkers. Circ Imaging 2009;2:107–15.
Schäfers KP, Stegger L. Combined imaging of molecular function and morphology with PET/CT and SPECT/CT: image fusion and motion correction. Basic Res Cardiol 2008;103:191–9.
Acknowledgments
This work was supported in part by grants from the Deutsche Forschungsgemeinschaft (DFG), Sonderforschungsbereich 656 Münster (projects C 2 and PM 3).
Author information
Authors and Affiliations
Corresponding author
Additional information
Katsuhiko Kato and Lars Stegger contributed equally to this work.
Rights and permissions
About this article
Cite this article
Kato, K., Schober, O., Ikeda, M. et al. Evaluation and comparison of 11C-choline uptake and calcification in aortic and common carotid arterial walls with combined PET/CT. Eur J Nucl Med Mol Imaging 36, 1622–1628 (2009). https://doi.org/10.1007/s00259-009-1152-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-009-1152-7