Abstract
Purpose
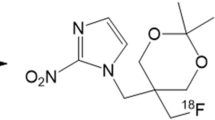
The aim of this study was to assess the pharmacokinetics, biodistribution and metabolism of [18F]EF3, a labelled 2-nitroimidazole hypoxia marker, in ten patients with head and neck cancer.
Methods
[18F]EF3 was administered intravenously (group 1, n = 5, mean dose ± SD: 324 ± 108 MBq; group 2, n = 5, mean dose ± SD: 1,134 ± 138 MBq) to patients (nine male, one female). Blood and urine samples and whole-body PET scans were obtained from 20 s to 4–6 h. Radioactivity was determined in several regions of interest.
Results
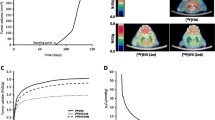
No serious adverse event was reported. [18F]EF3 concentration in blood exhibited a bi-exponential decline. [18F]EF3 was mainly eliminated in the urine. By 7 h 40 min after injection, 53 ± 14% of the injected dose was collected in the urine. There was no significant difference between the low- and high-dose groups. A progressive accumulation occurred also in the colon, indicating a hepatobiliary excretion. Except in organs involved in the elimination of [18F]EF3, the tumour-to-organ ratio remained close to or below unity in muscle, lungs, heart and brain at various times after injection. In one patient, tumour hypoxia was observed with a tumour-to-blood ratio ranging from 1.4 to 1.9. Last, [18F]EF3 remained very stable after injection, with percentage of native tracer above 87% in the serum and 84% in the urine.
Conclusion
Administration of [18F]EF3 in head and neck cancer patients is feasible and safe. Uptake and retention in tumour was observed, indicating the presence of hypoxia.




Similar content being viewed by others
References
Brizel D, Scully SP, Harrelson JM, Layfield LJ, Bean JM, Prosnitz LR. Tumour oxygenation predicts for the likelihood of distant metastases in human soft tissue sarcoma. Cancer Res. 1996;56:941–3.
Gatenby RA, Kessler HB, Rosenblaum JS, et al. Oxygen distribution in squamous cell carcinoma mestastases and its relationship to outcome of radiation therapy. Int J Radiat Oncol Biol Phys. 1988;14:831–8.
Hockel M, Schlenger K, Aral B, Mitze M, Schaffer U, Vaupel P. Association between tumour hypoxia and malignant progression in advanced cancer of the uterine cervix. Cancer Res. 1996;56:4509–15.
Stone HB, Brown JM, Phillips TL, Sutherland RM. Oxygen in human tumours: correlations between methods of measurements and response to therapy. Radiat Res. 1993;136:422–34.
Jonathan RA, Wijffels KI, Peeters W, de Wilde PC, Marres HA, Merkx MA, et al. The prognostic value of endogenous hypoxia-related markers for head and neck squamous cell carcinomas treated with ARCON. Radiother Oncol. 2006;79:288–97.
Hodgkiss RJ. Use of 2-nitroimidazoles as bioreductive markers for tumour hypoxia. Anticancer Drug Des. 1998;13:687–702.
Kaanders JHAM, Wijffels KIEM, Marres HAM, Pop LAM, Marres HAM, Ljungkvist ASE, et al. Pimonidazole binding and tumour vascularity predict for treatment outcome in head and neck cancer. Cancer Res. 2002;62:7066–74.
Evans SM, Judy KD, Dunphy I, Jenkins WT, Hwang WT, Nelson PT, et al. Hypoxia is important in the biology and aggression of human glial brain tumours. Clin Cancer Res. 2004;10:8177–84.
Evans SM, Fraker D, Hahn SM, Gleason K, Jenkins WT, Jenkins K, et al. EF5 binding and clinical outcome in human soft tissue sarcomas. Int J Radiat Oncol Biol Phys. 2006;64:922–7.
Kaanders JH, Bussink J, van der Kogel AJ. ARCON: a novel biology-based approach in radiotherapy. Lancet Oncol. 2002;3:728–37.
Rischin D, Hicks RJ, Fisher R, Binns D, Corry J, Porceddu S, Trans-Tasman Radiation Oncology Group Study 98.02. Prognostic significance of [18F]-misonidazole positron emission tomography-detected tumor hypoxia in patients with advanced head and neck cancer randomly assigned to chemoradiation with or without tirapazamine: a substudy of Trans-Tasman Radiation Oncology Group Study 98.02. J Clin Oncol. 2006;24:2098–104.
Gregoire V, Haustermans K, Geets X, Roels S, Lonneux M. PET-based treatment planning in radiotherapy: a new standard? J Nucl Med. 2007;48(Suppl 1):68S–77S.
Rajendran JG, Hendrickson KR, Spence AM, Muzi M, Krohn KA, Mankoff DA. Hypoxia imaging-directed radiation treatment planning. Eur J Nucl Med Mol Imaging. 2006;33(Suppl 1):44–53.
Eschmann SM, Paulsen F, Reimold M, Dittmann H, Welz S, Reischl G, et al. Prognostic impact of hypoxia imaging with 18F-misonidazole PET in non-small cell lung cancer and head and neck cancer before radiotherapy. J Nucl Med. 2005;46:253–60.
Mahy P, De Bast M, Levêque Ph, Gillart J, Labar D, Marchand J, et al. Preclinical validation of the hypoxic tracer 2-(2-nitroimidazol-1-yl)-N-(3,3,3-[18F]trifluoropropyl)-acetamide, [18F]-EF3. Eur J Nuc Med Mol Imaging 2004;31:1263–72.
Mahy P, De Bast M, Gillart J, Labar D, Grégoire V. Detection of tumour hypoxia: comparison between EF5 adducts and [18F]-EF3 uptake on an individual mouse tumour basis. Eur J Nuc Med Mol Imaging. 2006;33:553–6.
Christian N, Bol A, De Bast M, Labar D, Lee J, Mahy P, et al. Determination of tumour hypoxia with the PET tracer [18F]EF3: improvement of the tumour-to-background ratio in a mouse tumour model. Eur J Nucl Med Mol Imaging. 2007 Sep;34(9):1348–54.
Josse O, Labar D, Georges B, Gregoire V, Marchand-Brynaert J. Synthesis of [18F]-labeled EF3 [2-(2-nitroimidazol-1-yl)-N-(3,3,3-trifluoropropyl)-acetamide], a marker for PET detection of hypoxia. Bioorg Med Chem. 2001;9:665–75.
Lonneux M, Borbath I, Bol A, Coppens A, Sibomana M, Bausart R, et al. Attenuation correction in whole-body FDG oncological studies: the role of statistical reconstruction. Eur J Nucl Med. 1999;26:591–8.
Koch CJ, Hahn SM, Rockwell K Jr, Covey JM, McKenna WG, Evans SM. Pharmacokinetics of EF5 [2-(2-nitro-1-H-imidazol-1-yl)-N-(2,2,3,3,3-pentafluoropropyl) acetamide] in human patients: implications for hypoxia measurements in vivo by 2-nitroimidazoles. Cancer Chemother Pharmacol. 2001;48:177–87.
Ziemer LS, Evans SM, Kachur AV, Shuman AL, Cardi CA, Jenkins WT, et al. Noninvasive imaging of tumour hypoxia in rats using the 2-nitroimidazole 18F-EF5. Eur J Nucl Med 2003;30:259–66.
Graham MM, Peterson LM, Link JM, Evans ML, Rasey JS, Koh WJ, et al. Fluorine-18-fluoromisonidazole radiation dosimetry in imaging studies. J Nucl Med. 1997;38:1631–6.
Bruehlmeier M, Roelcke U, Schubiger PA, Ametamey SM. Assessment of hypoxia and perfusion in human brain tumours using PET with 18F-fluoromisonidazole and 15O–H2O. J Nucl Med. 2004;45:1851–9.
Yang DJ, Wallace S, Cherif A, Li C, Gretzer MB, Kim EE, et al. Development of F-18-labeled fluoroerythronitroimidazole as a PET agent for imaging tumour hypoxia. Radiology. 1995;194:795–800.
Koch CJ, Evans SM, Lord EM. Oxygen dependance of cellular uptake of EF5 [2-(2-nitro-1H-imidazol-1yl)-N-(2,2,3,3,3-pentafluoropropyl)acetamide]: analysis of drug adducts by fluorescent antibodies vs bound radioactivity. Br J Cancer. 1995;72:869–74.
Gronroos T, Eskola O, Lehtio K, Minn H, Marjamaki P, Bergman J, et al. Pharmacokinetics of [18F]FETNIM: a potential marker for PET. J Nucl Med. 2001;42:1397–404.
Lehtio K, Oikonen V, Gronroos T, Eskola O, Kalliokoski K, Bergman J, et al. Imaging of blood flow and hypoxia in head and neck cancer: initial evaluation with [(15)O]H(2)O and [(18)F]fluoroerythronitroimidazole PET. J Nucl Med. 2001;42:1643–52.
Koh WJ, Rasey JS, Evans ML, Grierson JR, Lewellen TK, Graham MM, et al. Imaging of hypoxia in human tumours with [F-18]fluoromisonidazole. Int J Radiat Oncol Biol Phys. 1992;22:199–212.
Tolvanen T, Lehtio K, Kulmala J, Oikonen V, Eskola O, Bergman J, et al. 18F-Fluoroerythronitroimidazole radiation dosimetry in cancer studies. J Nucl Med. 2002;43:1674–80.
Kumar PD, Stypinski D, Xia H, et al. Fluoroazomycin arabinoside (FAZA): synthesis, 2H and 3H-labelling and preliminary biological evaluation of a novel 2-nitroimidazole marker of tissue hypoxia. J Label Comp Radiopharm. 1999;42:3–16.
Souvatzoglou M, Grosu AL, Röper B, Krause BJ, Beck R, Reischl G, et al. Tumour hypoxia imaging with [18F]FAZA PET in head and neck cancer patients: a pilot study. Eur J Nucl Med Mol Imaging. 2007;34:1566–75.
Seddon BM, Payne GS, Simmons L, Ruddle R, Grimshaw R, Tan S, et al. A phase I study of SR-4554 via intravenous administration for noninvasive investigation of tumour hypoxia by magnetic resonance spectroscopy in patients with malignancy. Clin Cancer Res. 2003;9:5101–12.
Evans SM, Kachur AV, Shiue CA Hustinx R, Jenkins WT, Shive GG, Karp JS, et al. Noninvasive detection of tumour hypoxia using the 2-nitroimidazole [18F]EF1. J Nucl Med. 2000;41:327–36.
Rasey JS, Koh WJ, Evans ML, Peterson LM, Lewellen TK, Graham MM, et al. Quantifying regional hypoxia in human tumours with positron emission tomography of [18F]fluoromisonidazole: a pretherapy study of 37 patients. Int J Radiat Oncol Biol Phys 1996;36:417–28.
Chapman JD, Zanzocino P. On measuring hypoxia in individual tumours with radiolabeled agents. J Nucl Med. 2001;42:1653–5.
Chapman JD, Engelhardt EL, Stobbe CC, Schneider RF, Hanks GE. Measuring hypoxia and predicting tumour radioresistance with nuclear medicine assays. Radiother Oncol. 1998;46:229–37.
Partridge MA, Spinelli A, Rydera W, Hindorf C. The effect of beta + energy on performance of a small animal PET camera. Nucl Instrum Meth A. 2006;568:933–6.
Riedl CC, Brader P, Zanzonico P, Reid V, Woo Y, Wen B, et al. Tumor hypoxia imaging in orthotopic liver tumors and peritoneal metastasis: a comparative study featuring dynamic (18)F-MISO and (124)I-IAZG PET in the same study cohort. Eur J Nucl Med Mol Imaging. 2008;35:39–46.
Reischl G, Dorow DS, Cullinane C, Katsifis A, Roselt P, Binns D, et al. Imaging of tumor hypoxia with [124I]IAZA in comparison with [18F]FMISO and [18F]FAZA—first small animal PET results. J Pharm Pharm Sci. 2007;10:203–11.
Janssen HL, Haustermans KM, Balm AJ, Begg AC. Hypoxia in head and neck cancer: how much, how important? Head Neck. 2005;27:622–38.
Acknowledgements
This work was supported by research grants from the “Fonds de la Recherche Scientifique Médicale” (grants # 3.4571.95, 3.4610.99 and 3.4560.00) of Belgium and by grants from the “Fonds Joseph Maisin”, Brussels and the “Fondation Albert Joachim”, Herent, Belgium.
Author information
Authors and Affiliations
Corresponding author
Additional information
P. Mahy and X. Geets contributed equally to this paper.
Rights and permissions
About this article
Cite this article
Mahy, P., Geets, X., Lonneux, M. et al. Determination of tumour hypoxia with [18F]EF3 in patients with head and neck tumours: a phase I study to assess the tracer pharmacokinetics, biodistribution and metabolism. Eur J Nucl Med Mol Imaging 35, 1282–1289 (2008). https://doi.org/10.1007/s00259-008-0742-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-008-0742-0