Abstract
Purpose
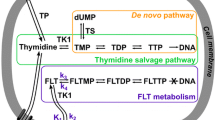
Clevudine (l-FMAU) an un-natural analogue of thymidine, is in clinical trials for the treatment of hepatitis B virus (HBV). l-FMAU is phosphorylated by cellular kinases such as thymidine kinase 1 and deoxycytidine kinase, and its triphosphate form inhibits HBV deoxyribonucleic acid synthesis. Thus, l-FMAU, radiolabeled with an appropriate isotope, may be useful for positron emission tomography (PET) imaging of tumor proliferation. We evaluated [18F]-l-FMAU as a PET imaging agent in tumor-bearing mice and compared the results with those of two other radiotracers, [18F]-d-FMAU and [18F]-FLT.
Methods
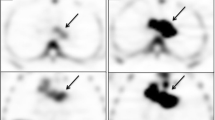
Subcutaneous xenografts of the human lung cancer cell lines H441 and H3255 were established in mice. A micro-PET scanner was used to obtain images of the tumor-bearing animals with [18F]-l-FMAU, [18F]-d-FMAU, and [18F]-FLT.
Results
At 2 h postinjection, the tumor uptake (% ID/g) of [18F]-l-FMAU, [18F]-d-FMAU, and [18F]-FLT in the faster-growing H441 cells was 3.13 ± 1.11, 7.74 ± 1.39, and 5.10 ± 1.45, respectively. The corresponding values for the slower-growing H3255 cells were 1.38 ± 0.81, 4.49 ± 1.08, and 0.57 ± 0.33. Tumor/muscle ratios of accumulation for [18F]-l-FMAU, [18F]-d-FMAU, and [18F]-FLT in H441 cells were 4.15 ± 1.82, 3.37 ± 1.19, and 12.94 ± 4.38, respectively, and the corresponding values in H3255 cells were 1.62 ± 0.50, 1.96 ± 0.74, and 1.50 ± 0.90.
Conclusions
[18F]-l-FMAU may be a useful agent for imaging tumor proliferation in fast-growing human lung cancers by PET.





Similar content being viewed by others
References
Watanabe KA, Reichman U, Hirota K, Lopez C, Fox JJ. Nucleosides 110. Synthesis and herpes virus activity of some 2′-fluoro-2′-deoxyarabinofuranosyl pyrimidine nucleosides. J Med Chem 1979;22:21-4.
Watanabe KA, Su TL, Riechman U, Greenberg N, Lopez C, Fox JJ. Nucleosides 129. Synthesis of antiviral nucleosides: 5-Alkenyl 1-(2-deoxy-2-fluoro-β-D-arabinofuranosyl)uracils. J Med Chem 1984;27:91-4.
Tann CH, Brodfuehrer PR, Brundidge SP, Sapino C, Howell HG. Fluorocarbohydrates in synthesis. An efficient synthesis of 1-(2-deoxy-2-fluoro-β-D-arabinofuranosyl)-5-iodouracil (β-FIAU) and 1-(2-deoxy-2-fluoro-β-D-arabinofuranosyl)thymine (β-FMAU). J Org Chem 1985;50:3644-7.
Buti M, Esteban R. Entecavir, FTC, L-FMAU, LdT and others. J Hepatology 2003;39:S139-42.
Giles FJ, Garcia-Manero G, Cortes JE, Baker SD, Miller CB, O’Brien SM, et al. Phase II study of troxacitabine, a novel dioxolane nucleoside analogue, in patients with refractory leukemia. J Clin Oncol 2002;20:656-64.
Hu R, Li L, Degreve B, Dutschman GE, Lam W, Cheng Y-C. Behavior of thymidine kinase towards monophosphate metabolites and its role in the metabolism of 1-(2′-deoxy-2′-fluoro-beta-L-arabinofuranosyl)-5-methyluracil (Clevudine) and 2′,3′-didehydro-2′,3′-dideoxythymidine in cells. Antimicrob Agents Chemother 2005;49:2044-9.
Chu CK, Ma T, Shanmuganathan K, Wang C, Xiang Y, Pai SB, et al. Use of 2′-fluoro-5-methyl-beta-L-arabinofuranosyluracil as a novel antiviral agent for hepatitis B virus and Epstein-Barr virus. Antimicrob Agents Chemother 1995;39:979-81.
Ma T, Pai SB, Zhu YL, Lin JS, Shanmuganathan K, Du J, et al. Structure-activity relationship of 1-(2-deoxy-2-fluoro-β-L-arabinofuranosyl)pyrimidine nucleosides as anti-hepatitis B virus agents. J Med Chem 1996;39:2835-43.
Liu S-H, Grove KL, Cheng Y-C. Unique metabolism of a novel antiviral l-nucleoside analog, 2′-fluoro-5-methyl-β-l-arabinofuranosyluracil: a substrate for both thymidine kinase and deoxycytidine kinase. Antimicrob Agents Chemother 1998;42:833-9.
Lee J, Yoo B-C, Lee H-S, Yoo H-W, Kang MJ, Kim D-H. Rapid quantitative determination of L-FMAU-TP from peripheral blood. The Drug Monit 2006;28:131-7.
Chong Y, Chu CK. Understanding the unique mechanism of L-FMAU (clevudine) against hepatitis B virus: Molecular dynamics studies. Bioorg Med Chem Lett 2002;12:3459-62.
Balakrishna Pai S, Liu SH, Zhu YL, Chu CK. Inhibition of hepatitis B virus by novel L-nucleoside 2′-deoxy-2′-fluoro-1-β-L-arabinofuranosyluracil. Antimicrob Agents Chemother 1996;40:380-6.
Chu CK, Boundinot FD, Peek SF, Hong JH, Choi Y, Korba BE, et al. Preclinical investigation of L-FMAU as an anti-hepatitis B virus agent. Antivir Therapy 1998;3:113-21.
Krishnan P, Fu Q, Lam W, Liou J-Y, Dutschman G, Cheng Y-C. Phosphorylation of pyrimidine deoxynucleoside analog diphosphates. Selective phosphorylation of L-nucleoside analog diphosphates by 3-phosphoglycerate kinase. J Biol Chem 2002;277:5453-9.
Kocic I. Clevudine (University of Georgia/Abbott/Bukwang/Triangle/Yale University). Curr Opin Investig Drugs 2000;1:308-13.
Apisarnthanarax S, Alauddin MM, Mourtada F, Ariga H, Raju U, Mawlawi O, et al. Early detection of chemoradioresponse in esophageal carcinoma by 3′-deoxy-3′-3H-fluorothymidine using preclinical tumor models. Clin Can Res 2006;12:4590-7.
Lifrange E, Kridelka F, Colin C. Stereotaxic needle-core biopsy and fine-needle aspiration biopsy in the diagnosis of nonpalpable breast lesions: controversies and future prospects. Eur J Radiol 1997;24:39-47.
Strauss LG, Conti PS. The applications of PET in clinical oncology. J Nucl Med 1991;32:623-48.
Sun H, Sloan A, Mangner TJ, Vaishampayan U, Muzik O, Collins JM, et al. Imaging DNA synthesis with [18F]FMAU and positron emission tomography in patients with cancer. Eur J Nucl Med Mol Imaging 2005;32:15-22.
Christman D, Crawford EJ, Friedkin M, Wolf AP. Detection of DNA synthesis in intact organism with positron emitting [Methyl-11C]thymidine. Proc Natl Acad Sci USA 1972;69:988-92.
Martiat Ph, Ferrant A, Labar D, Cogneau M, Bol A, Michel C, et al. In vivo measurement of C-11 thymidine uptake in non-Hodgkin’s lymphoma using positron emission tomography. J Nucl Med 1988;29:1633-7.
Shields AF, Lim K, Grierson JL, Krohn KA. Utilization of labeled thymidine in DNA synthesis: Studies for PET. J Nucl Med 1990;31:337-42.
Vander Borght T, Pauwels S, Lambotte L, Labar D, De Maeght S, Stroobandt G, et al. Brain tumor imaging with PET and 2-[11C]thymidine. J Nucl Med 1994;35:974-82.
Conti PS, Hilton J, Wong DF, Alauddin MM, Dannals RF, Ravert HT, et al. High performance liquid chromatography of methyl-C-11-thymidine and its major metabolites for clinical PET studies. Nucl Med Biol 1994;21:1045-51.
Conti PS, Alauddin MM, Fissekis JD, Schmall B, Watanabe KA. Synthesis of 2′-fluoro-5-[11C]methyl-1-β-D-arabinofuranosyluracil ([11C]-FMAU): A potential nucleoside analogue for in vivo study of cellular proliferation with PET. Nucl Med Biol 1995;22:783-9.
Alauddin MM, Conti PS, Fissekis JD. Synthesis of [18F]-labeled 2′-deoxy-2′-fluoro-1-β-D-arabinofuranosyluracil ([18F]FMAU). J Labelled Comp Radiopharm 2002;45:583-90.
Grierson JR, Shields AF. Radiosynthesis of 3′-deoxy-3′-fluoro-thymidine: 18F-FLT for imaging cellular proliferation in vivo. Nucl Med Biol 2000;27:143-56.
Martin SJ, Eisenbarth JA, Wagner-Utermann U, Mier W, Henze M, Pritzkow H, et al. A new precursor for the radiosynthesis of [18F]FLT. Nucl Med Biol 2002;29:263-73.
Conti PS, Bading JR, Alauddin MM, Fissekis JD, Berenji B. Preliminary clinical imaging trials with the tumor imaging 2¢-fluoro-5-[11C]-methyl-1-b-D-arabinofuranosyluracil (11C-FMAU). Comparison studies with FDG. Radiological Society of North America meeting (RSNA), 2002, Dec. 5-9, Chicago, IL.
Kenny LM, Vigushin DM, Al-Nahas A, Osman E, Luthra SK, Shousha S, et al. Quantification of cellular proliferation in tumor and normal tissues of patients with breast cancer by [18F]-fluorothymidine-positron emission tomography imaging: evaluation of analytical methods. Cancer Res 2005;65:10104-12.
Bading JR, Shahinian A, Bathija P, Conti PS. Pharmacokinetics of the thymidine analog 2’-fluoro-5-[14C]-methyl-1-β-D-arabinofuranosyluracil ([14C]FMAU in rat prostate tumor cells. Nucl Med Biol 2000;27:361-8.
Sherly JL, Kelly TJ. Regulation of human thymidine kinase during the cell cycle. J Biol Chem 1988;263:8350-8.
Van der Wilt CL, Kroep JR, Loves WJ, Rots MG, Van Groeningen CJ, Kaspers GJ, et al. Expression of deoxycytidine kinase in leukemic cells compared with solid tumor cell lines, liver metastases and normal liver. Eur J Cancer 2003;39:691-7.
Mukhopadhyay U, Pal A, Gelovani JG, Bornmann W, Alauddin MM. Radiosynthesis of 2′-deoxy-2′-[18F]-fluoro-5-methyl-1-β-L-arabinofuranosyluracil [18F]L-FMAU. Appl Rad Isotopes 2007;65:941-6.
Ory DS, Neugeboren BA, Mulligan RC. A stable human-derived packaging cell line for production of high titer retrovirus/vesicular stomatitis virus G pseudo types. Proc Natl Acad Sci USA 1996;93:11400-6.
Patlak CS, Blasberg RG, Fenstermacher JD. Graphical evaluation of blood-to-brain transfer constants from multiple-time uptake data. J Cereb Blood Flow Metab 1983;3:1-7.
Kong W, Engel K, Wang J. Mammalian nucleoside transporters. Curr Drug Metab 2004;5:63-84.
Philips FS, Feinberg A, Chou T-C, Vidal PM, Su T-L, Watanabe KA, et al. Distribution, metabolism and excretion of 1-(2-deoxy-2-fluoro-β-D-arabinofuranosyl)thymidine and 1-(2-deoxy-2-fluoro-β-D-arabinofuranosyl)-5-iodocytidine. Cancer Res 1983;43:3619-27.
Bading JR, Shahinian AH, Vail A, Bathija P, Koszalka GW, Koda RT, et al. Pharmacokinetics of the thymidine analog 2′-fluoro-5-methyl-1-beta-D-arabinofuranosyluracil (FMAU) in tumor-bearing rats. Nucl Med Biol 2004;31:407-18.
Wright JD, Ma T, Chu CK, Boudinot FD. Pharmacokinetics of 1-(2-deoxy-2-fluoro-β-L-arabinofuranosyluracil (L-FMAU) in rats. Pharm Res 1995;12:1350-3.
Shields AF, Briston DA, Chandupatla S, Douglas KA, Lawhorn-Crews J, Collins JM, Mangner TJ, Heilbrun LK, Muzik O. A simplified analysis of [18F]-3′-deoxy-3′-fluorothymidine metabolism and retention. Eur J Nucl Med Mol Imaging 2005;32:1269-75.
Acknowledgments
This work was supported by startup funds from The University of Texas M. D. Anderson Cancer Center to Drs. Mian Alauddin and Juri Gelovani.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nishii, R., Volgin, A.Y., Mawlawi, O. et al. Evaluation of 2′-deoxy-2′-[18F]fluoro-5-methyl-1-β-l-arabinofuranosyluracil ([18F]-l-FMAU) as a PET imaging agent for cellular proliferation: comparison with [18F]-d-FMAU and [18F]FLT. Eur J Nucl Med Mol Imaging 35, 990–998 (2008). https://doi.org/10.1007/s00259-007-0649-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-007-0649-1