Abstract
Purpose
In this study, we evaluated the 12-O-tetradecanoyl-phorbol-13-acetate (TPA)-induced acute and chronic inflammation in living mice by PET imaging of TNF-α and integrin αvβ3 expression.
Methods
TPA was topically applied to the right ear of BALB/c mice every other day to create the inflammation model. 64Cu-DOTA-etanercept and 64Cu-DOTA-E{E[c(RGDyK)]2}2 were used for PET imaging of TNF-α and integrin αvβ3 expression in both acute and chronic inflammation. Hematoxylin and eosin staining, ex vivo autoradiography, direct tissue sampling, and immunofluorescence staining were also performed to confirm the non-invasive PET imaging results.
Results
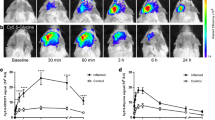
The ear thickness increased significantly and the TNF-α level more than tripled after a single TPA challenge. MicroPET imaging using 64Cu-DOTA-etanercept revealed high activity accumulation in the inflamed ear, reaching 11.1 ± 1.3, 13.0 ± 2.0, 10.9 ± 1.4, 10.2 ± 2.2%ID/g at 1, 4, 16, and 24 h post injection, respectively (n = 3). Repeated TPA challenges caused TPA-specific chronic inflammation and reduced 64Cu-DOTA-etanercept uptake due to lowered TNF-α expression. 64Cu-DOTA-E{E[c(RGDyK)]2}2 uptake in the chronically inflamed ears (after four and eight TPA challenges) was significantly higher than in the control ears and those after one TPA challenge. Immunofluorescence staining revealed increased integrin β3 expression, consistent with the non-invasive PET imaging results using 64Cu-DOTA-E{E[c(RGDyK)]2}2 as an integrin αvβ3-specific radiotracer. Biodistribution and autoradiography studies further confirmed the quantification capability of microPET imaging.
Conclusion
Successful PET imaging of TNF-α expression in acute inflammation and integrin αvβ3 expression in chronic inflammation provides the rationale for multiple target evaluation over time to fully understand the inflammation processes.





Similar content being viewed by others
References
Feghali CA, Wright TM. Cytokines in acute and chronic inflammation. Front Biosci 1997;2:d12–26.
Elenkov IJ, Iezzoni DG, Daly A, Harris AG, Chrousos GP. Cytokine dysregulation, inflammation and well-being. Neuroimmunomodulation 2005;12:255–69.
Franquet T, Stern EJ. Bronchiolar inflammatory diseases: high-resolution CT findings with histologic correlation. Eur Radiol 1999;9:1290–303.
Rennen HJ, Boerman OC, Oyen WJ, Corstens FH. Imaging infection/inflammation in the new millennium. Eur J Nucl Med 2001;28:241–52.
Tehranzadeh J, Ashikyan O, Dascalos J. Magnetic resonance imaging in early detection of rheumatoid arthritis. Semin Musculoskelet Radiol 2003;7:79–94.
Lavender JP, Lowe J, Barker JR, Burn JI, Chaudhri MA. Gallium 67 citrate scanning in neoplastic and inflammatory lesions. Br J Radiol 1971;44:361–6.
Fischman AJ, Rubin RH, White JA, Locke E, Wilkinson RA, Nedelman M, et al. Localization of Fc and Fab fragments of nonspecific polyclonal IgG at focal sites of inflammation. J Nucl Med 1990;31:1199–205.
Dams ET, Oyen WJ, Boerman OC, Storm G, Laverman P, Kok PJ, et al. 99mTc-PEG liposomes for the scintigraphic detection of infection and inflammation: clinical evaluation. J Nucl Med 2000;41:622–30.
Peters AM, Danpure HJ, Osman S, Hawker RJ, Henderson BL, Hodgson HJ, et al. Clinical experience with 99mTc-hexamethylpropylene-amineoxime for labelling leucocytes and imaging inflammation. Lancet 1986;2:946–9.
Becker W, Bair J, Behr T, Repp R, Streckenbach H, Beck H, et al. Detection of soft-tissue infections and osteomyelitis using a technetium-99m-labeled anti-granulocyte monoclonal antibody fragment. J Nucl Med 1994;35:1436–43.
Thakur ML, Marcus CS, Henneman P, Butler J, Sinow R, Diggles L, et al. Imaging inflammatory diseases with neutrophil-specific technetium-99m-labeled monoclonal antibody anti-SSEA-1. J Nucl Med 1996;37:1789–95.
van der Laken CJ, Boerman OC, Oyen WJ, van de Ven MT, Edwards DS, Barrett JA, et al. Technetium-99m-labeled chemotactic peptides in acute infection and sterile inflammation. J Nucl Med 1997;38:1310–5.
Barrera P, van der Laken CJ, Boerman OC, Oyen WJ, van de Ven MT, van Lent PL, et al. Radiolabelled interleukin-1 receptor antagonist for detection of synovitis in patients with rheumatoid arthritis. Rheumatology (Oxford) 2000;39:870–4.
Rennen HJ, Boerman OC, Oyen WJ, van der Meer JW, Corstens FH. Specific and rapid scintigraphic detection of infection with 99mTc-labeled interleukin-8. J Nucl Med 2001;42:117–23.
Signore A, Chianelli M, Annovazzi A, Bonanno E, Spagnoli LG, Pozzilli P, et al. 123I-interleukin-2 scintigraphy for in vivo assessment of intestinal mononuclear cell infiltration in Crohn’s disease. J Nucl Med 2000;41:242–9.
Moyer BR, Vallabhajosula S, Lister-James J, Bush LR, Cyr JE, Snow DA, et al. Technetium-99m-white blood cell-specific imaging agent developed from platelet factor 4 to detect infection. J Nucl Med 1996;37:673–9.
Stumpe KD, Dazzi H, Schaffner A, von Schulthess GK. Infection imaging using whole-body FDG-PET. Eur J Nucl Med 2000;27:822–32.
Leong KG, Karsan A. Signaling pathways mediated by tumor necrosis factor alpha. Histol Histopathol 2000;15:1303–25.
Cairns CB, Panacek EA, Harken AH, Banerjee A. Bench to bedside: tumor necrosis factor-alpha: from inflammation to resuscitation. Acad Emerg Med 2000;7:930–41.
Taylor PC. Anti-cytokines and cytokines in the treatment of rheumatoid arthritis. Curr Pharm Des 2003;9:1095–106.
Goldenberg MM. Etanercept, a novel drug for the treatment of patients with severe, active rheumatoid arthritis. Clin Ther 1999;21:75–87.
Goffe B. Etanercept (Enbrel)—an update. Skin Therapy Lett 2004;9:1–4.
Cai W, Chen X. Anti-angiogenic cancer therapy based on integrin αvβ3 antagonism. Anti-Cancer Agents Med Chem 2006;6:407–28.
Danese S, Sans M, de la Motte C, Graziani C, West G, Phillips MH, et al. Angiogenesis as a novel component of inflammatory bowel disease pathogenesis. Gastroenterology 2006;130:2060–73.
Wilder RL. Integrin αvβ3 as a target for treatment of rheumatoid arthritis and related rheumatic diseases. Ann Rheum Dis 2002;61 Suppl 2:ii96–9.
Storgard CM, Stupack DG, Jonczyk A, Goodman SL, Fox RI, Cheresh DA. Decreased angiogenesis and arthritic disease in rabbits treated with an αvβ3 antagonist. J Clin Invest 1999;103:47–54.
Wu Y, Zhang X, Xiong Z, Cheng Z, Fisher DR, Liu S, et al. MicroPET imaging of glioma αv-integrin expression using 64Cu-labeled tetrameric RGD peptide. J Nucl Med 2005;46:1707–18.
Cai W, Wu Y, Chen K, Cao Q, Tice DA, Chen X. In vitro and in vivo characterization of 64Cu-labeled Abegrin™, a humanized monoclonal antibody against integrin αvβ3. Cancer Res 2006;66:9673–81.
Meares CF, McCall MJ, Reardan DT, Goodwin DA, Diamanti CI, McTigue M. Conjugation of antibodies with bifunctional chelating agents: isothiocyanate and bromoacetamide reagents, methods of analysis, and subsequent addition of metal ions. Anal Biochem 1984;142:68–78.
Murakawa M, Yamaoka K, Tanaka Y, Fukuda Y. Involvement of tumor necrosis factor (TNF)-alpha in phorbol ester 12-O-tetradecanoylphorbol-13-acetate (TPA)-induced skin edema in mice. Biochem Pharmacol 2006;71:1331–6.
Cai W, Zhang X, Wu Y, Chen X. A thiol-reactive 18F-labeling agent, N-[2-(4-18F-fluorobenzamido)ethyl]maleimide (18F-FBEM), and the synthesis of RGD peptide-based tracer for PET imaging of αvβ3 integrin expression. J Nucl Med 2006;47:1172–80.
Zhang X, Cai W, Cao F, Schreibmann E, Wu Y, Wu JC, et al. 18F-labeled bombesin analogs for targeting GRP receptor-expressing prostate cancer. J Nucl Med 2006;47:492–501.
Cai W, Shin DW, Chen K, Gheysens O, Cao Q, Wang SX, et al. Peptide-labeled near-infrared quantum dots for imaging tumor vasculature in living subjects. Nano Lett 2006;6:669–76.
Cai W, Rao J, Gambhir SS, Chen X. How molecular imaging is speeding up anti-angiogenic drug development. Mol Cancer Ther 2006;5:2624–33.
Koutroubakis IE, Tsiolakidou G, Karmiris K, Kouroumalis EA. Role of angiogenesis in inflammatory bowel disease. Inflamm Bowel Dis 2006;12:515–23.
Jackson JR, Seed MP, Kircher CH, Willoughby DA, Winkler JD. The codependence of angiogenesis and chronic inflammation. Faseb J 1997;11:457–65.
Walsh DA. Angiogenesis and arthritis. Rheumatology (Oxford) 1999;38:103–12.
Cai W, Chen K, Mohamedali KA, Cao Q, Gambhir SS, Rosenblum MG, et al. PET of vascular endothelial growth factor receptor expression. J Nucl Med 2006;47:2048–56.
Pichler BJ, Kneilling M, Haubner R, Braumuller H, Schwaiger M, Rocken M, et al. Imaging of delayed-type hypersensitivity reaction by PET and 18F-galacto-RGD. J Nucl Med 2005;46:184–9.
Acknowledgements
This work was supported by the National Institute of Biomedical Imaging and Bioengineering (NIBIB) (R21 EB001785), the National Cancer Institute (NCI) (R21 CA102123, P50 CA114747, U54 CA119367, and R24 CA93862), the Department of Defense (DOD) (W81XWH-04-1-0697, W81XWH-06-1-0665, W81XWH-06-1-0042, W81XWH-07-1-0374, and DAMD17-03-1-0143), and a Benedict Cassen Postdoctoral Fellowship from the Education and Research Foundation of the Society of Nuclear Medicine (to W. Cai). We thank Drs. Gang Niu, Hui Wang, and Zhanhong Wu for their help with the biodistribution studies. We also thank the cyclotron team at the University of Wisconsin, Madison for 64Cu production.
Author information
Authors and Affiliations
Corresponding author
Additional information
Qizhen Cao and Weibo Cai contributed equally to this work.
Rights and permissions
About this article
Cite this article
Cao, Q., Cai, W., Li, ZB. et al. PET imaging of acute and chronic inflammation in living mice. Eur J Nucl Med Mol Imaging 34, 1832–1842 (2007). https://doi.org/10.1007/s00259-007-0451-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-007-0451-0