Abstract
Purpose
To evaluate low dose non-enhanced CT and standard dose contrast-enhanced CT in combined PET/CT protocols for staging and therapy planning of non-small cell lung cancer (NSCLC).
Methods
Retrospective analysis was performed of 50 consecutive patients with proven NSCLC who had been referred for primary staging (n=41) or restaging (n=9). All patients underwent a multi-phase PET/CT consisting of a low dose non-enhanced attenuation scan and an arterial and portal-venous contrast-enhanced CT scan followed by whole-body PET. Fused datasets of non-enhanced and contrast-enhanced PET/CT were compared per patient by using the TNM staging system, and per lesion regarding localisation, characterisation and delineation of tumour lesions. The staging results were validated either by histopathology or by clinical–radiological follow-up for ≥6 months.
Results
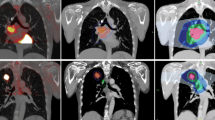
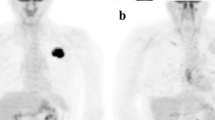
In 47/50 patients, the results of T staging did not differ between the two PET/CT protocols. Three patients could only be correctly classified as having T4 tumours after contrast application. Regarding N staging, both protocols yielded the same results. In M staging, there was only one patient with an improvement of the results as a result of contrast application. The lesion-based analysis of 92 sites showed no difference in the accuracy of lesion localisation and only one revision of lesion characterisation by contrast-enhanced PET/CT. The assessment of tumour delineation was altered by contrast application in 58/92 sites (p<0.0001). In 10/50 patients, contrast-enhanced PET/CT detected additional clinically important findings.
Conclusion
In patients with advanced NSCLC, contrast-enhanced CT as part of the PET/CT protocol more accurately assessed the TNM stage in 8% of patients compared with non-contrast PET/CT. However, for planning of 3D conformal radiotherapy and non-conventional surgery, contrast-enhanced PET/CT protocols are indispensable owing to their superiority in precisely defining the tumour extent.





Similar content being viewed by others
References
Deslauriers J, Gregoire J. Clinical and surgical staging of non-small cell lung cancer. Chest 2000;117:96S–103S
Barker JM, Silvestri GA. Lung cancer staging. Curr Opin Pulm Med 2002;8:287–293
Laroche C, Fairbairn I, Moss H, Pepke-Zaba J, Sharples L, Flower C, et al. Role of computed tomographic scanning of the thorax prior bronchoscopy in the investigation of suspected lung cancer. Thorax 2000;55:359–363
Steinert HC, Hauser M, Allemann F, Engel H, Berthold T, von Schulthess GK, et al. Non-small lung cancer: nodal staging with FDG PET versus CT with correlative lymph node mapping and sampling. Radiology 1997;202:441–446
Pieterman RM, van Putten JWG, Meuzelaar JJ, Mooyaart EL, Vaalburg W, Koeter GH, et al. Preoperative staging of non-small-cell lung cancer with positron-emission tomography. N Engl J Med 2000;343:254–561
Sachs S, Bilfinger T. The impact of positron emission tomography on clinical decision making in a university-based multidisciplinary lung cancer practice. Chest 2005;128:698–703
Lloyd C, Silvestri GA. Mediastinal staging of non-small-cell lung cancer. Cancer control 2001;8:311–317
Gould MK, Kuschner WG, Rydzak CE, Maclean CC, Demas AN, Shigemitsu H, et al. Test performance of positron emission tomography and computed tomography for mediastinal staging in patients with non-small-cell lung cancer. Ann Intern Med 2003;139:879–892
Gupta NC, Graeber GM, Bishop HA. Comparative efficacy of positron emission tomography with fluorodeoxyglucose in evaluation of small (<1 cm), intermediate (1 to 3 cm), and large (>3 cm) lymph node lesions. Chest 2000;117:773–778
Vansteenkiste JF, Stroobants SG, De Leyn PR, Dupont PJ, Bogaert J, Maes A, et al. Lymph node staging in non-small-cell lung cancer with FDG-PET scan: a prospective study on 690 lymph node stations from 68 patients. J Clin Oncol 1998;16:2142–2149
van Tinteren H, Hoekstra OS, Smit EF, van den Bergh JH, Schreurs AJ, Stallaert RA, et al. Effectiveness of positron emission tomography in the preoperative assessment of patients with suspected non-small-cell lung cancer: the PLUS multicentre randomised trial. Lancet 2002;359:1388–1393
Antoch G, Stattaus J, Nemat AT, Marnitz S, Beyer T, Kuehl H, et al. Non-small cell lung cancer: dual-modality PET/CT in preoperative staging. Radiology 2003;229:526–533
Lardinois D, Weder W, Hany TF, Kamel EM, Korom S, Seifert B, et al. Staging of non-small-cell lung cancer with integrated positron emission tomography and computed tomography. N Engl J Med 2003;348:2500–2507
Goerres GW, von Schulthess GK, Steinert HC. Why most PET of lung and head-and-neck cancer will be PET/CT. J Nucl Med 2004;45(suppl):66S–71S
Antoch G, Freudenberg LS, Beyer T, Bockisch A, Debatin J. To enhance or not to enhance? 18F-FDG and CT contrast agents in dual-modality 18F-FDG PET/CT. J Nucl Med 2004;45(suppl):56S–65S
Strobel K, Thuerl M, Hany TF. How much intravenous contrast is needed in FDG-PET/CT? Nuklearmedizin 2005;44:S32–S37
von Schulthess GK. Integrated modality imaging with PET-CT and SPECT-CT: CT issues. Eur Radiol Suppl 2005; 15 Suppl 4:D121–D126
Brechtel K, Klein M, Vogel M, Mueller M, Aschoff P, Beyer T, et al. Optimized contrast-enhanced CT protocols for diagnostic whole-body 18F-FDG-PET/CT: technical aspects of single-phase versus multi-phase CT imaging. J Nucl Med 2006;47:470–476
Mountain CF. Revisions in the international system for staging lung cancer. Chest 1997;111:1710–1717
Glazer GM, Gross BH, Quint LE, Francis IR, Bookstein FL, Orringer MB. Normal mediastinal lymph nodes: number and size according to American Thoracic Society mapping. AJR 1985;144:261–265
Schöder H, Erdi YE, Larson SM, Yeung HWD. PET/CT: a new imaging technology in nuclear medicine. Eur J Nucl Med Mol Imaging 2003;30:1419–1437
Beyer T, Antoch G, Müller S, Egelhof T, Freudenberg LS, Debatin J, et al. Acquisition protocol considerations for combined PET/CT imaging. J Nucl Med 2004;45 (suppl):25S–35S
Beyer T, Antoch G, Bockisch A, Stattaus J. Optimized intravenous contrast administration for diagnostic whole-body 18F-FDG PET/CT. J Nucl Med 2005;46:429–435
Detterbeck FC, Jones DR, Kernstine KH, Naunheim KS; American College of Physicians. Lung cancer. Special treatment issues. Chest 2003;123:244S–258S
Brianzoni E, Rossi G, Ancidei S, Berbellini A, Capoccetti F, Cidda C, et al. Radiotherapy planning: PET/CT scanner performances in the definition of gross tumour volume and clinical target volume. Eur J Nucl Med Mol Imaging 2005;32:1392–1399
Bar-Shalom R, Yefremov N, Guralnik L, Gaitini D, Frenkel A, Kuten A, et al. Clinical performance of PET/CT in evaluation of cancer: additional value for diagnostic imaging and patient management. J Nucl Med 2003;44:1200–1209
Berthelsen AK, Holm S, Loft A, Klausen TL, Andersen F, Højgaard L. PET/CT with intravenous contrast can be used for PET attenuation correction in cancer patients. Eur J Nucl Med Mol Imaging 2005;32:1167–7511
Kapoor V, McCook BM, Torok FS. An introduction to PET-CT imaging. RadioGraphics 2004;24:523–543
Schoeder H, Yeung HWD, Larson SM. CT in PET/CT:essential features of interpretation. J Nucl Med 2005;46:1249–1251
Osman MM, Cohade C, Fishman EK, Wahl RL. Clinically significant incidental findings on the unenhanced CT portion of PET/CT studies; frequency in 250 patients. J Nucl Med 2005;46:1352–1355
Acknowledgements
We would like to thank our technicians Henriette Heners, Sylvia Storz, Benjamin Gerras, Anke Reckwell, Gabi Boutin and Tina Brutschy for their helpful assistance in the acquisition of the PET/CT data. We gratefully acknowledge the continuing support of our colleagues of the Radiopharmacy Department.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pfannenberg, A.C., Aschoff, P., Brechtel, K. et al. Low dose non-enhanced CT versus standard dose contrast-enhanced CT in combined PET/CT protocols for staging and therapy planning in non-small cell lung cancer. Eur J Nucl Med Mol Imaging 34, 36–44 (2007). https://doi.org/10.1007/s00259-006-0186-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-006-0186-3