Abstract
Purpose
Hypoxia is associated with tumor aggressiveness and is an important cause of resistance to radiation therapy and chemotherapy. Assays of tumor hypoxia could provide selection tools for hypoxia-modifying treatments. The purpose of this study was to develop and characterize a rodent tumor model with a reporter gene construct that would be transactivated by the hypoxia-inducible molecular switch, i.e., the upregulation of HIF-1.
Methods
The reporter gene construct is the herpes simplex virus 1-thymidine kinase (HSV1-tk) fused with the enhanced green fluorescent protein (eGFP) under the regulation of an artificial hypoxia-responsive enhancer/promoter. In this model, tumor hypoxia would up-regulate HIF-1, and through the hypoxia-responsive promoter transactivate the HSV1-tkeGFP fusion gene. The expression of this reporter gene can be assessed with the 124I-labeled reporter substrate 2′-fluoro-2′-deoxy-1-β-d-arabinofuranosyl-5-iodouracil (124I-FIAU), which is phosphorylated by the HSV1-tk enzyme and trapped in the hypoxic cells. Animal positron emission tomography (microPET) and phosphor plate imaging (PPI) were used in this study to visualize the trapped 124I-FIAU, providing a distribution of the hypoxia-induced molecular events. The distribution of 124I-FIAU was also compared with that of an exogenous hypoxic cell marker, 18F-fluoromisonidazole (FMISO).
Results
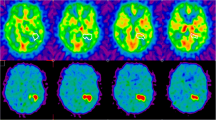
Our results showed that 124I-FIAU microPET imaging of the hypoxia-induced reporter gene expression is feasible, and that the intratumoral distributions of 124I-FIAU and 18F-FMISO are similar. In tumor sections, detailed radioactivity distributions were obtained with PPI which also showed similarity between 124I-FIAU and 18F-FMISO.
Conclusion
This reporter system is sufficiently sensitive to detect hypoxia-induced transcriptional activation by noninvasive imaging and might provide a valuable tool in studying tumor hypoxia and in validating existing and future exogenous markers for tumor hypoxia.




Similar content being viewed by others
References
Thrall DE, Rosner GL, Azuma C, McEntee MC, Raleigh JA. Hypoxia marker labeling in tumor biopsies: quantification of labeling variation and criteria for biopsy sectioning. Radiother Oncol 1997;53:171–6. DOI 10.1016/S0167-8140(97)01931-2
Brizel DM, Dodge RK, Clough RW, Dewhirst MW. Oxygenation of head and neck cancer: changes during radiotherapy and impact on treatment outcome. Radiother Oncol 1999;53:113–7. DOI 10.1016/S0167-8140(99)00102-4
Höckel M, Schlenger K, Aral B, Mitze M, Schäffer U, Vaupel P. Association between tumor hypoxia and malignant progression in advanced cancer of the uterine cervix. Cancer Res 1996;56:4509–15.
Nordsmark M, Overgaard J. A confirmatory prognostic study on oxygenation status and loco-regional control in advanced head and neck squamous cell carcinoma treated by radiation therapy. Radiother Oncol 2000;57:39–43. DOI 10.1016/S0167-8140(00)00223-1
Brizel DM, Scully SP, Harrelson JM, Layfield LJ, Bean JM, Prosnitz LR, Dewhirst MW. Tumor oxygenation predicts for the likelihood of distant metastases in human soft tissue sarcoma. Cancer Res 1996;56:941–3.
Dehdashti F, Grigsby PW, Mintun MA, Lewis JS, Siegel BA, Welch MJ. Assessing tumor hypoxia in cervical cancer by positron emission tomography with 60Cu-ATSM: relationship to therapeutic outcome—a preliminary report. Int J Radiat Oncol Biol Phys 2003;55:1233–8. DOI 10.1016/S0360-3016(02)04477-2
Dehdashti F, Mintun MA, Lewis JS, Bradley J, Govindan R, Laforest R, et al. In vivo assessment of tumor hypoxia in lung cancer with 60Cu-ATSM. Eur J Nucl Med Mol Imaging 2003;30:844–50. DOI 10.1007/s00259-003-1130-4
Chapman JD, Zanzonico P, Ling CC. On measuring hypoxia in individual tumors with radiolabeled agents. J Nucl Med 2001;42:1653–5.
Höckel M, Knoop C, Schlenger K, Vorndran B, Knapstein PG, Vaupel P. Intratumoral pO2 histography as predictive assay in advanced cancer of the uterine cervix. Adv Exp Med Biol 1994;345:445–50.
Loncaster JA, Harris AL, Davidson SE, Logue JP, Hunter RD, Wycoff CC, et al. Carbonic anhydrase (CA IX) expression, a potential new intrinsic marker of hypoxia: correlations with tumor oxygenation measurements and prognosis in locally advanced carcinoma of the cervix. Cancer Res 2001;61:6394–9.
Nordsmark M, Bentzen SM, Overgaard J. Measurement of human tumour oxygenation status by a polarographic needle electrode. Acta Oncol 1994;33:383–9.
Nordsmark M, Loncaster J, Chou S-C, Havsteen H, Lindegaard JC, Davidson SE, et al. Invasive oxygen measurements and pimonidazole labeling in human cervix carcinoma. Int J Radiat Oncol Biol Phys 2001;49:581–6. DOI 10.1016/S0360-3016(00)01493-0
Rasey JS, Koh WJ, Evans ML, Peterson LM, Lewellen TK, Grahm MM, Krohn KA. Quantifying regional hypoxia in human tumors with positron emission tomography of [18F]fluoromisonidazole: a pretherapy study of 37 patients. Int J Radiat Oncol Biol Phys 1996;36:417–28. DOI 10.1016/S0360-3016(96)00325-2
West CML, Cooper RA, Loncaster JA, Wilks DP, Bromley M. Tumor vascularity: a histological measure of angiogenesis and hypoxia. Cancer Res 2001;61:2907–10.
Lehtio K, Oikonen V, Gronroos T, Eskola O, Kalliokoski K, Bergman J, et al. Imaging of blood flow and hypoxia in head and neck cancer: initial evaluation with [15O]H2O and [18F]fluoroerythronitroimidazole PET. J Nucl Med 2001;42:1653–5.
Dolbier WRJ, Li AR, Koch CJ, Shiue CY, Kachur AV. [18F]-EF5, a marker for PET detection of hypoxia: synthesis of precuror and a new fluorination procedure. Appl Radiat Isot 2001;54:73–80.
Chao KSC, Bosch WR, Mutic S, Lewis JS, Dehdashti F, Mintun MA, et al. A novel approach to overcome hypoxic tumor resistance: Cu-ATSM-guided intensity-modulated radiation therapy. Int J Radiat Oncol Biol Phys 2001;49:1171–82.
Bentzen L, Keiding S, Nordsmark M, Falborg L, Hansen SB, Keller J, et al. Tumour oxygenation assessed by 18F-fluoromisonidazole PET and polarographic needle electrodes in human soft tissue tumours. Radiother Oncol 2003;67:339–44.
Semenza GL. HIF-1, O2, and the 3 PHDs: how animal cells signal hypoxia to the nucleus. Cell 2001;107:1–3. DOI 10.1016/S0092-8674(01)00518-9
Semenza GL. Targeting HIF-1 for cancer therapy. Nat Rev Cancer 2003;3:721–82. DOI 10.1038/nrc1187
Yancopoulos GD, Davis S, Gale NW, Rudge JS, Wiegand SJ, Holash J. Vascular-specific growth factors and blood vessel formation. Nature 2000;407:242–8. DOI 10.1038/35025215
Yuan J, Narayanan L, Rockwell S, Glazer PM. Diminished DNA repair and elevated mutagenesis in mammalian cells exposed to hypoxia and low pH. Cancer Res 2000;60:4372–6.
Rofstad EK. Microenvironment-induced cancer metastasis. Int J Radiat Biol 2000;76:589–605.
Chavez JC, LaManna JC. Activation of hypoxia-inducible factor-1 in the rat cerebral cortex after transient global ischemia: potential role of insulin-like growth factor-1. J Neurosci 2002;22:8922–31.
Airley RE, Loncaster J, Raleigh JA, Harris AL, Davidson SE, Hunter RD, et al. Glut-1 and CAIX as intrinsic markers of hypoxia in carcinoma of the cervix: relationship to pimonidazole binding. Int J Cancer 2003;104:85–91.
Vordermark D, Brown JM. Evaluation of hypoxia-inducible factor-1α (HIF-1α) as an intrinsic marker of tumor hypoxia in U87 MG human glioblastoma: in vitro and xenograft studies. Int J Radiat Oncol Biol Phys 2003;56:1184–93.
Serganova I, Doubrovin M, Vider J, Ponomarev V, Soghomonyan S, Beresten T, et al. Molecular imaging of spatial heterogeneity and temporal dynamics of hypoxia-induced HIF-1 signal transduction activity in tumors in living mice. Cancer Res 2004;64:6101–8.
Tjuvajev JG, Stockhammer G, Desi R, Uehara H, Watanabe K, Gansbacher B, Blasberg RG. Imaging the expression of transfected genes in vivo. Cancer Res 1995;55:1352–60.
Tjuvajev JG, Avril N, Oku T, Sasajima T, Miyagawa T, Joshi R, et al. Imaging herpes virus thymidine kinase gene transfer and expression by positron emission tomography. Cancer Res 1998;58:4333–41.
Ponomarev V, Doubrovin M, Lyddane C, Beresten T, Balatoni J, Bornman W, et al. Imaging TCR-depenent NFAT-mediated T-cell activation with positron emission tomography in vivo. Neoplasia 2001;3:480–6. DOI 10.1038/sj.1522-8002.7900204
Doubrovin M, Ponomarev V, Serganova I, Soghomonian S, Myagawa T, Beresten T, et al. A new reporter gene system for molecular-genetic imaging of CNS with intact blood–brain barrier: bacterial xanthine phosphoribosyl transferase-radiolabeled xanthine. Mol Imaging 2003;2:36–48.
Riviere I, Sadelain M. In: Robbins P, editor. Gene therapy protocols: methods in molecular biology. Totowa: Humana; 1997. p. 59–78.
Gallardo HF, Tan C, Ory D, Sadelain M. Recombinant retroviruses pseudotyped with the vesicular stomatitis virus G glycoprotein mediate both stable gene transfer and pseudotransduction in human peripheral blood lymphocytes. Blood 1997;90:952–7.
Zanzonico P, O’Donoghue J, Chapman JD, Schneider R, Cai S, Larson S, et al. Iodine-124-labeled iodo-azomycin-galactoside imaging of tumor hypoxia in mice with serial microPET scanning. Eur J Nucl Med Mol Imaging 2004;31:117–28. DOI 10.1007/s00259-003-1322-y
Tjuvajev JG, Finn R, Watanabe K, Joshi R, Oku T, Kennedy J, et al. Noninvasive imaging of herpes virus thymidine kinase gene transfer and expression: a potential method for monitoring clinical gene therapy. Cancer Res 1996;56:4087–95.
Hackman T, Doubrovin M, Balatoni J, Beresten T, Ponomarev V, Beattie B, et al. Imaging expression of cytosine deaminase-herpes virus thymidine kinase fusion gene expression with [124I]FIAU and PET. Mol Imaging 2002;1:36–42.
Lim JL, Berridge MS. An efficient radiosynthesis of [18F]fluoromisonidazole. Appl Radiat Isot 1993;44:1085–91.
Balatoni J, Finn R, Tjuvajev J, Larson S, Blasberg R. Synthesis and quality assurance of radioiodinated 2-fluoro-2-deoxy-1-d-arabinofuranosyl-5-iodouracil. J Labelled Comp Radiopharm 1997;103:4087–95.
Martin GV, Caldwell JH, Graham MM, Grierson JR, Kroll K, Cowan MJ, et al. Noninvasive detection of hypoxic myocardium using fluorine-18-fluoromisonidazole and positron emission tomography. J Nucl Med 1992;33:2202–8.
Rasey JS, Koh WJ, Grierson JR, Grunbaum Z, Krohn KA. Radiolabelled fluoromisonidazole as an imaging agent for tumor hypoxia. Int J Radiat Oncol Biol Phys 1989;17:985–91.
Acknowledgment
This work was supported in part by grant RO1 CA84596 from the NCI, National Institutes of Health, USA.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wen, B., Burgman, P., Zanzonico, P. et al. A preclinical model for noninvasive imaging of hypoxia-induced gene expression; comparison with an exogenous marker of tumor hypoxia. Eur J Nucl Med Mol Imaging 31, 1530–1538 (2004). https://doi.org/10.1007/s00259-004-1673-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-004-1673-z