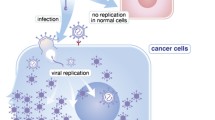
Abstract
Medullary thyroid carcinoma (MTC), a neoplasm of thyroid C-cells, is characterized by dominant activating mutations in the RET proto-oncogene. Currently therapy is restricted to surgical removal of all neoplastic tissue lacking alternative forms of treatment such as chemotherapy or radiotherapy. Therefore MTC is a particularly attractive target for gene therapeutic approaches. Many promising gene therapy strategies have been used in various animal models of MTC, showing enhanced antitumoral efficacy, and these will hopefully extend our current standard of care in the future. These approaches can tentatively be subdivided into four groups: (a) Inhibition of oncogenic RET signaling, (b) suicide gene therapy, (c) immunotherapy, and (d) combination of immunotherapy and suicide approaches. To block oncogenic signal transduction dominant-negative RET mutants were delivered into tumor cells and found to possess strong antineoplastic activity, including tumor growth suppression and increased animal survival. Suicide gene therapeutic approaches applied to MTC treatment featured either gene transfer of herpes simplex virus thymidine kinase with concomitant application of ganciclovir or delivery of nitric oxide synthase II. Here antitumor effects were attributed to the occurrence of substantial bystander activities. Immunotherapy approaches comprised stimulation of immune response by delivery of interleukin 2 or 12. Finally, treatment with herpes simplex virus thymidine kinase/ganciclovir in combination with interleukin 2 was found to be superior over either treatment alone. This review discusses the various gene therapeutic approaches applied to MTC treatment in detail, gives an overview on the diverse vector systems used to achieve efficient transduction of thyroid cancer cells, and points out the strategies employed to accomplish target cell selective gene expression thereby contributing to enhanced safety of gene therapy for MTC

Similar content being viewed by others
Abbreviations
- GCV :
-
Ganciclovir
- HSV-TK :
-
Herpes simplex virus thymidine kinase
- IL :
-
Interleukin
- MEN :
-
Multiple endocrine neoplasia
- MTC :
-
Medullary thyroid carcinoma
- NK :
-
Natural killer
- NOS II :
-
Nitric oxide synthase II
- RET :
-
Rearranged during transfection
References
Vitale G, Caraglia M, Ciccarelli A, Lupoli G, Abbruzzese A, Tagliaferri P, Lupoli G (2001) Current approaches and perspectives in the therapy of medullary thyroid carcinoma. Cancer 91:1797–1808
Hundahl SA, Fleming ID, Fremgen AM, Menck HR (1998) A national cancer database report on 53856 cases of thyroid carcinoma treated in the US 1985–1995. Cancer 83:2638–2648
Eng C (1999) RET proto-oncogene in the development of human cancer. J Clin Oncol 17:380–393
Hazard JB (1977) The C-cells (parafollicular cells) of the thyroid gland and medullary thyroid carcinoma. J Pathol 88:213–250
Orlandi F, Caraci P, Mussa A, Saggiorato E, Pancani G, Angeli A (2001) Treatment of medullary thyroid carcinoma: an update. Endocr Relat Cancer 8:135–147
Ponder BA, Smith D (1996) The MEN II syndromes and the role aof the ret proto-oncogene. Adv Cancer Res 70:179–222
Mulligan LM, Kwok JBJ, Healey CS, Elsdon MJ, Eng C, Gardner E, Love DR, Mole SE, Moore JK, Papi L, Ponder MA, Telenius H, Tunnacliffe A, Ponder BA (1993) Germ-line mutations of the RET proto-oncogene in multiple endocrine neoplasia type 2A. Nature 363:548–560
Takahashi M (2001) The RET/GDNF signaling pathway and human diseases. Cytokine Growth Factor Rev 12:361–373
Asai N, Iwashita T, Matsuyama M, Takahashi M (1995) Mechanism of activation of ret proto-oncogene by multiple endocrine neoplasia 2A mutation. Mol Cell Biol 15:1613–1619
Gimm O (2001) Thyroid cancer. Cancer Lett 163:143–156
Frilling A, Weber F, Tecklenborg C, Broelsch CE (2003) Prophylactic thyroidectomy in multiple endocrine neoplasia: the impact of molecular mechanisms of the RET proto-oncogene. Langenbecks Arch Surg 388:17–26
Schmutzler C, Koehrle J (2000) Innovative strategies for the treatment of thyroid cancer. Eur J Endocrinol 143:15–24
Willis AC, Chen X (2002) The promise and obstacle of p53 as a cancer therapeutic agent. Curr Mol Med 2:329–345
Herfarth KKF, Wick MR, Marshall HN, Gartner E, Lum S, Moley JF (1997) Absence of TP53 alterations in pheochromocytomas and medullary thyroid carcinomas. Genes Chromosom Cancer 20:24–29
Yane K, Konishi N, Kitahori Y, Naito H, Koaichi K, Ohnishi T, Miyahara H, Matsunaga T, Hiasa Y (1996) Lack of p16/CKDN2 alterations in thyroid carcinomas. Cancer Lett 101:85–92
Segouffin-Cariou C, Billaud M (2000) Transforming ability of MEN2A-RET requires activation of the phosphatidylinositol 3-kinase/AKT signaling pathway. J Biol Chem 275:3568–3576
Vivanco I, Sawyers CL (2002) The phosphatidyl 3-kinase Akt pathway in human cancer. Nat Rev Cancer 2:489–501
Weering DHJ van, Bos JL (1998) Signal transduction by the receptor tyrosine kinase RET. Recent Results Cancer Res 154:271–281
Marshall GM, Peaston AE, Hocker JE, Smith SA, Hansford LM, Tobias V, Norris MD, Haber M, Smith DP, Lorenzo MJ, Ponder BA, Hancock JF (1997) Expression of multiple endocrine neoplasia 2B RET in neuroblastoma cells alters cell adhesion in vitro, enhances metastatic behaviour in vivo and activates Jun kinase. Cancer Res 57:5399–5405
Lui VW, He Y, Huang L (2001) Specific down-regulation of HER-2/neu mediated by a chimeric U6 hammerhead ribozyme results in growth inhibition of human ovarian carcinoma. Mol Ther 3:169–177
Chin L, Tam A, Pomerantz J, Wong M, Holash J, Bardeesy M, Shen Q, O'Hagan R, Pantginis J, Zhou H, Horner JW, Cordon-Cardo C, Yancopoulos GD, DePinho RA (1999) Essential role for oncogenic Ras in tumor maintenance. Nature 400:468–472
Drosten M, Frilling A, Stiewe T, Pützer BM (2002) A new therapeutic approach in medullary thyroid cancer treatment: inhibition of oncogenic RET signaling by adenoviral vector-mediated expression of a dominant-negative RET mutant. Surgery 132:991–997
Drosten M, Stiewe T, Pützer BM (2003) Antitumor capacity of a dominant-negative RET proto-oncogene mutant in a medullary thyroid carcinoma model. Hum Gene Ther (in press)
Cosma MP, Cardone M, Carlomagno F, Colantuoni V (1998) Mutations in the extracellular domain cause RET loss of function by a dominant negative mechanism. Mol Cell Biol 18:3321–3329
Messina M, Yu DM, Learoyd DL, Both GW, Molloy PL, Robinson BG (2000) High level, tissue-specific expression of a modified calcitonin/calcitonin gene-related peptide promoter in a human medullary thyroid carcinoma cell line. Mol Cell Endocrinol 164:219–224
Parthasarathy R, Cote GJ, Gagel RF (1999) Hammerhead ribozyme-mediated inactivation of mutant RET in medullary thyroid carcinoma. Cancer Res 59:3911–3914
Hennige AM, Lammers R, Höppner W, Arlt D, Strack V, Teichmann R, Machicao F, Ullrich A, Häring HU, Kellerer M (2001) Inhibition of Ret oncogene activity by the protein tyrosine phosphatase SHP1. Endocrinology 143:4441–4447
Qiao S, Iwashita T, Furukawa T, Yamamoto M, Sobue G, Takahashi M (2001) Differential effects of leukocyte common antigen-related protein on biochemical and biological activities of RET-MEN2A and RET-MEN2B mutant proteins. J Biol Chem 276:9460–9467
Aghi M, Hochberg F, Breakefield XO (2000) Prodrug activation enzymes in cancer gene therapy. J Gene Med 2:148–164
Molten FL (1986) Tumor chemosensitivity conferred by inserted herpes thymidine kinase genes: a paradigm for a prospective cancer control strategy. Cancer Res 46:5276–5281
Fillat C, Carrio M, Cascante A, Sangro B (2003) Suicide gene therapy mediated by the Herpes simplex virus thymidin kinase gene/Ganciclovir system: fifteen years of application. Curr Gene Ther 3:13–26
Dillen IJ van, Mulder NH, Vaalburg V, de Vries EF, Hospers GA (2002) Influence of the bystander on HSV-tk/GCV gene therapy. A review. Curr Gene Ther 2:307–322
Ishii-Morita H, Agbaria R, Mullen CA, Hirano H, Koeplin DA, Ram Z, Oldfield EH, Johns DG, Blaese RM (1997) Mechanism of 'bystander effect' killing in the herpes simplex virus thymidine kinase gene therapy model of cancer treatment. Gene Ther 4:244–251
Fick J, Barker FG, Dazin P, Westphale EM, Beyer EC, Isreal MA (1995) The extent of heterocellular communication mediated by gap junctions is predictive of bystander tumor cytotoxicity in vitro. Proc Natl Acad Sci USA 92:11071–11075
Minemura K, Takeda T, Minemura K, Nagasawa T, Zhang R, Leopardi R, DeGroot LJ (2000) Cell-specific induction of sensitivity to ganciclovir in medullary thyroid carcinoma by adenovirus-mediated gene transfer of Herpes simplex virus thymidine kinase. Endocrinology 141:1814–1822
Zhang R, DeGroot LJ (2000) Gene therapy of established medullary thyroid carcinoma with herpes simplex viral thymidine kinase in a rat tumor model: relationship of bystander effect and antitumor efficacy. Thyroid 10:313–319
Jiang S, Altmann A, Grimm D, Kleinschmidt JA, Schilling T, Germann C, Haberkorn U (2001) Tissue-specific gene expression in medullary thyroid carcinoma cells employing calcitonin regulatory elements and AAV vectors. Cancer Gene Ther 8:469–472
Nathan C, Xie QW (1994) Nitric oxide synthases: roles, tolls and controls. Cell 78:915–918
Drapier JC, Hibbs JB Jr (1988) Differentiation of murine macrophages to express nonspecific cytotoxicity for tumor cells results in L-arginine-dependent inhibition of mitochondrial iron-sulfur enzymes in the macrophage effector cells. J Immunol 140:2829–2838
Burney S, Tamir S, Gal A, Tannenbaum SR (1997) A mechanistic analysis of niric oxide-induced cellular toxicity. Nitric Oxide 1:130–144
Lepoivre M, Chenais B, Yapo A, Lemaire G, Thelander L, Tenu JP (1990) Alterations of ribonucleotide activity following induction of the nitrite-generation pathway in adenocarcinoma cells. J Biol Chem 265:14143–14149
Lancaster JF Jr (1994) Stimulation of the diffusion and reaction of the endogenously produced nitric oxide. Proc Natl Acad Sci USA 91:8137–8141
Schwentker A, Billiar TR (2002) Inducible nitric oxide synthase: from cloning to therapeutic applications. World J Surg 26:772–778
Soler MN, Bobe P, Benihoud K, Lemaire G, Roos BA, Lausson S (2000) Gene therapy of rat medullary thyroid cancer by naked nitric oxide synthase II DNA injection. J Gene Med 2:344–352
Messina M, Yu DM, Both GW, Molloy PL, Robinson BG (2003) Calcitonin-specific transcription and splicing targets gene-directed enzyme prodrug therapy to medullary thyroid carcinoma cells. J Clin Endocrinol Metab 88:1310–1318
Parker WB, Allan PW, Shaddix SC, Rose LM, Speegle HF, Gillespie GY, Bennett LL Jr (1998) Metabolism and metabolic actions of 6-methylpurine and 2-fluoroadenine inhuman cells. Biochem Pharmacol 55:1673–1681
Khong HP, Restifo NP (2002) Natural selection of tumor variants in the generation of 'tumor escape' phenotypes. Nat Immunol 3:999–1005
Dunn GP, Bruce AT, Ikeda H, Old LJ, Schreiber RD (2002) Cancer immunoediting: from immunosurveillance to tumor escape. Nat Immunol 3:991–998
Parney IF, Chang LJ (2003) Cancer immunogene therapy: a review. J Biomed Sci 10:37–43
Haupt K, Siegel F, Lu M, Yang D, Hilken G, Mann K, Roggendorf M, Saller B (2001) Induction of a cellular and humoral immune response against Preprocalcitonin by genetic immunisation: a potential new treatment for medullary thyroid carcinoma. Endocrinology 142:1017–1023
Addison CL, Braciak T, Ralston R, Muller WJ, Gauldie J, Graham FL (1995) Intratumoral injection of an adenovirus encoding interleukin-2 induces regression an immunity in a murine breast cancer model. Proc Natl Acad Sci USA 92:8522–8526
Rosenberg SA (2001) Progress in human tumour immunology and immunotherapy. Nature 411:380–384
Lausson S, Fournes B, Borrel C, Milhaud G, Treilhou-Lahille F (1996) Immune response against medullary thyroid carcinoma (MTC) induced by parental and/or interleukin-2-secreting MTC cells in a rat model of human familial medullary thyroid carcinoma. Cancer Immunol Immunother 43:116–123
Zhang R, Minemura K, DeGroot LJ (1998) Immunotherapy for medullary thyroid carcinoma by a replication-defective adenovirus transducing murine interleukin-2. Endocrinology 139:601–608
Zhang R, Baunoch D, DeGroot LJ (1998) Genetic immunotherapy for medullary thyroid carcinoma: destruction of tumors in mice by in vivo delivery of adenoviral vector transducing the murine interleukin-2 gene. Thyroid 8:1137–1146
Zhang R, Straus FH, DeGroot LJ (1999) Effective genetic therapy of established medullary thyroid carcinomas with murine interleukin-2: dissemination and cytotoxicity studies in a rat tumor model. Endocrinology 140:2152–2158
Wolf SF, Temple PA, Kobayashi M, Young D, Dicig M, Lowe L, Dzialo R, Fitz L, Hewick RM, Kelleher K, Herrmann SH, Clark SC, Azzoni L, Chan SH, Trinchieri G, Perussia B (1991) Cloning of cDNA for natural killer cell stimulating factor, a heterodimeric cytokine with multiple biologic effects on T and natural killer cells. J Immunol 146:3074–3081
Trinchieri G (1995) Interleukin-12: a proinflammatory cytokine with immunoregulatory function that bridge innate resistance and antigen-specific adaptive immunity. Annu Rev Immunol 13:251–276
Zitvogel L, Tahara H, Cai Q, Storkus W, Muller G, Wolf SF, Gately M, Robbins PD, Lotze MT (1994) Construction and characterization of retroviral vectors expressing biologically active human interleukin-12. Hum Gene Ther 5:1493–1506
Melero I, Mazzolini G, Narvaiza I, Qian C, Chen L, Prieto J (2001) IL-12 gene therapy for cancer: in synergy with other immunotherapies. Trends Immunol 22:113–115
Orange JS, Salazar-Mather TP, Opal SM, Spencer RL, Miller AH, McEwan BS, Biron CA (1995) Mechanism of interleukin 12-mediated toxicities during experimental viral infections: role of tumor necrosis factor and glucocorticoids. J Exp Med 181:901–914
Zhang R, DeGroot LJ (2000) Genetic immunotherapy of established tumours with adenoviral vectors transducing murine interleukin-12 (mIL-12) subunits in a rat medullary thyroid carcinoma model. Clin Endocrinol (Oxf) 52:687–694
Yamazaki M, Zhang R, Straus FH, Messina M, Robinson BG, Hashizume K, DeGroot LJ (2002) Effective gene therapy for medullary thyroid carcinoma using recombinant adenovirus inducing tumor-specific expression of interleukin-12. Gene Ther 9:64–74
Soler MN, Milhaud G, Lekmine F, Treilhou-Lahille F, Klatzmann D, Lausson S (1999) Treatment of medullary thyroid carcinoma by combined expression of suicide and interleukin-2 genes. Cancer Immunol Immunother 48:91–99
Zhang R, DeGroot LJ (2001) An adenoviral vector expressing functional heterogeneous proteins herpes simplex viral thymidine kinase and human interleukin-2 has enhanced in vivo antitumor activity against medullary thyroid carcinoma. Endocr Relat Cancer 8:315–325
Hinze R, Gimm O, Taubert H, Bauer G, Dralle H, Holzhausen HJ, Rath FW (2000) Regulation of proliferation and apoptosis in sporadic and heretitary medullary thyroid carcinomas and their putative precurser lesions. Virchows Arch 437:256–263
Shen C, Buck AK, Liu X, Winkler M, Reske SN (2003) Gene silencing by adenovirus-delivered siRNA. FEBS Lett 539:111–114
Cohen MS, Hussain HB, Moley JF (2002) Inhibition of medullary thyroid carcinoma cell proliferation and RET phosphorylation by tyrosine kinase inhibitors. Surgery 132:960–966
Carlomagno F, Vitagliano D, Guida T, Ciardiello F, Tortora G, Vecchio G, Ryan AJ, Fontanini G, Fusco A, Santoro M (2002) ZD6474, an orally available inhibitor of KDR tyrosine kinase activity, efficiently blocks oncogenic RET kinases. Cancer Res 62:7284–7290
Stewart AK, Lassam NJ, Quirt IC, Bailey DJ, Rotstein LE, Krajden M, Dessureault S, Gallinger S, Cappe D, Wan Y, Addison CL, Moen RC, Gauldie J, Graham FL (1999) Adenovector-mediated gene delivery of interleukin-2 in metastatic breast cancer and melanoma: results of a phase 1 clinical trial. Gene Ther 6:350–363
DeGroot LJ, Zhang R (2001) Clinical review 131. Gene Therapy for thyroid cancer: where do we stand? J Clin Endocrinol Metab 86:2923–2928
Scappaticci FA (2002) Mechanisms and future directions for angiogenesis based-cancer therapies. J Clin Oncol 20:3906–3927
Chiocca EA (2002) Oncolytic viruses. Nat Rev Cancer 2:938–950
Nemunaitis J, Ganly I, Khuri F, Arseneau J, Kuhn J, McCarty T, Landers S, Maples P, Romel L, Randlev B, Reid T, Kaye S, Kirn D (2000) Selective replication and oncolysis in p53 mutant tumors with ONYX-015, an E1B55kD gene-deleted adenovirus, in patients with advanced head and neck cancer: a phase II trial. Cancer Res 60:6359–6366
Rothmann T, Hengstermann A, Whitaker NJ, Scheffner M, zur Hausen H (1998) Replication of ONYX-015, a potential anticancer adenovirus, is independent of p53 status in tumor cells. J Virol 72:9470–9478
Balague C, Noya F, Alemany R, Chow LT, Curiel DT (2001) Human papillomavirus E6E7-mediated adenovirus cell killing: selectivity of mutant adenovirus replication in organotypic cultures of human keratinocytes. J Virol 75:7602–7611
Li Y, Yu DC, Chen Y, Amin P, Zhang H, Nguyen N, Henderson DR (2001) A hepatocellular carcinoma-specific adenovirus variant, CV890, eliminates distant human liver tumors in combination with doxorubicin. Cancer Res 61:6428–6436
Wickham TJ (2003) Ligand-directed targeting of genes to the site of disease. Nat Med 9:135–139
Nicklin SA, Baker AH (2002) Tropism-modified adenoviral and adeno-associated viral vectors for gene therapy. Curr Gene Ther 2:273–293
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Drosten, M., Pützer, B.M. Gene therapeutic approaches for medullary thyroid carcinoma treatment. J Mol Med 81, 411–419 (2003). https://doi.org/10.1007/s00109-003-0455-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-003-0455-6