Summary
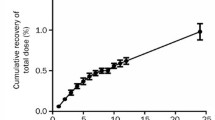
The pharmacokinetics of Indocyanine Green (ICG) has been studied in 15 patients given 0.5, 1.0 and 2.0 mg · kg−1. The plasma disappearance and biliary excretion rate were measured in patients with tightly fitting catheters under slight negative pressure in order to achieve complete collection of bile. Recovery of unchanged ICG in bile over 18 h after the i.v. injection was 80% of the dose in all three dose groups.
Plasma disappearance in all 3 groups was biphasic, showing an initial phase with a t1/2 of 3–4 min and a secondary phase with a dose-dependent apparent t1/2 of 67.6, 72.5 and 88.7 min, respectively. After 0.5 and 1.0 mg · kg−1 the biliary excretion rate curves showed an ascending phase with a mean t1/2 of 5 min and a descending phase with a mean t1/2 of 72 min. It was inferred that the secondary component of the plasma-decay mainly reflected the biliary excretion rate. After 2.0 mg · kg−1 in some patients the biliary excretion curve showed features of saturation; the t1/2 of the descending phase ranged from 73 to 440 min, and the time of maximal excretion was increased from 1.3 to 2.7 h after injection, whilst the mean maximal excretion rate was in the same range as the excretion rate after the 1.0 mg · kg−1 dose. The non-linear pharmacokinetics was only moderately reflected in the measured plasma disappearance patterns. Two compartment analysis of the plasma levels indicated a clearance of 230–260 ml · min−1, whereas the clearance conventionally calculated from the initial t1/2 was 475 ml · min−1. The volume of the central compartment in 70 kg patients was 2.31, which is about the plasma volume. The fictive volume of distribution in the liver (V2) was 70–90 l, indicating marked hepatic storage of ICG. This was probably due to the very low liver-to-plasma transport rate (k21) of 0.006–0.10 min−1. Thus, the biliary excretion of ICG can be quantified by 2-compartment pharmacokinetic analysis of plasma disappearance curves, including a secondary phase. The latter, slow component was apparent at very low plasma levels due to the marked hepatic storage, and it was also influenced by retention of small amounts of impurities or degradation products. Improved detectability of this phase cannot simply be obtained by increasing the dose, since at doses exceeding 1.0 mg · kg−1 non-linear elimination may complicate the pharmacokinetic analysis.
Similar content being viewed by others
References
Baker KJ, Bradley SE (1966) Binding of sulphobromophthalein (BSP) sodium and indocyanine green (ICG) by plasma α-lipoproteins. Proc Soc Exptl Biol Med 122: 957–963
Björnsson OG, Murphy R, Chadwick VS (1982) Physiochemical studies of indocyanine green (ICG): Absorbance/-concentration relationship, pH tolerance and assay precision in various solvents. Experienta 38: 1441–1442
Branch RA, James JA, Read AC (1976) The clearance of antipyrine and indocyanine green in normal subjects and in patients with chronic liver disease. Clin Pharmacol Ther 20: 81–89
Capron J, Erlinger E, Feldmann G (1975) Increased sulphobromophthalein clearance in a patient receiving phenobarbital and other anticonvulsant drugs. Gastroenterology 69: 756–760
Carson ER, Jones CA (1979) Use of kinetic analysis and mathematical modelling in the study of metabolic pathways in vivo. Application to hepatic organic anion metabolism. N Engl J Med 300: 1016–1027
Cherrick GR, Stein WS, Levy CM, Davidson CS (1960) Indocyanine green: observations on its physical properties, plasma decay and hepatic extraction. J Clin Invest 39: 592–600
Donn KH, Powell JR, Rogers JF, Plachetta JR (1984) Lack of histamine H2-receptor antagonists on indocyanine green disposition measured by two methods. J Clin Pharmacol 24: 300–370
Dunk AA, Jenkins WJ, Burroughs AK, Walt RP, Osuafor TOK, Sherlock S, Mackie S, Dick R (1983) The effect of ranitidine on the plasma clearance and hepatic extraction of indocyanine green in patients with chronic liver disease. Br J Clin Pharmacol 10: 117–120
Erlinger E, Dhumeaux MD, Desjeux JF, Benkamou JP (1973) Hepatic handling of unconjugated dyes in the Dubin-Johnson syndrome. Gastroenterology 64: 106–110
Gentile S, Bajema BL, Baldini G, Lunazzi G, Groothuis GMM, Tiribelli C, Meijer DKF, Sottocasa GL (1985) Measurement of the association of cholephilic organic anions with different binding proteins. Biochem Pharmacol 34: 2439–2444
Groszmann RJ (1983) The measurement of liver blood flow using clearance techniques. Hepatology 3: 1039–1040
Heintz R, Svensson CK, Stoeckel K, Bowers GJ, Lalka D (1986) Indocyanine green: Pharmacokinetics in the rabbit and relevant studies of its stability and purity. J Pharm Sci 45: 398–402
Kurtz W, Kojouharoff G, Leuschner U (1986) A 60-minute indocyanine green test with two compartment analysis. Gastroenterology 90: 1740 (abstract)
Levy CM, Bender J, Silverberg M, Naylor J (1963) Physiology of dye extraction by the liver: Comparative studies on sulfobromophthalein and indocyanine green. Ann NY Acad Sci 111: 161–174
Martin JF, Vierling JM, Wolkoff AW, Scharschmidt BF, Vergalla J, Waggoner JG, Berk PD (1976) Abnormal hepatic transport of indocyanine green in Gilbert's syndrome. Gastroenterology 70: 385–391
Meijer DKF, Weitering JG, Vermeer GA (1983) Pharmacokinetics of biliary excretion in man V.Dibrosulfophthalein. Eur J Pharmacol 24: 549–556
Meijer DKF, Blom A, Weitering JG, Hornsveld R (1984) Pharmacokinetics of the hepatic transport of organic anions: influence of extra- and intracellular binding on hepatic storage of dibrosulfophthalein and interactions with indocyanine green. J Pharmacokinet Biopharm 12: 43–65
Mikulecky M (1976) Indocyanine green plasma clearance in normal human males and females. Acta Med Acad Sci Hung 33: 133–141
Ohkubo H, Hizotaka M, Okuda K (1978) Effects of caloric restriction on the kinetics of indocyanine green in patients with liver diseases and in the rat. Am J Dig Dis 23: 1017–1024
Okuda K, Ohkubo H, Musha H, Kotoda K, Abe H, Tanikawa K (1976) Marked delay of indocyanine green plasma clearance with near-normal bromosulphophthalein retention test: A constitutional abnormality? Gut 17: 588–594
Paumgartner G, Probst P, Kruines R, Levy CM (1970) Kinetics of indocyanine green removal from the blood. Ann NY Acad Sci 170: 134–147
Rappaport PL, Thiessen JJ (1982) High pressure liquid chromatographic analysis of indocyanine green. J Pharm Sci 71: 157–161
Ruifrok PG (1982) Automated pharmacokinetic analysis: Experience of a user: Biopharm Drug Dispos 3: 243–253
Schwartz RD, Sidell FR, Cucinella SA (1974) Effects of physical stress on the disposition of drugs eliminated by the liver in man. J Pharmacol Exp Ther 188: 1–7
Stoeckel K, McNamara PJ, McLean AJ, Du Souich P, Lalka D, Gibaldi M (1980) Non-linear pharmacokinetics of indocyanine green in the rabbit and rat. J Pharmacokinet Biopharm 8: 483–496
Svensson CK, Edwards DJ, Lalka D, Mauriello PM, Middleton E (1982) Comparison of chromatographic and spectrophotometric analysis of indocyanine green in plasma-following administration of multiple doses to humans. J Pharm Sci 71: 1305–1306
Villeneuve JP, Huot R, Marleau D, Huet PM (1982) The estimation of hepatic blood flow with indocyanine green: Comparison between continuous infusion and single injection methods. Am J Gastroenterol 77: 233–237
Winkler K, Larsen JA, Munkner T, Tygstrup N (1965) Determination of the hepatic blood flow in man by simultaneous use of five test substances measured in two parts of the liver. Scand J Clin Lab Invest 17: 423–432
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Meijer, D.K.F., Weert, B. & Vermeer, G.A. Pharmacokinetics of biliary excretion in man. VI. Indocyanine green. Eur J Clin Pharmacol 35, 295–303 (1988). https://doi.org/10.1007/BF00558268
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00558268