Summary
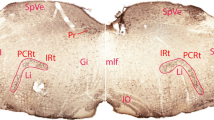
After injections in the cat of Rhodamine labelled latex microspheres in the amygdala and of Fast Blue in the cerebellum neurons labelled with one of these tracers as well as some double labelled neurons were found in the parabrachial nucleus, the nucleus locus coeruleus and some adjacent nuclei (the nucleus subcoeruleus and the pontine tegmental reticular formation). All double labelled cells were located on the ipsilateral side. A few double labelled neurons were also found bilaterally in the dorsal raphe nucleus. It therefore appears that a certain number of cerebellar projecting neurons in these brain stem nuclei by means of divergent axon collaterals also project to the amygdala. The location of the double labelled cells found in this study suggests that at least some of the neurons are catecholaminergic. The findings are related to previous reports on the distribution of catecholaminergic neurons and on the amygdaloid and cerebellar projections from this part of the brain stem, and the possible involvement of these connections in cerebellar non-somatic responses are discussed. Some comments are made concerning the use of fluorescent latex microspheres for double labelling studies in combination with another fluorescent tracer.
Similar content being viewed by others
References
Andrezik JA, Dormer KJ, Foreman RD, Person RJ (1984) Fastigial nucleus projections to the brain stem in beagles: pathways for autonomic regulation. Neuroscience 11:497–507
Batini C, Buisseret-Delmas C, Corvisier J, Hardy O, Jassik-Gerschenfeld D (1978) Brain stem nuclei giving fibers to lobulus VI and VII of the cerebellar vermis. Brain Res 153:241–261
Bentivoglio M, Kuypers HGJM, Catsman-Berrevoets CE (1980a) Retrograde neuronal labeling by means of bisbenzimide and nuclear yellow (Hoechst S769121). Measures to prevent diffusion of the tracers out of retrogradely labeled neurons. Neurosci Lett 18:19–24
Bentivoglio M, Kuypers HGJM, Catsman-Berrevoets CE, Loewe H, Dann O (1980b) Two new fluorescent retrograde neuronal tracers which are transported over long distances. Neurosci Lett 18:25–30
Blanks RH, Precht W, Torigoe Y (1983) Afferent projections to the cerebellar flocculus in the pigmented rat demonstrated by retrograde transport of horseradish peroxidase. Exp Brain Res 52:293–306
Bloom FE (1973) Ultrastructural identification of catecholamine-containing central synaptic terminals. J Histochem Cytochem 21:333–348
Bloom FE, Battenberg ELF (1976) A rapid, simple and more sensitive method for the demonstration of central catecholamine-containing neurons and axons by glyocylic acid-induced fluorescence. II. A detailed description of methodology. J Histochem Cytochem 24:561–571
Bloom FE, Hoffer BJ, Siggins GR (1971) Studies on norepinephrine-containing afferents to Purkinje cells of rat cerebellum. I. Localization of the fibers and their synapses. Brain Res 25:501–521
Bolme P, Fuxe K, Lidbrink P (1972) On the function of central catecholamine neurons-their role in cardiovascular and arousal mechanisms. Res Commun Chem Pathol Pharmacol 4:637–697
Bowden DM, German DC, Poynter WD (1978) An autoradiographic, semistereotaxic mapping of major projections from locus coeruleus and adjacent nuclei in Macaca Mulatta. Brain Res 145:257–276
Chan-Palay V (1977) Cerebellar dentate nucleus: organization, cytology and transmitters. Springer, Berlin Heidelberg New York
Chu N-S, Bloom FE (1974) The catecholamine-containing neurons in the cat dorsolateral pontine tegmentum: distributions of the cell bodies and some axonal projections. Brain Res 66:1–21
Crispino L, Bullock TH (1984) Cerebellum mediates modality-specific modulation of sensory responses of midbrain and forebrain in rat. Proc. Natl Acad Sci USA 81:2917–2920
Dahlström A, Fuxe K (1964) Evidence for the existence of monoamine neurons in the central nervous system. I. Demonstration of monoamines in the cell bodies of brain stem neurons. Acta Physiol Scand [Suppl] 232:1–55
Dietrichs E (1983) Cerebellar cortical afferents from the periaqueductal grey in the cat. Neurosci Lett 41:21–26
Dietrichs E (1984) Cerebellar autonomic function: direct hypothalamo-cerebellar pathway. Science 223:591–593
Dietrichs E, Haines DE (1984) Demonstration of hypothalamo-cerebellar and cerebello-hypothalamic fibres in a prosimian primate (Galago crassicaudatus). Anat Embryol 170:313–318
Dietrichs E, Haines DE (1985a) Observations on the cerebello-hypothalamic projection, with comments on nonsomatic cerebellar circuits. Arch Ital Biol 123:133–139
Dietrichs E, Haines DE (1985b) Do the same hypothalamic neurons project to both amygdala and cerebellum? Brain Res (in press)
Dietrichs E, Zheng ZH (1984) Are hypothalamo-cerebellar fibers collaterals from the hypothalamo-spinal projection? Brain Res 296:225–231
Dietrichs E, Walberg F, Haines DE (1985a) Cerebellar nuclear afferents from feline hypothalamus demonstrated by retrograde transport after implantation of crystalline WGA-HRP. Neurosci Lett 54:129–133
Dietrichs E, Haines DE, Qvist H (1985b) Indirect hypothalamo-cerebellar pathway? Demonstration of hypothalamic efferents to the lateral reticular nucleus. Exp Brain Res (in press)
Fallon JH, Koziell DA, Moore RY (1978) Catecholamine innervation of the basal forebrain. II. Amygdala, suprarhinal cortex and entorhinal cortex. J Comp Neurol 180:509–532
Foote SL, Bloom FE, Aston-Jones G (1983) Nucleus locus cerulues: new evidence of anatomical and physiological specificity. Physiol Rev 63:844–914
Fulton JF (1937) The interrelation of cerebrum and cerebellum in the regulation of somatic and autonomic functions. William & Wilkins, Baltimore, pp 135–182 (The Harvey Lectures, vol 31)
Haines DE, Dietrichs E (1984) An HRP study of hypothalamo-cerebellar and cerebello-hypothalamic connections in squirrel monkey (Saimiri sciureus). J Comp Neurol 229:559–575
Haines DE, Dietrichs E, Sowa TE (1984) Hypothalamo-cerebellar and cerebello-hypothalamic pathways: a review and hypothesis concerning cerebellar circuits which may influence autonomic centers and affective behavior. Brain Behav Evol 24:198–220
Haines DE, Sowa TE, Dietrichs E (1985) Connections between the cerebellum and hypothalamus in the tree shrew (Tupaia glis). Brain Res 328:367–373
Hökfelt T, Fuxe K (1969) Cerebellar monoamine nerve terminals, a new type of afferent fibers to the cortex cerebelli. Exp Brain Res 9:63–72
Johnson FH, Russel GV (1952) The locus coeruleus as a pneumotaxic center. Anat Rec 112:348
Jones BE, Friedman L (1983) Atlas of catecholamine perikarya, varicosites and pathways in the brainstem of the cat. J Comp Neurol 215:382–396
Jones BE, Moore RY (1977) Ascending projections of the locus coeruleus in the rat. II. Autoradiographic study. Brain Res 127:23–53
Kaada BR (1972) Stimulation and regional ablation of the amygdaloid complex with reference to functional representations: In: Eleftheriou BE (ed) The neurobiology of the amygdala. Plenum Press, New York, pp 205–281
Kaelber WW (1978) The relationship of the locus coeruleus to the amygdala in the cat. J Anat 127:469–473
Katz LC, Burkhalter A, Dreyer WJ (1984) Fluorescent latex microspheres as a retrograde neuronal marker for in vivo and in vitro studies of visual cortex. Nature 310:498–500
Keizer K, Kuypers HGJM, Huisman AM, Dann O (1983) Diamidino yellow dihydrochloride (DY-2HCI); a new fluorescent retrograde neuronal tracer, which migrates only very slowly out of the cell. Exp Brain Res 51:179–191
Kimoto Y, Satoh K, Sakumoto T, Tohyama M, Shimizu N (1978) Afferent fiber connections from the lower brain stem to the rat cerebellum by the horseradish peroxidase method combined with MAO staining, with special reference to noradrenergic neurons. J Hirnsforsch 19:85–100
Krettek JE, Price JL (1978) A description of the amygdaloid complex in the rat and cat with observations on intra-amygdaloid axonal connections. J Comp Neurol 178:255–280
Kuru M, Yamamoto H (1964) Fiber connections of the pontine detrussor nucleus. J Comp Neurol 123:161–185
Kuypers HGJM, Catsman-Berrevoets CE, Padt RE (1977) Retrograde axonal transport of fluorescent substances in the rat's forebrain. Neurosci Lett 6:127–135
Landis S, Bloom FE (1975) Ultrastructural identification of noradrenergic boutons in mutant and normal mouse cerebellar cortex. Brain Res 96:299–305
Landis S, Shoemaker WJ, Schlumpf M, Bloom FE (1975) Catecholamines in mutant mouse cerebellum: fluorescence microscopic and chemical studies. Brain Res 93:253–266
Martner J (1975) Cerebellar influences on autonomic mechanisms. An experimental study in the cat with special reference to the fastigial nucleus. Acta Physiol Scand [Suppl] 425:1–42
Mason ST, Fibiger HC (1979) Regional topography within noradrenergic locus coeruleus as revealed by retrograde transport of horseradish peroxidase. J Comp Neurol 187:703–724
Mehler WR (1980) Subcortical afferent connections of the amygdala in the monkey. J Comp Neurol 190:733–762
Mizuno N, Nomura S, Takeuchi Y (1980) The parabrachial nucleus as an intermediate relay station of the visceral afferent pathwyas in the cat. In: Ito M, Tsukahara N, Kubota K, Yagi K (eds) Integrative control functions of the brain, vol 3. Kodansha, Tokyo, pp 51–64
Moruzzi G (1940) Paleocerebellar inhibition of vasomotor and respiratory carotid sinus reflexes. J Neurophysiol 3:20–31
Mugnaini E, Dahl AL (1975) Mode of distribution of aminergic fibers in the cerebellar cortex of the chicken. J Comp Neurol 162:417–432
Nomura SN, Mizuno N, Itoh K, Matsuda K, Sugimoto T, Nakamura Y (1979) Localization of parabrachial nucleus neurons projecting to the thalamus or the amygdala in the cat using horseradish peroxidase. Exp Neurol 64:375–385
Norgren R (1976) Taste pathways to hypothalamus and amygdala. J Comp Neurol 166:17–30
Norgren R, Leonard CM (1971) Taste pathways in rat brainstem. Science 173:1136–1139
Norgren R, Leonard CM (1973) Ascending central gustatory pathways. J Comp Neurol 150:217–237
Norita M, Kawamura K (1980) Subcortical afferents to the monkey amygdala: an HRP study. Brain Res 190:225–230
Olson L, Fuxe K (1971) On the projections from the locus coeruleus noradrenaline neurons: the cerebellar innervation. Brain Res 28:165–171
Ottersen OP (1981) Afferent connections to the amygdaloid complex of the rat with some observations in the cat. III. Afferents from the lower brain stem. J Comp Neurol 202:335–356
Ottersen OP, Ben-Ari Y (1978) Pontine and mesencephalic afferents to the central nucleus of the amygdala of the rat. Neurosci Lett 8:329–334
Persson M, Wiklund L (1981) Scattered catecholaminergic cells in the dorsolateral tegmentum caudal to locus coeruleus in cat. Neurosci Lett 23:275–280
Pickel VM, Krebs H, Bloom FE (1973) Proliferation of norepinephrine-containing axons in rat cerebellar cortex after peduncle lesions. Brain Res 59:169–179
Pickel VM, Segal M, Bloom FE (1974) A radioautographic study of the efferent pathways of the nucleus locus coeruleus. J Comp Neurol 155:15–42
Poitras D, Parent A (1978) Atlas of the distribution of monoamine-containing nerve cell bodies in the brain stem of the cat. J Comp Neurol 179:699–718
Roffler-Tarlov S, Landis SC, Zigmond MJ (1984) Effects of Purkinje cell degeneration on the noradrenergic projection to mouse cerebellar cortex. Brain Res 298:303–311
Russchen FT (1982) Amygdalopetal projections in the cat. II. Subcortical afferent connections. A study with retrograde tracing techniques. J Comp Neurol 207:157–176
Russchen FT, Lohman AHM (1979) Afferent connections of the amygdala in the cat. Folia Anat Iugoslavica [Suppl] 9:57–63
Saper CB, Loewy AD (1980) Efferent connections of the parabrachial nucleus in the rat. Brain Res 197:291–317
Snider RS (1972) Some cerebellar influences on autonomic function. In: Hackman CH (ed) Limbic system mechanisms and autonomic fuction. Thomas, Springfield, Ill, pp 87–90
Somana R, Walberg F (1978) The cerebellar projection from locus coeruleus as studied with retrograde transport of horseradish peroxidase in the cat. Anat Embryol 155:87–94
Somana R, Walberg F (1979a) The cerebellar projection from the parabrachial nucleus in the cat. Brain Res 172:144–149
Somana R, Walberg F (1979b) Cerebellar afferents from the nucleus of the solitary tract. Neurosci Lett 11:41–47
Steindler DA (1981) Locus coeruleus neurons have axons that branch to the forebrain and cerebellum. Brain Res 223:367–373
Taber E (1961) The cytoarchitecture of the brain stem of the cat. I. Brain stem nuclei of cat. J Comp Neurol 116:27–70
Takeuchi Y, McLean JH, Hopkins DA (1982) Reciprocal connections between the amygdala and parabrachial nuclei: ultrastructural demonstration by degeneration and axonal transport of horseradish peroxidase in the cat. Brain Res 239:583–588
Tohyama M (1976) Comparative anatomy of cerebellar catecholamine innervation from teleosts to mammals. J Hirnforsch 17:43–60
Ungerstedt U (1971) Stereotaxic mapping of the monoamine pathways in the rat brain. Acta Physiol Scand [Suppl] 367:1–48
Van der Kooy D, Kuypers HGJM, Catsman-Berrevoets CE (1978) Single mammillary body cells with divergent axon collaterals. Demonstration by a simple, fluorescent retrograde double labeling technique in the rat. Brain Res 158:189–196
Veening JG (1978) Subcortical afferents of the amygdaloid complex in the rat. Neurosci Lett 8:196–202
Voshart K, van der Kooy D (1981) The organization of the efferent projections of the parabrachial nucleus to the forebrain in the rat: a retrograde fluorescent double-labeling study. Brain Res 212:271–286
Wiklund L, Léger L, Persson M (1981) Monoamine cell distribution in the cat brain stem. A fluorescence histochemical study with quantification of indolaminergic and locus coeruleus cell groups. J Comp Neurol 203:613–647
Yao Z, Zhou J (1983) Afferent connections of the amygdaloid complex — studies with retrograde axonal transport method of HRP in the central and medial nuclei of amygdala in the rat. Acta Anat Sinica 14:368–373
Zheng Z-H, Dietrichs E, Walberg F (1982) Cerebellar afferent fibres from the dorsal motor vagal nucleus in the cat. Neurosci Lett 32:113–118
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dietrichs, E. Divergent axon collaterals to cerebellum and amygdala from neurons in the parabrachial nucleus, the nucleus locus coeruleus and some adjacent nuclei. Anat Embryol 172, 375–382 (1985). https://doi.org/10.1007/BF00318986
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00318986