Abstract
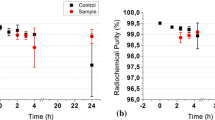
Several chromatographic methods have been used for determining the radiochemical purity of 99mTc-diethyl HIDA (Solco HIDA). Good separation of 99mTc-HIDA from 99mTc-hydrolyzate and nonreduced 99mTcO -4 was obtained in a short time using Gelman ITLC (SA) and 2N NaCl as the mobile phase. In vitro stability was followed by the same method. It was found that the presence of oxygen from air enhances the percentage of 99mTcO -4 in the preparation. Formation of a chelate of 99mTc-HIDA with a high stability constant was established by using Sephadex G-25 filtration. The degree of binding of 99mTc-diethyl HIDA to blood plasma proteins, examined by the same method.
In vivo stability was examined by analyzing bile and urine, which were found to contain 99mTc-HIDA chelate. Biodistribution of the preparation was also investigated in experimental animals.
Similar content being viewed by others
References
Eckelman WC, Meinken G, Richards P (1971) Chemical state of 99mTc in biomedical products. J Nucl Med 12: 596–600
Loberg MD, Cooper M, Harvey E, Callery P, Faith W (1976) Development of new radiopharmaceuticals based on N-substitution of iminodiacetic acid. J Nucl Med 17: 633
Loberg MD, Fields AT (1977) Stability of 99mTc-labeled N-substituted iminodiacetic acids: Ligand exchange reaction between 99mTc-HIDA and EDTA. Int J Appl Radiat Isot 28: 687
Pauwels EKJ, Feitsman RIJ (1977) Radiochemical Quality Control of 99mTc-Labeled Radiopharmaceuticals. Eur J Nucl Med 2: 97–103
Sundrehagen E (1979) Polymer Formation and Hydrolysation of 99Tc (IV). Int J App Rad Isotopes 30: 739–743
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jovanović, V., Konstantinovska, D. & Memedović, T. Determination of radiochemical purity and stability of 99mTc-diethyl HIDA. Eur J Nucl Med 6, 375–378 (1981). https://doi.org/10.1007/BF00251341
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00251341