Targeted radionuclide therapy (TRT), also known as radionuclide therapy or molecular targeted radiotherapy, has emerged as a model of precision medicine. Its efficacy relies on targeting a hallmark expressed by cancer cells rather than the type of cancer, making it a prime example of a tissue-of-origin–agnostic approach. For example, within the pool of tumors expressing somatostatin receptors (SSTs) and thus eligible for TRT, commonly known as peptide receptor radionuclide therapy in this setting, there exists a broad spectrum of malignancies. These range from pediatric tumors, such as neuroblastoma and medulloblastoma, through pulmonary and gastroenteropancreatic neuroendocrine neoplasia (NEN) to the spectrum of pheochromocytoma/paraganglioma syndromes, among others. Beyond the target expression, which can be variable itself, within these diseases is a substantial variation in biologic characteristics including diverse cellular origins, markedly differing growth rates, heterogeneous but overlapping mutational profiles, and patterns of tumor spread. In defining tumoral heterogeneity, we have to consider also the impact of tissue microenvironment, which can vary between tumor types. Consequently, the challenges of selecting which patients to treat, predicting their response to treatment, and prescribing the optimum treatment protocol are manifold. These considerations also include differing sensitivities to radiation influenced by both the tissue of origin and the tumor’s genetic makeup, whether through driver mutations or secondary epigenomic alterations. In a simplistic theranostic approach, SST expression on a whole-body scale as assessed by SST PET and histopathologic features acts as a predictive biomarker for identifying suitable candidates for TRT. Although TRT is currently administered as a fixed activity every 6–8 wk over 2–6 cycles and has been shown to be efficacious across a broad spectrum of tumor types with relatively low toxicity, this approach does not take into account specific tumor or patient characteristics. For example, patients with a large disease burden, different lesion sizes or sites, tumors with low or heterogeneous SST expression, or likely increased radioresistance mechanisms related to altered tissue microenvironment in large deposits may benefit from higher administered activities, different intervals between cycles, or different radionuclides. Although there are some logistic advantages to treating the patients with a standardized approach, it is somewhat antithetic to the concept of precision medicine. We believe that it would therefore be important to shift toward more personalized theranostic strategies based on tumor biology. In this perspective, we combine observations from our personal experiences of delivering theranostics; our synthesis of the available published data, particularly from recent clinical trials; and general principles of radiation biology to reimagine how it might be possible to change both the delivery of theranostics and the endpoints used to assess efficacy. Importantly, we recognize that some of our opinions currently lack validation since existing trials have largely failed to address the importance of either tumor biology or disease burden and have largely adopted a protocolized rather than personalized prescription of the administered activity, the number of cycles, and the timing between them. Nevertheless, we hope that our observations will stimulate researchers and industry to reconsider the design of future trials of TRT to address the holes in the existing evidence base.
CONSIDERATION OF QUALITY OF LIFE
Although overall survival (OS) and progression-free survival (PFS) are traditional measures of the efficacy of cancer therapies, in more indolent tumor types and in certain selected populations, including the elderly, quality of life is an increasingly important factor in treatment selection. In addition to premature deaths, NEN is particularly characterized by hormone-related symptoms that can markedly reduce quality of life. Evaluation of treatment outcomes in patients with functional tumors provides the opportunity to assess the rapidity of action of TRT and beneficially impact quality of life. Several studies have demonstrated the positive antihormonal effect of TRT (1). In a recent study that included patients with metastatic insulinoma, hormonal response was observed after the first cycle in 65% of cases (2). As a feature of differentiation, hormone secretion can be associated with indolent cancer biology and lead to a large area under the curve for suffering due to long survival. In such patients, limiting the number of treatments to that required for hormonal control may delay the onset of treatment resistance or off-target toxicities while not manifestly negatively impacting survival. We contend that for indolent NEN with hormone-related symptoms, patient-reported outcomes, preferably using appropriately validated quality-of-life instruments that cover appropriate symptoms relevant to NEN, should be the primary endpoint of such clinical trials and that eligibility criteria could be expanded to include patients with poorer performance status and without the requirement for disease progression.
INTEGRATION OF COMBINATION THERAPIES INTO TREATMENT PARADIGMS
There is potentially a wide range of oncologic therapies that could be combined with TRT in terms of both drug types and sequencing. These rely mostly on preclinical data or experience extrapolated from combination therapies with external-beam radiotherapy, for which synergistic or additive therapeutic effects have been demonstrated. However, these may increase concerns regarding cumulative toxicities (especially bone marrow damage) (3) and an increased cost burden. In our opinion, thrilling approaches include combining TRT with immune checkpoint therapy or inhibiting DNA damage response and repair (4), such as PARP inhibitors that convert the predominantly single-strand DNA breaks associated with β-particle TRT into double-strand breaks.
Although low responses to immune checkpoint inhibitors have been observed with most neuroendocrine neoplasms because of relatively low mutational burdens, combination with immune checkpoint therapy or approaches that improve neoantigen presentation may be an alternative mechanism for immune priming and might not need as high a radiation exposure as required to have direct cytocidal effects. This might have advantages for Auger electron–emitting radionuclides (5).
The challenge of introducing inhibitors of DNA damage response and repair into the clinic will be managing the likely enhancement of toxicity to normal tissues. However, the reliance on different repair pathways in tumors and normal tissues may provide protection. For example, slowly growing tumors are reliant on nonhomologous end-joining for repair of double-strand DNA breaks, whereas proliferating bone marrow will generally use homologous recombination. Accordingly, DNA-protein kinase inhibitors that inhibit nonhomologous end-joining may provide differential radiosensitization (6).
The use of predictive modeling and machine learning may help to design clinical studies optimally and predict outcomes at an intermediate stage with good reliability. Another approach would be to invest the neoadjuvant space that can serve as a platform for research and TRT development. This would enrich translational research programs and help to evaluate the impact of TRT on genomic, immune microenvironment signature, and other important parameters.
Balancing the incremental risks versus benefits of combination therapies is facilitated by the likelihood of premature death from failure of disease control. There are 2 major considerations in this regard: the ability to deliver adequate radiation to achieve disease control and the biology of the tumor, particularly with respect to radiosensitivity and the likelihood of premature death based on proliferative activity.
EFFECT OF ABSORBED DOSE ON RESPONSE
The goal of any form of radiation therapy is conventionally held to be to deliver the highest radiation possible to tumor while minimizing dose to normal tissues. Several studies have demonstrated that tumor radiation dose decreases during treatment, with the maximum lesion dose typically being observed at cycle 1 (7). In responding tumors, off-target radiation to normal tissues is also least with cycle 1 because of sink effects. Importantly, below a specific radiation dose threshold, achieving a response becomes unlikely and is further modified by genetic susceptibilities. In the setting of intrinsic radioresistance, escalating the administered activity is unlikely to enhance response but increases the risk of side effects (8). Accordingly, prediction of radiation dose delivery, or at least verification of radiation dose delivery by posttreatment imaging, becomes important to understanding the likelihood of response and potentially modifying treatment planning—ideally before, or at least during, the course of treatment (9). In recent years, the implementation of a new generation of extremely sensitive digital PET/CT scanners, potentially allowing late-time-point imaging for prospective dosimetry, and multidetector CZT-based SPECT/CT systems and artificial intelligence–based dosimetry software has increased the practicality of routine posttreatment dosimetry in clinical practice. When the radiation dose is estimated to be too low for a favorable outcome, treatment could be suspended or strategies adopted to augment therapeutic efficacy, including use of radiosensitizing strategies such as DNA damage response-modifying agents as discussed above.
EFFECT OF CANCER BIOLOGY ON DISEASE CONTROL
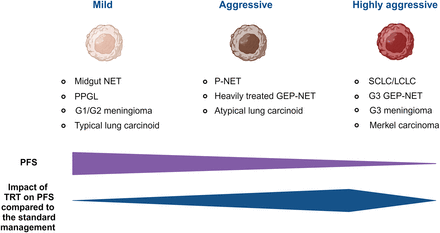
In understanding response to TRT, it is important to recognize the highly variable natural history of various SST-expressing tumors even in the absence of effective treatment. PFS, for example, differs markedly between grade 1 and 3 NEN. Because actively dividing cells are generally more radiosensitive than indolent tumors, the objective response rate (ORR) tends to be higher in more aggressive tumors despite the tendency for PFS to be shorter because of more rapid repopulation of cells not eradicated by treatment. Several studies have demonstrated a relatively high ORR in grade 3 NEN (10). As a result, the appropriate measure of efficacy chosen may also vary by tumor type and grade. Data from the SEPTRALU registry show the efficacy and safety of [177Lu]Lu-DOTATATE in a wide range of SST-expressing NENs (n = 522), regardless of location (8). The best RECIST 1.1 responses were complete response, 0.7%; partial response, 33.2%; stable disease, 52.1%; and tumor progression, 14%, with efficacy varying with tumor subtype, despite benefit in all subgroups. Median PFS was 31.3 mo in midgut NENs, 30.6 mo in PPGL NENs, 19.8 mo in pancreatic NENs, and 17.6 mo in bronchopulmonary NENs, again emphasizing the importance of tissue of origin. Indeed, dichotomous results between PFS and ORR are reported in different phase II and III clinical trials. The observed PFS after TRT relies heavily on the specific cancer subtype, with midgut being higher than pancreatic neuroendocrine tumors (NETs), whereas the ORR is generally higher for pancreatic NETs than for small intestinal NETs or for grade 2/3 than for grade 1 NETs (11). The incremental impact of TRT on OS, compared with standard treatment, may thus be greater for grade 2/3 than grade 1 NEN (Fig. 1) even though PFS is substantially shorter in absolute terms.
Distinction between disease-related PFS and expected impact of TRT on PFS compared with standard management in various SST-expressing tumors. G = grade; GEP = gastrointestinal and pancreatic; LCLC = large cell lung cancer; NET = neuroendocrine tumor; P-NET = pancreatic NET; PPGL = pheochromocytoma/paraganglioma; SCLC = small cell lung cancer.
Despite the lack of robust data from clinical trials, we can identify 3 main groups of cancer biology that may aid in guiding treatment with TRT, predicting likely effects and refining objectives more effectively.
Indolent Cancer Biology
The most robust data on the efficacy of TRT in indolent NEN derive from the NETTER-1 open-label, randomized, controlled phase 3 trial in progressive midgut NETs after first-line somatostatin analog therapy (12). NETTER-1 compared [177Lu]Lu-DOTATATE (4 administrations of 7.4 GBq) with an augmented dose of octreotide, long-acting release (60 mg), in patients with progressive, SST-expressing small-intestinal NEN (ileal in 75% of cases). Most patients on this trial had grade 1 tumors. Although the interim analysis was encouraging for [177Lu]Lu-DOTATATE in terms of PFS, differences in median OS did not reach statistical significance (48 vs. 36.3 mo, P = 0.30) and the ORR was relatively low, albeit higher than in the control arm (18% vs. 3%, P < 0.001) (13). This may seem somewhat counterintuitive since these tumors exhibit high SST expression allowing delivery of multiple therapeutic cycles and thereby achieving high cumulative tumor radiation doses. This exemplar of an indolent cancer biology is probably relevant to pheochromocytoma/paraganglioma, grade 1 and 2 meningioma, and typical lung carcinoids. Accordingly, it would be anticipated that the effect of TRT on OS in these tumors, compared with the standard management, might be limited mostly to disease stabilization. In this setting, the impact of treatment on quality of life becomes even more important, and potential delayed toxicities related to treatments are a major consideration. Ongoing academic trials aim to evaluate, for example, whether a longer interval between cycles (>8 wk) for these types of tumors could be equally effective and less toxic as part of a maintenance treatment strategy. Additionally, control of hormone-related symptoms, when present, provides an objective benefit of TRT and may be a more important and appropriate surrogate of response than PFS or OS. It can also often be objectively validated by reduced hormone levels.
Aggressive Cancer Biology
In the NETTER 2 trial, which included more aggressive disease (grades 2 and 3 gastrointestinal and pancreatic NETs) randomized to [177Lu]Lu-DOTATATE or an augmented dose of octreotide, long-acting release (60 mg), the reported PFS was 22.8 mo in TRT arm versus 8.5 mo in the control arm, with an ORR of 43% versus 9.3%, respectively (14). In the OCLURANDOM study (noncomparative phase II randomized study), which involved patients with heavily treated pancreatic NETs (56% and 58% of patients received chemotherapy in the [177Lu]Lu-DOTATATE and sunitinib arms, respectively), preliminary results have shown good efficacy for peptide receptor radionuclide therapy with [177Lu]Lu-DOTATATE, with an 80% 12-mo PFS rate (sunitinib, 42% 12-mo PFS rate) and a median PFS in the peptide receptor radionuclide therapy arm of 20.7 mo (presented as oral communications at the 2022 meetings of the European Society for Medical Oncology and the European Association of Nuclear Medicine). The study sample closely mirrors the results of the original sunitinib study participants and suggests superior disease control (PFS of 11.4 mo in the earlier study). Collectively, these studies prove a great impetus toward the use of TRT in aggressive cancer models, encompassing patients with pancreatic NETs, heavily treated gastrointestinal and pancreatic NETs, and atypical lung carcinoids. Nevertheless, it is likely that study endpoints around PFS and OS will become more relevant in these diseases than for tumors with more indolent biology.
Highly Aggressive Cancer Biology
The subgroup of highly aggressive cancer biology includes small cell lung cancer/large cell lung cancer, grade 3 gastrointestinal and pancreatic NETs, grade 3 meningioma, and Merkel carcinoma. Because of their aggressive nature, the potential therapeutic effect can be rapidly counterbalanced by proliferation, making it difficult to administer a complete therapeutic schedule using the current standard protocol. Furthermore, initiating the first cycle may pose a challenge due to both the eligibility period and the delay in starting the treatment. This is even more true in the setting of trials. Hence, prompt initiation of TRT and shorter cycle intervals could be crucial for managing the rapid repopulation of these tumors. The incorporation of chemotherapy, which offers direct cytotoxic effects on tumors and enhances radiosensitivity, is gaining significance in addressing these types of tumors. Since these aggressive tumors also tend to have a higher mutational burden (a predictive biomarker of response to immune checkpoint inhibitor efficacy), combination with these agents may further enhance the efficacy of TRT. An SST antagonist, [177Lu]Lu-satoreotide tetraxetan, which has been evaluated in a phase I/II trial (15), is being trialed in combination with immune checkpoint therapy in small cell lung cancer. Importantly, in treating heterogeneous tumors at a microscopic scale and when prior or concurrent treatments may enhance off-target toxicity; in using TRT when disease volume is highest, promoting crossfire irradiation of even cells lacking SST expression near those that do; and in minimizing compounding toxicity by leveraging the sink effect, combination of TRT with chemotherapy or in other combinations applied upfront rather than as a salvage therapy has logical appeal.
USE OF ALTERNATIVE RADIONUCLIDES
Although 177Lu has become the most widely used therapeutic radionuclide, there are many other options that could be considered for use with SST agents. Although 90Y has fallen out of use because of off-target toxicity to the kidneys, it has theoretic advantages for large tumor masses, and the off-target effects can be mitigated by leveraging the tumor sink effect. Beginning a course of treatment with this radionuclide and transitioning to 177Lu has been shown to be highly effective in controlling disease and to be well tolerated (16). Although potentially more toxic to normal tissues, the use of α-particles also offers a unique advantage in inducing more lethal double-strand breaks than do β-particles and in targeting microscopic dormant clusters of cells. 225Ac, 211At, and 212Pb emerge as the foremost candidates among α-emitters for this purpose (17,18). In this regard, targeted α-therapy should take the lead either alone or in therapeutic combination. Clinical trials are currently in progress, yet numerous hurdles persist regarding α-emitters. These include establishing reliable supply chains, enhancing understanding of the correlation between administered activity and absorbed dose in both tissue and tumor, and addressing the long-term adverse effects and their links with the absorbed dose. The latter point is crucial, especially considering that many patients with NETs have a long life expectancy despite significant tumor burden. Although results from a 5-y long-term follow-up study of [225Ac]Ac-DOTATOC are reassuring (19), we urgently need data from prospective trials to further validate these results.
An exciting option with respect to the combination of a low– and high–linear-energy-transfer agent is 161Tb, which has a β-energy and half-life similar to those of 177Lu but additional Auger electron emissions that will also augment radiation dose to micrometastases.
CONCLUSIONS AND PERSPECTIVES
It is evident that the efficacy and benefit of TRT in NETs vary significantly within disease groups and between patients and that many factors beyond simply assessing target expression must be considered. It is likely that advances in the molecular characterization of these tumors will refine the proposed subclasses. The concept of precision medicine is underpinned by the ability to perform these analyses accurately and to integrate them into care practice. To elevate the standard of care for patients harboring these tumors, it is imperative to foster collaboration among health care organizations, industry leaders, academic institutions, and relevant professional/patient-related societies. This effort should manifest in global initiatives, including the creation of registries and databases and the organization of symposia and congresses, all geared toward catalyzing clinical trials, research programs, and the dissemination of vital information in the rapidly evolving field of theranostics.
DISCLOSURE
No potential conflict of interest relevant to this article was reported.
Footnotes
Published online Sep. 19, 2024.
- © 2024 by the Society of Nuclear Medicine and Molecular Imaging.
REFERENCES
- Received for publication May 26, 2024.
- Accepted for publication August 2, 2024.