Visual Abstract
Abstract
Although immunotherapy has revolutionized the entire cancer treatment landscape, small fractions of patients respond to immunotherapy. Early identification of responders may improve patient management during immunotherapy. In this study, we evaluated a PET approach for monitoring immunotherapy in lung cancer by imaging the upregulation of lymphocyte activation gene 3 (LAG-3)–expressing (LAG-3+) tumor-infiltrating lymphocytes (TILs). Methods: We synthesized a LAG-3–targeted molecular imaging probe, [68Ga]Ga-NOTA-C25 and performed a series of in vitro and in vivo assays to test its specificity. Next, [68Ga]Ga-NOTA-C25 PET was used to monitor immunotherapy in murine lung cancer–bearing mice and in humanized mouse models for assessing clinical translational potential, with confirmation by immunostaining and flow cytometry analysis. Results: [68Ga]Ga-NOTA-C25 PET could noninvasively detect intertumoral differences in LAG-3+ TIL levels in different tumor models. Importantly, in Lewis lung carcinoma tumor models treated with an agonist of a stimulator of interferon genes, [68Ga]Ga-NOTA-C25 PET also detected an immunophenotyping transition of the tumor from “cold” to “hot” before changes in tumor size. Meanwhile, animals carrying “hot” tumor showed more significant tumor inhibition and longer survival than those carrying “cold” tumor. [68Ga]Ga-NOTA-C25 PET also showed markedly higher tumor uptake in immune system–humanized mice carrying human non–small cell lung cancer than immunodeficient models. Conclusion: [68Ga]Ga-NOTA-C25 PET could be used to noninvasively monitor the early response to immunotherapy by imaging LAG-3+ TILs in lung cancer. [68Ga]Ga-NOTA-C25 PET also exhibited excellent translational potential, with great significance for the precise management of lung cancer patients receiving immunotherapy.
Lung cancer is the leading cause of cancer death globally, and approximately 80% of lung cancers are non–small cell lung cancer (NSCLC) (1). Although immunotherapy has been approved for NSCLC, with certain survival benefits (2), numerous patients have been unresponsive (3). Cancer immunotherapy depends on rescuing CD8+ T cells from inhibitory status to activation, for the elimination of tumor cells (4). During this process, immunophenotypes of the tumor can be categorized by the abundance of tumor-infiltrating lymphocytes (TILs) in the tumor immune microenvironment. Specifically, immune “hot” tumors have abundant TILs, whereas “cold” tumors lack TILs (5). It has been well established that the treatment response to immunotherapy is strongly associated with tumor TIL abundance, particularly CD8+ T cells, which play a central role in improving the response to cancer immunotherapy (6).
Lymphocyte activation gene 3 (LAG-3) is a type I transmembrane protein mainly expressed in T cell–based TILs. It has become an important next-generation immune checkpoint after programmed cell death protein 1 (PD-1)/programmed death ligand 1 and cytotoxic T lymphocyte-associated antigen 4 (7). Naïve and resting T cells do not express LAG-3, but activated T cells could upregulate LAG-3 expression (8,9). Thus, LAG-3 expression upregulation correlates with the early phase of T cell activation, as observed in lung cancer patients (7,9,10). The Food and Drug Administration approved the first LAG-3 immune checkpoint inhibitor (ICI; relatlimab) in combination with nivolumab for cancer immunotherapy in 2022 (11). Clinical studies showed that relatlimab combined with nivolumab significantly improved progression-free survival compared with nivolumab monotherapy in NSCLC (8,12). Moreover, a high level of LAG-3–expressing (LAG-3+) TILs was shown to be an independent positive prognostic factor in stage I–IIIB lung cancer patients (9). Hence, LAG-3+ TILs may be a useful biomarker for identifying the immunotherapy response at early times.
PET/CT has been extensively applied in clinical molecular imaging for tumor theranostics (13). PD-1/programmed death ligand 1–targeted PET has been used to predict the response to ICIs and has been shown to be superior to immunohistochemistry (14). Recent imaging studies focused on early-stage immune biomarkers (OX-40 and inducible costimulator) (15,16) and downstream biomarkers (interleukin 2 and granzyme B) of the immune reaction (17,18). These studies demonstrated potential in assessing the tumor immunotherapy response, particularly conspicuous for granzyme B PET (19). However, a weak correlation was also reported in a clinical trial (20). Effort needs to be put toward developing novel molecular imaging biomarkers for evaluating immunotherapy.
C25, a 9-amino-acid cyclic peptide (CVPMTYRAC), was identified recently as having a high affinity for both human and murine LAG-3 (21). As is well known, peptide-based molecular imaging has been widely studied due to multiple advantages, including favorable pharmacokinetics and easy modification and radiolabeling (22). In this study, we report a LAG-3–targeted molecular imaging probe, [68Ga]Ga-NOTA-C25, and assessed the feasibility of [68Ga]Ga-NOTA-C25 PET in detecting LAG-3+ TILs and monitoring the treatment response to monotherapy in lung cancer tumor models.
MATERIALS AND METHODS
Synthesis and Characterization of [68Ga]Ga-NOTA-C25
The precursor NOTA-C25 was rationally designed and synthesized. NOTA-C25 (50.37 μg; 0.038 μmol) was added to [68Ga]GaCl3 (296 MBq in 0.6 mL of 0.05 M HCl), and the pH was adjusted to 4.6 for radiolabeling. The reaction mixture was purified with a C18 cartridge by radio–thin-layer chromatography to obtain [68Ga]Ga-NOTA-C25 for quality control and stability. The specific activity of [68Ga]Ga-NOTA-C25 was calculated on the basis of radiolabeling records (n ≥ 3). Under the same protocol, [68Ga]Ga-NOTA-scrambled C25 was also prepared for subsequent in vitro and in vivo assays. More details are provided in the supplemental materials (supplemental materials are available at http://jnm.snmjournals.org).
Immune Stimulation for Flow Cytometry and Cell Uptake
Immune cells from C57BL/6 mice and human peripheral blood mononuclear cells (hPBMCs; catalog no. HPA01091; Hope Bio-technology) were used. Immune stimulation with an activation cocktail (catalog no. 423301; BioLegend) on C57BL/6 mouse–derived immune cells and phytohemagglutinin-M (catalog no. 11082132001; Sigma) on hPBMCs was performed for flow cytometry analysis of LAG-3 and CD8. Cell uptake of [68Ga]Ga-NOTA-C25 or [68Ga]Ga-NOTA-scrambled C25 (0.037 MBq/mL; 6.788 × 10−9 μM) was performed by blocking assays with nonradiolabeled C25 (9.613 × 10−6 μM) and anti–LAG-3 antibody (αLAG-3; 1.333 × 10−7 μM) to test probe specificity. Detailed protocols are provided in the supplemental materials.
Cells and Animal Models
Cells (murine lung cancer: CMT-167, KLN-205, and Lewis lung carcinoma (LLC); human lung cancer: H460) were cultured at 37°C with 5% CO2 to prepare animal models by subcutaneous transplantation in the right shoulder at different densities. Tumor volume was measured every other day using a caliper, and tumor volume was calculated as (length × width2)/2. All animal protocols were approved by the Animal Care Committee at the Fourth Hospital of Harbin Medical University. More details are provided in the supplemental materials.
PET/CT Imaging and Biodistribution Study
When the tumor volume reached about 300 mm3, CMT-167, KLN-205, and LLC tumor–bearing mice were injected with approximately 7.4 MBq of [68Ga]Ga-NOTA-C25 or [68Ga]Ga-NOTA-scrambled C25 (∼1.8 μg in 100 μL; 0.0014 μmol; with a specific activity of 5.45 ± 0.25 [mean ± SD] GBq/μmol) via the tail vein. PET/CT scanning and biodistribution were performed at different time points after injection of the probe. In blocking assays, C25 peptide (100 μg in 100 μL; 0.0961 μmol) or 200 μg (in 100 μL; 0.0014 μmol) of antimouse LAG-3 antibody (catalog no. BE0174; Bioxcell) was coinjected with [68Ga]Ga-NOTA-C25 into CMT-167– and KLN-205–bearing mice for blocked PET and biodistribution (23). Furthermore, PET/CT imaging and biodistribution based on [68Ga]Ga-NOTA-scrambled C25 were also performed in KLN-205 models with the same protocol as that mentioned earlier. Detailed information is provided in the supplemental materials.
In Vivo PET for Monitoring Immunotherapy
When the tumor volume reached 50–100 mm3 at 10 d after transplantation, LLC models were randomly divided into 4 groups (n ≥ 6) for receiving immunotherapy with anti–PD-1 antibody (αPD-1; intraperitoneal administration of 10 mg/kg of body weight), an agonist of a stimulator of interferon genes (STING) (benzothiophene oxobutanoic acid (MSA-2); subcutaneous administration of 25 mg/kg of body weight), combination therapy (αPD-1 + MSA-2), and phosphate-buffered saline (PBS) as a control. Tumor volume and survival time were recorded. [68Ga]Ga-NOTA-C25 PET/CT was performed at days 4 and 13 during treatment, and animals were euthanized to collect tumors, tumor-draining lymph nodes (TDLN), spleen, and thymus for biodistribution or flow cytometry detection of CD8+ LAG-3+ T cells. Detailed protocols are provided in the supplemental materials.
LAG-3 PET in Immune System–Humanized NSCLC Models
Immune system–humanized NSCLC models were established in H460 tumor models by injecting hPBMCs and not injecting hPBMCs as an immunodeficient control, with flow cytometry verification of the proportion of hCD3+ hCD45+ lymphocytes as previously published (24,25). [68Ga]Ga-NOTA-C25 PET/CT was performed as described earlier. More details are provided in the supplemental materials.
Immunofluorescence Staining
Tumor sections from different murine tumor models, treated animals, or humanized animal models were fixed for immunofluorescence staining of CD3 and LAG-3. Detailed protocols are provided in the supplemental materials.
In Vivo Biologic Activity Experiments
LLC subcutaneous xenograft mouse models were established. When the tumor volume reached 200–300 mm3, LLC tumor models were divided into 3 groups for injection with [68Ga]Ga-NOTA-C25 (7.4 MBq; 1.8 μg in 100 μL; 0.0014 μmol), a blocking dose of C25 (100 μg in 100 μL; 0.0961 μmol), and PBS as a control. Samples of peripheral blood, spleen, and tumor were harvested from these 3 groups at 1 and 48 h after injection for flow cytometry tests of CD8+ γ-interferon–positive percentages. Detailed information is provided in the supplemental materials.
Statistical Analysis
All data analysis was performed on GraphPad Prism 9.0 (GraphPad Software). A 1- or 2-way ANOVA and an unpaired 2-tailed Student t test were used for statistical analysis when appropriate. Results are presented as mean ± SD. A P value of less than 0.05 was considered as statistically significant.
RESULTS
Synthesis and In Vitro Characterization of [68Ga]Ga-NOTA-C25
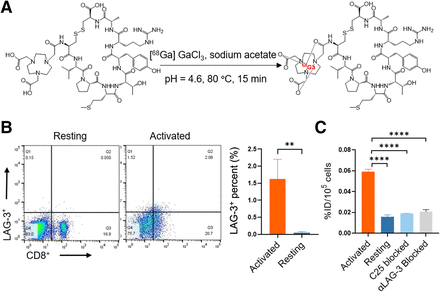
NOTA-C25 was obtained with a purity of 98.54% and confirmed through high-resolution mass spectrometry to have a molecular weight of 1325.5541 (Suppl. Fig. 1). [68Ga]Ga-NOTA-C25 was efficiently radiolabeled, with a radiochemical yield of greater than 85% and a radiochemical purity of greater than 99% (Fig. 1A; Suppl. Fig. 2A). The molar activity of [68Ga]Ga-NOTA-C25 was calculated as 5.45 ± 0.25 GBq/μmol. [68Ga]Ga-NOTA-C25 remained stable in both serum and PBS for at least 240 min of incubation (Suppl. Fig. 2). The oil–water partition coefficient was determined to be −3.550 ± 0.311, indicating the probe’s hydrophilicity. In addition, [68Ga]Ga-NOTA-scrambled C25 was prepared using the same protocol as that mentioned earlier and served as a scrambled probe in subsequent experiments.
(A) Radiochemical synthesis of [68Ga]Ga-NOTA-C25. (B) Flow cytometry and quantitative analysis of CD8+ LAG-3+ in murine immune cells that were activated or resting. (C) Cell uptake of [68Ga]Ga-NOTA-C25 (6.788 × 10−9 μM) in murine immune cells with blocking assays by nonradiolabeled C25 (9.613 × 10−6 μM) and αLAG-3 (1.333 × 10−7 μM). n = 3. **P < 0.01. ****P < 0.0001.
Immune stimulation and cell uptake experiments were performed on murine CD8+ T cells. Results showed that CD8+ T cells exhibited significantly higher LAG-3 expression after stimulation than resting groups (P < 0.01) (Fig. 1B), as detected by flow cytometry. Consistently, stimulation-activated CD8+ T cells exhibited higher [68Ga]Ga-NOTA-C25 uptake than resting groups, and the uptake in activated groups could be specifically blocked by coincubation with nonradiolabeled C25 peptide and αLAG-3 (P < 0.0001) (Fig. 1C). However, there was no significant difference in cell uptake of [68Ga]Ga-NOTA-scrambled C25 among activated, resting, and blocked groups of immune cells, corroborating the specificity of [68Ga]Ga-NOTA-C25 for LAG-3 (Suppl. Fig. 3).
Imaging LAG-3+ TILs In Vivo by [68Ga]Ga-NOTA-C25 PET
[68Ga]Ga-NOTA-C25 PET was used to detect LAG-3+ TILs in vivo in CMT-167, KLN-205, and LLC tumor models. Representative PET/CT images and quantitative analysis are showed in Figure 2. [68Ga]Ga-NOTA-C25 could reach whole-body distribution as early as 30 min after injection and showed “fast-in” and “fast out” trends, with gradually reduced uptake over time in all tumors. Tumor uptake of [68Ga]Ga-NOTA-C25 was highest in KLN-205 tumors, moderate in CMT-167 tumors, and at baseline levels in LLC tumors, with similar trends in tumor/muscle ratios. Blocking PET with nonradiolabeled C25 showed that the tumor uptake and tumor/muscle ratio could both be significantly decreased in CMT-167 and KLN-205 tumor models; however, a greater impact of blocking was observed in the latter. Biodistribution (Suppl. Fig. 4; Suppl. Fig. 5; Suppl. Table 1) showed consistent trends and confirmed the PET results. Differences in [68Ga]Ga-NOTA-C25 distribution among CMT-167, KLN-205, and LLC tumors demonstrated the potential of LAG-3 PET in detecting tumor immune microenvironment heterogeneity.
Representative PET/CT images and quantitative analysis in KLN-205, CMT-167, and LLC tumor–bearing mice at 30, 60, and 120 min after injection of [68Ga]Ga-NOTA-C25 with blocking assays by nonradiolabeled C25 (100 μg in 100 μL; 0.0961 μmol). Yellow arrows indicate tumor. n ≥ 3. *P < 0.05. **P < 0.01. ***P < 0.001. ****P < 0.0001.
αLAG-3–blocked PET at 60 min after injection (Suppl. Figs. 4B and 4C) showed reductions in the tumor uptake and tumor/muscle ratio similar to those seen with C25-blocked PET in both CMT-167 and KLN-205 tumor models. Biodistribution results were consistent with PET results (Suppl. Fig. 5; Suppl. Table 2). However, KLN-205 tumors merely showed a baseline uptake of [68Ga]Ga-NOTA-scrambled C25 at all time points, and the biodistribution of [68Ga]Ga-NOTA-scrambled C25 in KLN-205 tumors at 60 min after injection also confirmed the PET results (Suppl. Fig. 6). These results indicated the specificity of [68Ga]Ga-NOTA-C25 in vivo. Moreover, there was a good correlation of [68Ga]Ga-NOTA-C25 uptake between biodistribution and PET results for KLN-205, CMT-167, and LLC tumors (Suppl. Fig. 7).
Immunostaining results (Suppl. Fig. 8) showed that CD3+ LAG-3+ fluorescence intensity was more abundant in CMT-167 and KLN-205 tumors than in LLC tumors. In summary, these findings demonstrated the feasibility of [68Ga]Ga-NOTA-C25 PET for detecting LAG-3+ TILs in vivo, and LLC tumors could be categorized as immune “cold” by [68Ga]Ga-NOTA-C25 PET.
In Vivo Detection of LAG-3+ TIL Upregulation Induced by STING Agonists in Lung Cancer
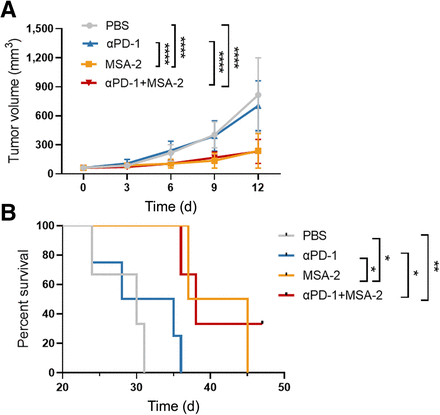
The ability of [68Ga]Ga-NOTA-C25 PET to monitor LAG-3+ TIL dynamics was assessed in LLC tumors, which were determined to be immune “cold” as described earlier. [68Ga]Ga-NOTA-C25 PET was performed at days 4 and 13 to monitor LAG-3+ TIL dynamics (Fig. 3A). At day 4, tumor uptake of [68Ga]Ga-NOTA-C25 in the MSA-2 and MSA-2 + αPD-1 groups was significantly increased compared with that in the αPD-1 or PBS group (Fig. 3B). A similar trend was observed at day 13. However, MSA-2 could induce incremental immune stimulation, as revealed by higher tumor uptake in the MSA-2 group (1.30 ± 0.22 percentage injected dose [%ID]/g) and the MSA-2 + αPD-1 group (1.85 ± 0.41 %ID/g) at day 13 than at day 4 (P < 0.05). Interestingly, the spleen could be clearly visualized by PET at day 13 in the MSA-2 + αPD-1 and MSA-2 groups, suggesting that the STING agonist or a combination with αPD-1 may increase peripheral immune activation (Fig. 3B).
(A) Time line of immunotherapy and PET imaging in LLC tumor–bearing mice. (B) Representative [68Ga]Ga-NOTA-C25 PET/CT and quantitative analysis in LLC models treated with PBS, αPD-1, MSA-2, and αPD-1 + MSA-2 at days 4 and 13. Yellow arrows indicate tumor. Red arrows indicate spleen. (C) Biodistribution in tumor and main immune organs excised from LLC models at days 4 and 13. n ≥ 6. *P < 0.05. **P < 0.01. ***P < 0.001. ****P < 0.0001.
These results were corroborated by the ex vivo biodistribution (Fig. 3C). Specifically, higher probe uptake was observed in tumors, TNLD, and spleen in the MSA-2 + αPD-1 and MSA-2 groups than in the αPD-1 or PBS group at day 4 and even more obviously at day 13. These results further indicated that MSA-2–based treatment caused incremental immune stimulation over time. Flow cytometry analysis at day 13 also showed that the STING agonist induced significant immune responses in the MSA-2 + αPD-1 and MSA-2 groups, as revealed by a higher abundance of CD8+ LAG-3+ T cells in tumors, TDLN, and spleen in these groups than in the αPD-1 or PBS group (Fig. 4). Moreover, there was a good correlation between [68Ga]Ga-NOTA-C25 PET and CD8+ LAG-3+ T cell levels in tumors at day 13 for the combination therapy and PBS groups (Suppl. Fig. 9). Immunostaining of LAG-3 and CD3 on tumor sections at day 13 showed trends similar to those seen with PET, thereby confirming that MSA-2 + αPD-1 and MSA-2 could induce higher infiltration of LAG-3+ TILs than αPD-1 or PBS (Fig. 5).
Flow cytometry detection of CD8+LAG-3+ T cells and quantitative analysis in tumor, spleen, and TDLN excised from LLC tumor models at day 13. n ≥ 6. *P < 0.05. **P < 0.01. ***P < 0.001. ****P < 0.0001.
Immunofluorescence images and quantitative analysis of LAG-3 and CD3 staining in tissue sections from LLC models at day 13. Blue = DAPI. Green = CD3. Red = LAG-3. Scale bar, 50 μm. n = 3. *P < 0.05. **P < 0.01. ***P < 0.001. ****P < 0.0001.
LAG-3 PET Imaging for Monitoring Immunotherapy
Having demonstrated that [68Ga]Ga-NOTA-C25 PET can accurately monitor LAG-3+ TIL upregulation during immunotherapy, we subsequently analyzed its value in estimating the immunotherapy response and survival benefit in lung cancer. Tumor volume changes and survival (excluding animal death or animal condition beyond the ethical standard) during treatment were recorded for the aforementioned treatment groups (Fig. 6A; Suppl. Fig. 10A). Tumor volume increased significantly faster in the PBS and αPD-1 groups than in the MSA-2 and MSA-2 + αPD-1 groups. Importantly, these differences in tumor size were detectable from day 6, whereas differences in tumor uptake measured by [68Ga]Ga-NOTA-C25 PET could be discerned as earlier as day 4. These results showed that LAG-3 PET can monitor the early response to immunotherapy before tumor size changes. The MSA-2 + αPD-1 group exhibited the best survival with the longest time, more than 47 d, significantly longer than those of the PBS group (P < 0.01) and the αPD-1 group (P < 0.05) (Fig. 6B). Similar trends were observed in the MSA-2 group. There was no significant weight loss during treatment (Suppl. Fig. 10B). In addition, we confirmed that the dosages of the probe and blocking C25 could not induce immunostimulatory effects (Suppl. Fig. 11). Overall, these findings suggested that LAG-3 PET was a promising molecular imaging approach for monitoring immunotherapy in lung cancer by visualizing the immunophenotyping transition in tumors from immune “cold” to immune “hot.”
(A) Tumor growth curves of LLC tumor–bearing mice treated with PBS, αPD-1, MSA-2, and αPD-1 + MSA-2. (B) Survival analysis of LLC models for different groups. n ≥ 6. *P < 0.05. **P < 0.01. ****P < 0.0001.
LAG-3 PET in Immune System–Humanized NSCLC
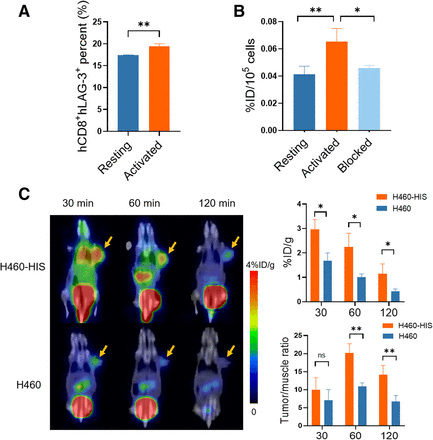
Stimulation-activated hPBMCs expressed higher hCD8+ hLAG-3+ than resting hPBMCs (P < 0.01) (Suppl. Fig. 12; Fig. 7A). Consistently, activated hPBMCs exhibited higher [68Ga]Ga-NOTA-C25 uptake than resting hPBMCs (P < 0.01) (Fig. 7B) and could be specifically blocked by nonradiolabeled C25 (P < 0.05). Next, we assessed the in vivo targeting of [68Ga]Ga-NOTA-C25 to hLAG-3+ TILs in human NSCLC H460 tumor–bearing mice established with a humanized immune system (H460-HIS), using immunoincompetent H460 models as controls. The establishment of the H460-HIS and H460 models was verified by flow cytometry analysis (Suppl. Fig. 13) as previously reported (24). Representative PET/CT images and quantitative analysis (Fig. 7C) showed that H460-HIS tumors exhibited higher tumor uptake than H460 tumors (P < 0.05). H460-HIS showed the highest tumor/muscle ratio at 60 min (20.23 ± 2.05), significantly higher than that of H460 (10.98 ± 0.75) (P < 0.01). H460 also showed lower background radioactivity than H460-HIS due to the lack of hLAG-3+ TILs. These results further indicated the accuracy of [68Ga]Ga-NOTA-C25 PET in detecting hLAG-3+ TILs.
(A) Quantitative analysis of flow cytometry for hCD8+hLAG-3+ in hPBMCs. (B) Cell uptake and blocking assays (nonradiolabeled C25, 9.613 × 10−6 μM) of [68Ga]Ga-NOTA-C25 in hPBMCS. (C) Representative PET/CT images and quantitative analysis in H460 and H460-HIS tumor models at 60 min after injection of [68Ga]Ga-NOTA-C25. Yellow arrows indicate tumor. n = 3. *P < 0.05. **P < 0.01.
Consistent with the PET results, hLAG-3+ TIL levels were significantly higher in H460-HIS tumors than in H460 tumors, as detected by flow cytometry and immunostaining (Suppl. Fig. 14). In the biodistribution analysis (Suppl. Table 3), H460-HIS tumors also exhibited higher tumor uptake than H460 tumors (P < 0.05). Similar trends were observed in major lymphoid organs. These results indicated the excellent performance of [68Ga]Ga-NOTA-C25 PET in noninvasively detecting hLAG-3+ TILs and highlighted the clinical translational potential of [68Ga]Ga-NOTA-C25 PET for monitoring immunotherapy in lung cancer patients.
DISCUSSION
Under immunosurveillance and selective pressure of the host immune system, tumor cells experience a transition from elimination to equilibrium and, finally, to immune escape (26,27). Immunotherapy strategies have been developed to overcome the escape mechanism and reactivate the host immune system to eliminate tumor cells; these include ICIs, cancer vaccines, and adoptive immune cell therapy. Among these, ICIs have raised the most research interest over the past decade, leading to significant advances in the field (6,28). To date, 8 ICIs have been approved by the Food and Drug Administration; these include the first approved LAG-3 ICI in combination with nivolumab (11,29). However, a low response rate was reported for all ICIs (30,31). Thus, the development of approaches for the early identification of responders in the precise management of immunotherapy is critical. Studies showed that molecular imaging is an excellent tool for noninvasively monitoring the immunotherapy response (32). Since LAG-3 is a promising next-generation immune checkpoint, when effective immunotherapies relieve immunosuppression and expose tumor antigens to the immune system, LAG-3 may upregulate specifically in TILs, particularly in CD8+ T cells (7,10,33); therefore, it may be an ideal molecular imaging biomarker for monitoring the early immunotherapy response. Compared with previously reported LAG-3 single-domain antibody–based micro-SPECT imaging (34), in the present study, we reported a LAG-3–specific peptide-based PET agent and acquired clear images with a clinical scanner in both mice and humanized mouse models. In particular, we verified the feasibility of LAG-3 PET for monitoring the early response to immunotherapy in lung cancer, providing more evidence that LAG-3 served as a molecular imaging marker in cancer immunotherapy. More recently, exciting advancement in the clinical translation of LAG-3 imaging has been made; a LAG-3 antibody–based PET agent, [89Zr]Zr-BI-754111, also showed favorable performance for monitoring immune infiltration in patients after PD-1 treatment (35). However, peptide probes are superior to antibodies as diagnostic agents, offering rapid imaging and clearance, favorable pharmacokinetics, and low cost (22).
Studies have shown that a STING agonist can increase the immunotherapy response in solid tumors by enhancing immune infiltration, particularly in immune “cold” tumors (36–38). Hence, in the present study, LLC tumors infiltrated with low levels of LAG-3+ TILs were selected for STING agonist–based immunotherapy. Our results showed that MSA-2–based immunotherapy was effective; importantly, [68Ga]Ga-NOTA-C25 PET could detect a tumor immunophenotyping transition from “cold” to “hot”—an early immunotherapy response—before tumor size changes. In addition, animals carrying LAG-3 PET–detected “hot” tumors exhibited significant tumor growth retardation and longer survival. In contradistinction, “cold” tumors progressed rapidly, with shorter survival. Above all, our results demonstrated that LAG-3 PET is reliable for monitoring the early immunotherapy response in lung cancer. However, we noted that lung uptake of [68Ga]Ga-NOTA-C25 could be blocked in both PET and biodistribution analyses; this condition might have been caused by immune cells residing in the lungs, recruited immune cells, and a complex immune microenvironment.
Rapid tumor accumulation and background clearance of [68Ga]Ga-NOTA-C25 promoted optimal PET imaging. However, longitudinal studies may need to focus on improving the specificity of the peptide-based probe. The fact that high-resolution small-animal PET/CT was not used here may have limited the imaging of smaller organs, such as TDLN. However, clear images obtained from clinical PET/CT with wide fields of view were undoubtedly beneficial for clinical translation. In addition, LAG-3 PET may also play a significant role in guiding the application of Food and Drug Administration–approved LAG-3–based combination therapy for cancer treatment.
CONCLUSION
[68Ga]Ga-NOTA-C25 PET is a reliable approach to noninvasively detecting LAG-3+ TILs with excellent clinical translation potential. [68Ga]Ga-NOTA-C25 PET can visualize the tumor immunophenotyping transition from “cold” to “hot” under effective immunotherapy and may be used to stratify lung cancer patients for the early identification of immunotherapy responders.
KEY POINTS
QUESTION: Can LAG-3 PET be used to monitor the early response to immunotherapy in lung cancer?
PERTINENT FINDINGS: [68Ga]Ga-NOTA-C25 PET could detect a tumor immunophenotyping transition during immunotherapy by targeting LAG-3+ TILs and was a reliable approach for monitoring the early treatment response before tumor size changes.
IMPLICATIONS FOR PATIENT CARE: [68Ga]Ga-NOTA-C25 PET showed excellent clinical translation potential and is a promising noninvasive imaging method for monitoring the immunotherapy response to provide precise management of immunotherapy in lung cancer patients.
DISCLOSURE
Financial support was provided by National Natural Science Foundation of China (81627901, 82102101, 82202226), Natural Science Foundation of Heilongjiang Province (JQ2020H002, JJ2022LH0959), HMU Marshal Initiative Funding (HMUMIF-21003), Tou-Yan Innovation Team Program of Heilongjiang Province (2019-15), and Heilongjiang Provincial Key Laboratory of Molecular Imaging Foundation. No other potential conflict of interest relevant to this article was reported.
ACKNOWLEDGMENT
Some graphic elements were provided by Figdraw with permission (WWSWO8cfbf).
Footnotes
↵* Contributed equally to this work.
Published online Nov. 16, 2023.
- © 2024 by the Society of Nuclear Medicine and Molecular Imaging.
REFERENCES
- Received for publication May 7, 2023.
- Revision received October 14, 2023.