Abstract
Integrin αvβ3 has been proposed as a potential imaging target for radiolabeled RGD peptides and a molecular marker for the estimation of tumor angiogenesis, yet it has not been applied in differentiated thyroid cancer (DTC) patients with radioactive iodine–refractory (RAIR) lesions. The current study was conducted to assess the potential of integrin αvβ3 imaging in the detection of RAIR DTC lesions using 99mTc-PEG4-E[PEG4-c(RGDfK)]2 (99mTc-3PRGD2), thus providing a feasible antiangiogenetic therapeutic target. Methods: Ten DTC patients (2 men, 8 women; mean age ± SD, 56.4 ± 9.8 y; age range, 42–73 y) with multiple RAIR metastases were recruited; all patients had both elevated thyroglobulin levels (thyroglobulin-positive) and negative 131I whole-body scan (WBS) results. Clinical data were collected including history, 131I WBS, contemporary CT, ultrasonography, thyroid-stimulating hormone, thyroglobulin, and antithyroglobulin. One or 2 target lesions were selected on the contemporary CT images using Response Evaluation Criteria in Solid Tumors 1.0 for all patients, 7 of whom were chosen for the calculation of the rates of lesion growth within the 3 mo before the study. WBS at 30 min and regional SPECT for lesions at 1 h were performed after the intravenous injection of 99mTc-3PRGD2. Two experienced nuclear medicine physicians read the images in a masked fashion. The tumor-to-background ratios were calculated for further analysis. Results: All the target RAIR metastatic lesions were identified as positive on 99mTc-3PRGD2 SPECT images. There was a significant correlation between the mean tumor-to-background ratios and mean growth rates of target lesions (r = 0.878, P = 0.009). Conclusion: The RAIR (131I WBS–negative/thyroglobulin-positive) metastatic lesions can be traced using 99mTc-3PRGD2 imaging, meaning these lesions are highly neovascularized. 99mTc-3PRGD2 angiogenesis imaging can be used for the localization and growth evaluation of RAIR lesions, providing a new therapeutic target and a novel imaging modality to monitor the efficacy of certain antiangiogenetic therapy.
It is well known that differentiated thyroid cancer (DTC) includes papillary thyroid cancer and follicular thyroid cancer. Most DTC patients can survive disease-free for more than 30 y with appropriate surgery and radioactive iodine therapy. However, 2%–5% of these tumors will lose their differentiated phenotypes, develop a recurrent lack of avidity for radioactive iodine, and lead to the failure of 131I treatment (1). These cases of DTC are commonly called radioactive iodine–refractory (RAIR) DTC, with negative 131I whole-body scan results and elevated unstimulated thyroglobulin (>2.0 ng/mL, thyroglobulin-positive), indicating dedifferentiation of those lesions. RAIR DTC is prone to be more aggressive and metastatic, rendering cells that are more malignant and refractory, with a median patient survival of less than 5 y (2). Cancer guidelines of the American Thyroid Association recommend the use of 18F-FDG PET/CT for the detection of RAIR DTC lesions. However, 18F-FDG trapping is not tumor-specific, and at present in China most patients cannot afford the expense of 18F-FDG PET for diagnosis, rather than treatment. A new cost-effective method for early detection and personalized therapy is highly desirable for these patients.
Integrin αvβ3 has been confirmed in various malignant tumors with high density, and it is essential for cell migration and invasion and plays an important role in tumor angiogenesis (3). Integrin αvβ3 expression is high on the surface of activated endothelial cells in newly formed blood vessels bit is low in both resting endothelial cells and most normal organ systems, thus representing an interesting molecular marker for angiogenesis imaging (4). The adhesion of endothelial cells and extracellular matrix, mediated by integrin αvβ3, is vital for vascular endothelial growth factor receptor-2 activation (5). Ligands bearing RGD (Arg-Gly-Asp) peptide have a high affinity and specificity for integrin αvβ3, especially cyclic RGD dimmers with PEG4 linkers (6). Unlike 18F-FDG PET/CT as a diagnosis-only modality, integrin αvβ3 imaging by radiolabeled RGD peptide provides a specific method for visualizing tumor angiogenesis and a therapeutic target for antiangiogenetic and antiintegrin drugs (7).
The RGD molecules 99mTc-NC100692, 18F-P-PRGD2, 18F-AH111585, and 18F-galacto-RGD have already been used in the molecular imaging of cancer cell lines, animal models, or clinical cancer patients (including in murine osteosarcoma, human colon and renal adenocarcinoma, rat pancreatic tumors, ovarian carcinoma, human breast carcinoma, human glioblastoma, and human melanoma) to trace tumor angiogenesis and metastasis with PET or SPECT (8–13). The integrin αvβ3 expression level on tumor cell membranes correlates well with the RGD radiotracer tumor uptake on scintigraphy imaging (14). Compared with the 18F-labeled tracers for PET, 99mTc-labeled nuclides are SPECT tracers that hold the advantage of being more broadly available. Moreover, the product can be kit-formulated and ready for routine clinical use with a simple, convenient labeling procedure and easy quality control. 99mTc-PEG4-E[PEG4-c(RGDfK)]2 (99mTc-3PRGD2) is a new 99mTc-labeled cyclic RGD dimer peptide with increased receptor binding affinity and improved kinetics for the in vivo imaging of integrin αvβ3 expression in xenografted tumor–bearing models (6,15,16). The blood clearance kinetics, biodistribution, and radiation dosimetry of a kit-formulated 99mTc-3PRGD2 were also evaluated in nonhuman primates in our department, and no adverse reactions were observed to date (17). A multicenter study of 99mTc-3PRGD2 for integrin receptor imaging of lung cancer was sponsored by our department last year. To date, RGD molecular imaging including 99mTc-3PRGD2 has not been used in thyroid cancer, in particular 131I WBS–negative/thyroglobulin-positive patients. This study provides clinical integrin imaging data for RAIR DTC.
MATERIALS AND METHODS
Study Design and Patient Recruitment
The study protocol was approved by the ethics committee of Chinese Academy of Medical Sciences and Peking Union Medical College Hospital (PUMCH) and by the institutional review boards of the PUMCH. Written informed consent was obtained from all subjects. Subject inclusion was based on the following presumptive diagnosis criteria: age over 18 y; a negative pregnancy test; clinically acceptable renal and hepatic function (as judged by an investigator according to blood examinations); and RAIR DTC patients with metastatic lesions, treated by radioactive iodine many times, who were 131I WBS–negative/thyroglobulin-positive. In addition, other findings from clinical staging procedures served as the standard of reference (including anti-thyroglobulin levels, 18FDG PET/CT, bone scintigraphy, contemporary CT, and ultrasonography). Because of ethical concerns, biopsies and histopathology were not available for most of the analyzed metastases. The exclusion criteria included 131I WBS positivity, thyroglobulin negativity, pregnancy, lactation, and impaired renal or liver function.
Ten RAIR DTC patients (2 men, 8 women; mean age ± SD, 56.4 ± 9.8 y; age range, 42–73 y; median body weight ± SD, 65.7 ± 12.8 kg [weight range, 44.0–87.5 kg]) were recruited from the Nuclear Medicine Department of PUMCH.
The protocol outlined admission, dosing, and safety checks on the same day up to 2.5 h after the administration of 99mTc-3PRGD2. Imaging and safety assessments were performed during the first 2.5 h after injection, after which the subjects were allowed to leave the site.
Radiosynthesis of 99mTc(Hydrazinonicotinamide-3PRGD2)(Tricine) (TPPTS) and Agent Injection
All chemicals obtained commercially were of analytic grade and used without further purification. Succinic acid, trisodium triphenylphosphine-3,3′,3″-trisulfonate (TPPTS), and tricine were purchased from Sigma-Aldrich. Cyclic RGD peptide dimer 3PRGD2 was custom-made by Peptides International, Inc. Na99mTcO4 was obtained from a commercial 99Mo/99mTc generator (Beijing Atom High Tech Co., Ltd.). Synthesis of the labeling precursor, kit preparation, and subsequent 99mTc labeling were performed as previously described (17). Briefly, the kit for preparation of 99mTc-3PRGD2 was formulated by combining 20 μg of hydrazinonicotinamide-3PRGD2, 5 mg of TPPTS, 6.5 mg of tricine, 40 mg of mannitol, 38.5 mg of disodium succinate hexahydrate, and 12.7 mg of succinic acid. For 99mTc radiolabeling, the kit vial was added to 1 mL of 99mTcO4-saline solution (1,110–1,850 MBq [30–50 mCi]) and then water-bathed at 100°C for 20 min. The resulting solution was analyzed by instant thin-layer chromatography using Gelman Sciences silica-gel paper strips and a 1:1 mixture of acetone and saline as eluant. For clinical use, the radiochemical purity was always greater than 95%. The reaction mixture was then diluted to 370 MBq (10 mCi) per milliliter with saline and filtered with a 0.20-μm Millex-LG filter (EMD Millipore). Each patient was injected with 99mTc-3PRGD2 (11.1 MBq/kg [0.3 mCi/kg]). Before imaging, all the subjects were asked to urinate completely.
Imaging Protocols
With the patients’ arms laid beside their body, anterior and posterior planar scans of the whole body were started at 30 min after injection, using a dual-head γ-camera with low-energy high-resolution collimators and a 20% energy acceptance window centered on 140 keV. SPECT and coregistered spiral CT were performed at 1 h after injection with the same γ-camera. The speed of whole-body scanning was set at 10 cm/min, followed by SPECT (zoom, ×1; 60 s/frame/12°; 30 steps/360°) with the patients’ arms raised above their head. The images were obtained using the optimized parameters of the Philips Precedence SPECT system. The DICOM image files of each patient were saved and transferred to Extended Brilliance workspace NM1.0 (Philips) for centralized reconstruction, reading, and analysis.
Image Analysis
The 99mTc-3PRGD2 images were evaluated by 2 experienced nuclear medicine physicians through consensus reading masked to the source, history, and pathologic conditions of the patients. For analysis, 1 or 2 target lesions were selected and measured by the Response Evaluation Criteria in Solid Tumors (RECIST) 1.0 (18). The total number of selected target lesions was 18, and they were all negative on 131I WBS. The variations of lesion sizes were documented, and the mean growth rates of target lesions for 7 patients in the 3 mo before the study were calculated and recorded (dynamic follow-up of CT images in the past 3 mo were not available for the other 3 patients). Tumor-to-background (T/B) ratios of target lesions on SPECT were measured and calculated by the same person using a consistent standard. Briefly, on the section on which target lesions were measured according to RECIST 1.0, regions of interest (ROI) were drawn on the entire target lesions on SPECT cross-sections. And on the same section, a blood-pool background ROI with a diameter of 20 mm was set over the contralateral normal lung tissue or surrounding normal soft tissue. The T/B ratios were calculated by mean counts of the tumor ROI divided by mean counts of the background ROI.
Statistics Analysis
All quantitative data were expressed as mean ± SD. The correlation between mean T/B ratios and mean growth rates of the target lesions was evaluated by linear regression analysis and by calculation of the Pearson correlation coefficient r. Statistical significance was tested using SPSS (version 19.0; IBM) at the level of 0.01.
RESULTS
Distribution of 99mTc-3PRGD2 in Human Body
The image quality of 99mTc-3PRGD2 scans was good for all 10 enrolled patients. The distribution of 99mTc-3PRGD2 in the human body could be observed clearly on the anterior and posterior whole-body images (Fig. 1C). Except for the intense accumulation in the kidneys and bladder corresponding to the main excretion pathway, there was moderate 99mTc-3PRGD2 uptake in the liver, spleen, intestines, nasopharyngeal region, and bone marrow. Because pulmonary metastases account for more than 50% of thyroid cancer metastases (19), the low background in the lungs and mediastinum allowed for easy discrimination of thoracic lesions.
Elevated levels of unstimulated thyroglobulin (ng/mL) and antithyroglobulin (IU/mL) with corresponding TSH (A), 131I-negative image with TSH stimulation (TSH > 30 μIU/mL) (B), and RGD-positive planar (C) and SPECT images (D). Tg = thyroglobulin.
Evaluation for 99mTc-3PRGD2 Imaging of Target Metastatic RAIR Lesions in DTC Patients
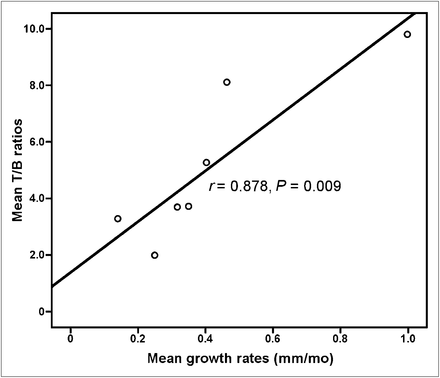
The characteristics of enrolled patients are shown in Table 1. All 18 selected target lesions were identified on SPECT images with the prescribed 99mTc-3PRGD2 administration imaging protocol. The mean T/B ratio of all 18 target lesions was 5.2 ± 2.8. A statistically significant correlation was found between the mean T/B ratios and mean growth rates of target lesions, with a correlation coefficient of 0.878 (P = 0.009) (Fig. 2). Figure 1 illustrates the variations of TSH, thyroglobulin, and antithyroglobulin levels; negative 131I WBS results with TSH stimulation; and positive 99mTc-3PRGD2 planar and SPECT images of RAIR lesions in patient 1.
Patient Characteristics (n = 10)
Correlation of mean T/B ratios and mean growth rates. Statistically significant correlation is found between mean T/B ratios and mean growth rates of target lesions, with correlation coefficient of 0.878 (P = 0.009).
DISCUSSION
The management of RAIR DTC metastatic lesions has caused a dilemma. Surgery is not eligible for multiple metastases. Neither 131I WBS for early detection nor radioactive iodine for effective therapy works because RAIR lesions usually lose the ability to concentrate 131I. RAIR cancer cells proliferate quickly, leading to active metabolism and the lack of oxygen, with vigorous angiogenesis and higher expression of glut protein on the cell membrane (20). 18F-FDG PET/CT, an imaging technique based on the high expression of glut protein, provides unique information about the molecular and glucose metabolic changes associated with DTC. The guidelines of the American Thyroid Association recommend that if an empiric dose (3.7–7.4 GBq [100–200 mCi]) of radioactive iodine fails to localize the persistent disease, 18F-FDG PET/CT should be considered, especially in DTC patients with stimulated serum thyroglobulin levels greater than 10–20 ng/mL or in those with aggressive histologies, to localize metastatic lesions that may require treatment or continued close observation (21). However, it is important to realize that 18F-FDG trapping is not tumor-specific. Both malignant thyroid tumors and some benign thyroid diseases, such as Graves disease and hyperactive thyroid adenoma, can take up 18F-FDG. Moreover, cervical lymph nodes with inflammation or other inflamed tissues could show high 18F-FDG uptake on the PET scans (22). At present in China, SPECT is more widely available than PET. Radiotracers for SPECT are easier to prepare and usually have a longer half-life than those for PET. Most patients cannot afford the expense of 18F-FDG PET for diagnosis, rather than treatment. A new SPECT agent with a therapeutic target is urgently needed both for diagnosing and for guiding further treatment.
99mTc-3PRGD2, a novel specific radiotracer for targeting integrin αvβ3, has been selected as a candidate for clinical evaluations because of its high uptake, long retention time, and high metabolic stability in athymic nude mice bearing U87MG gliomas and MDA-MB-435 breast cancer xenografts (6,15). 99mTc-3PRGD2 has recently been translated into clinical trials following its preliminary applications and several other RGD-based tracers’ applications in preclinical experiments and clinical trial (10–12,15). A multicenter study of 99mTc-3PRGD2 for integrin receptor imaging of lung cancer was organized by our department last year, and results indicated that 99mTc-3PRGD2 imaging at 1 h was sensitive enough for the detection of lung cancer, with a sensitivity of 88% for semiquantitative analysis that could reach 93%–97% in visual analysis when considering the volume effect, necrosis, and metastasis (23). The present study is the first application of RGD imaging in DTC patients with RAIR lesions. On the planar images, the distribution of 99mTc-3PRGD2 in the human body could be observed clearly, presenting the same results as those in lung cancer patients. Except for the intense accumulation in the kidneys and bladder corresponding to the main excretion pathway, there was moderate 99mTc-3PRGD2 uptake in the liver, spleen, intestines, nasopharyngeal region, and bone marrow. On the SPECT images, all target lesions could be identified as positive, with various higher uptake of 99mTc-3PRGD2 than blood-pool background, contrary to their negative uptake demonstrated by 131I WBS. The mean T/B ratio of all target lesions was 5.2 ± 2.8. To reflect the recent status of tumor growth, the mean growth rates of target lesions in 7 patients in the 3 mo before the study were calculated and recorded (dynamic follow-up of CT images in the past 3 mo were not available for the other 3 patients). The mean T/B ratios of target lesions escalated as the mean growth rates increased, and significant correlation was found between the two (r = 0.878, P = 0.009) (Fig. 2). The correlation suggests that active angiogenesis with higher integrin αvβ3 density on RAIR DTC cells and tumor neovasculature is needed for rapid growth of tumor lesions. The higher the 99mTc-3PRGD2 uptake is in target lesions, the more rapid is the tumor growth, and vice versa. 99mTc-3PRGD2 also holds promise for monitoring response to antiangiogenic therapy, as demonstrated by other RGD molecules (7,24).
Further analysis illustrates that all the lesions larger than 1 cm were observed clearly on 99mTc-3PRGD2 SPECT images. Among these lesions, some presented higher uptake whereas others presented relatively lower uptake. Not all of the lesions smaller than 1 cm were detected. The major cause of this phenomenon is the heterogeneous property of the tumor itself; however, partial-volume effects in small lesions may be another reason. Six of 10 patients had undergone an 18F-FDG PET/CT scan. Integrin imaging was in accordance with 18F-FDG uptake in almost all of the individual metastatic lesions larger than 1 cm (which is a limitation of SPECT because of poor resolution). The metastatic RAIR DTC lesions can have both a high glucose metabolic rate and vigorous angiogenesis. The comparison of 99mTc-3PRGD2 with 18F-FDG PET/CT and further monitoring of its therapeutic efficacy are in progress.
CONCLUSION
In this study, significant uptake of 99mTc-3PRGD2 could be observed in RAIR DTC lesions, contrary to their negative 131I WBS results. A correlation between the mean T/B ratios and mean growth rates of lesions was confirmed, indicating active angiogenesis and malignant phenotypes of RAIR DTC. 99mTc-3PRGD2 with SPECT is a promising modality for diagnosing and guiding further treatment of RAIR DTC.
DISCLOSURE STATEMENT
The costs of publication of this article were defrayed in part by the payment of page charges. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
No potential conflict of interest relevant to this article was reported.
Footnotes
Published online Oct. 15, 2012.
- © 2012 by the Society of Nuclear Medicine and Molecular Imaging, Inc.
REFERENCES
- Received for publication April 26, 2012.
- Accepted for publication July 20, 2012.